Journal of Gynecological Research and Obstetrics
Xiphopagic conjoined twins: A case report with pre and postnatal imaging
Delta Hospital CHIREC Brussels, Belgium
Author and article information
Cite this as
Jottrand Z, Geeraerd F, D’Hondt A, Donner C, Avni FA (2023) Xiphopagic conjoined twins: A case report with pre and postnatal imaging. J Gynecol Res Obstet. 2023; 9(1): 3-6. Available from: 10.17352/jgro.000119
Copyright License
© 2023 Jottrand Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Siamese or conjoined twins represent a rare but specific malformation occurring in monozygotic and mono-amniotic twin pregnancies. We report a case of xiphopagic conjoined twins and compare the contribution and correlation between ante- and post-mortem imaging for the detailed evaluation of the malformation. Our case highlights the importance of postnatal imaging to better understand this complex type of fetal malformation.
Siamese or conjoined twins (CT) are a very rare developmental anomaly resulting from the incomplete separation of embryonic button cells at the blastocyst stage [1]. The etiology remains poorly understood; two theories have been developed: the partial fusion of the two primitive lines of two embryos or an incomplete division of an embryo after the 14th day post-fertilization. CT has always the same gender.
The prevalence of CT is rare and is explained by the rarity of twin pregnancies (2% of pregnancies) and monozygotic (35% of twin pregnancies). CT represents one in 50,000 to 100,000 births [1,2] of which 65% are stillborn, and 35% of those born alive will die in the first 24 hours. Only 25% will survive up to an age when surgical separation can be considered [3,4].
Several classifications have been proposed; they are based on the most significant joining site as well as common organs and symmetry [5]. One of them classified CT according to the site of fusion and was always joined at identical anatomy points as described in Table 1 [3].
Cases of CT have been observed and reported for many years [6]. The first antenatal diagnosis of a CT (at 35 GW) was reported by Wilson and al in 1976 [6]; he showed the importance of early diagnosis, establishing the site of attachment and describing associated malformations. To date, advances in medical imaging allow an earlier and quite precise diagnosis. This helps to specify the classification according to the site of union and common organs. An early antenatal diagnosis is essential to optimize a multidisciplinary approach considering both the pregnant mother and her fetus taking into account the expectations of the parents.
Case report
We report the case of a second pregnancy in a 27-year-old patient. The couple was non-consanguineous. Their first child was born without any anomaly in 2019. The patient suffered from iron deficiency anemia and hypothyroidism, both treated during pregnancy. There were no previous twin pregnancies in their respective families.
The first-trimester ultrasound was performed at 14 WG and revealed a mono-chorionic, mono-amniotic twin pregnancy, with joint embryos apparently omphalo - and thoracopagus. At the end of this ultrasound, the diagnosis as well as its poor prognosis were explained to the parents. They were referred to our fetal medicine unit and met with members of the neonatology team, a pediatric surgeon, and a pediatric cardiologist. All confirmed the severity of the anomaly. A significant language barrier complicated significantly the discussions. The parents decided to continue the pregnancy. The main reason was religion as well as hope that at least one of the 2 twins would survive.
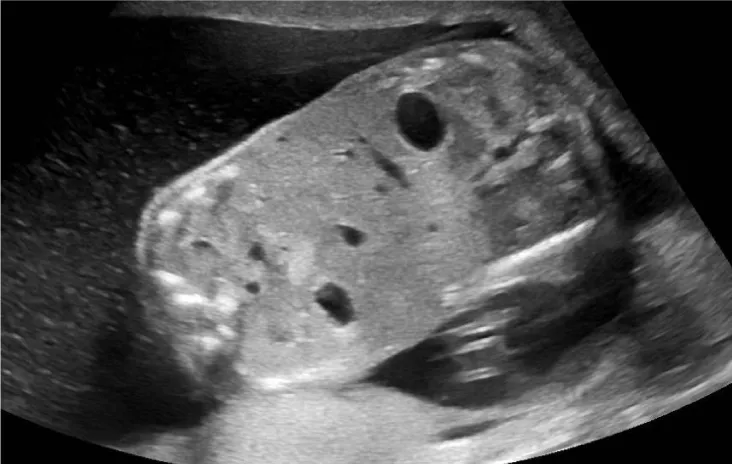
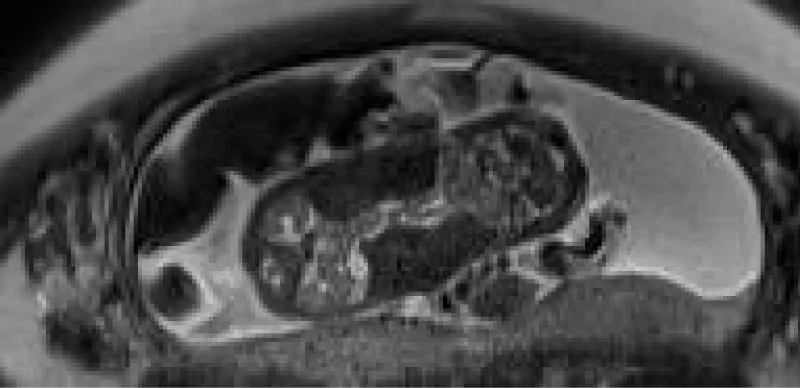
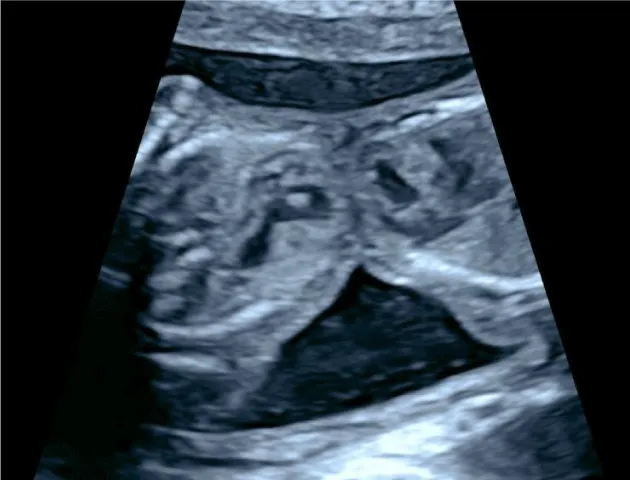
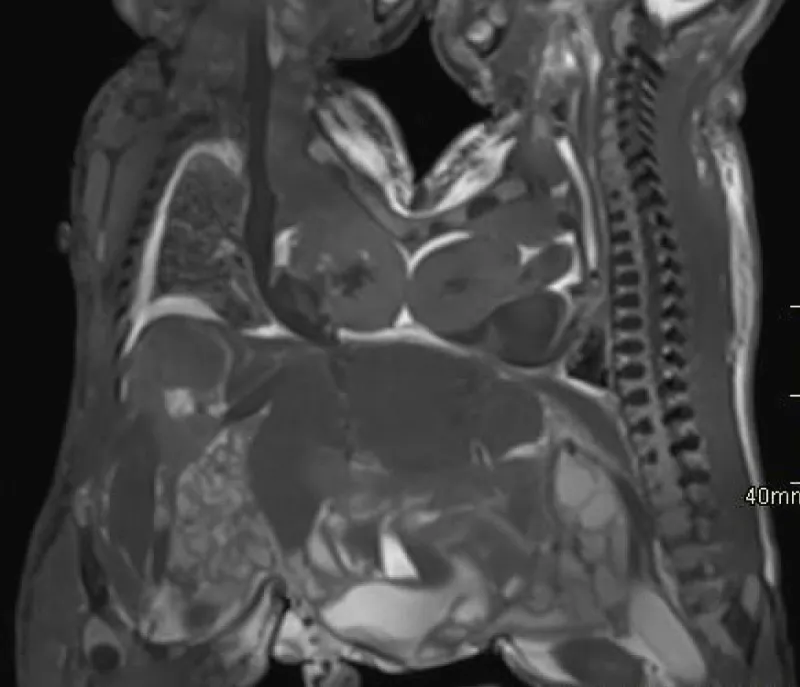
Given the parents’ decision to continue the pregnancy, multiple imaging tests were performed to further clarify the nature of the malformations and to bring additional information to the parents. A fetal ultrasound and MRI were performed at 20 WG confirming the presence of 2 females CT, xiphopagic having the liver (Figure 1) and some parts of the small bowel loops in common (Figure 2). Both fetuses appeared to have congenital heart malformation (Figure 3). Bilateral univentricular heart was confirmed by a fetal cardiologist. All the anomalies are described in Table 2.
Further multidisciplinary discussions were organized to confirm the severity of the malformation. They were explained to the parents insisting on the additional risk for the mother in case of a cesarean section later in pregnancy. The parents agreed on delivery around 28 GW with palliative care for the CT. The cesarean section was performed at 29 GW. The female CTs weighed around 1320g each. They were united by the lower chests and upper abdomens. Their Apgar scores were 4, 2, 1 at 1, 5, and 10’. Both babies died due to respiratory distress after one hour. Post-mortem imaging tests were performed.
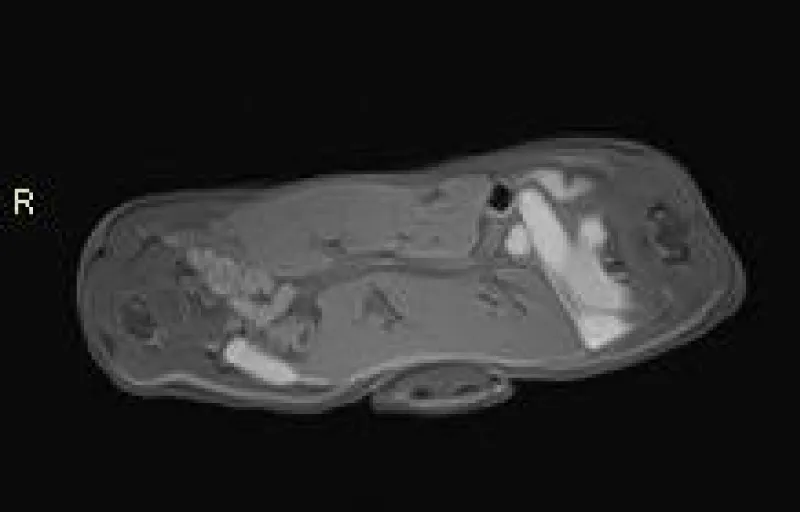
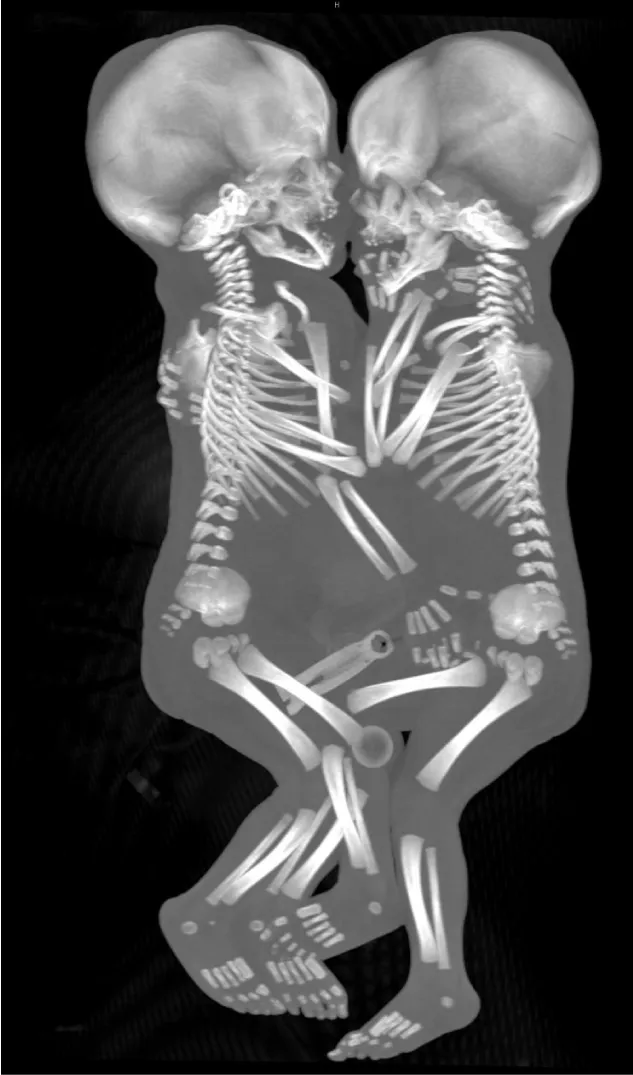
The post-mortem MRI and CT confirmed the known data, namely, a common liver, intestinal loops crossing the midline, and two univentricular hearts (Figures 4,5). Furthermore, these examinations revealed a fusion of the two lower sternums on the midline and a common diaphragm (Figures 5,6). This may explain partially the postnatal respiratory distress breathing. Partial fusion of both hearts was also suspected. Finally, close contact between the hearts rendered it impossible to verify whether they are completely separated. A 3D bone reconstruction by a CT scanner made it possible to visualize the skeleton of these twins (Figure 6).
Discussion
CT is a rare condition and represents a challenge to radiologists, obstetricians, neonatologists, and surgeons. The challenge for prenatal imaging is to determine the exact anatomy of the 2 fetuses: common organs, any malformations, and the presence of cross-circulation in order to assess the prognosis, neonatal management, and the possibility of a postnatal surgery [7].
Presently, the diagnosis of conjoined twins is possible as early as the first-trimester ultrasound, as it was in our case [8].
As recommended by the literature, our patient was monitored closely: several ultrasound examinations were performed as well as a fetal MRI (at 20 WG). In our case, fetal MRI confirmed the abnormalities detected on ultrasound. Fetal MRI provided additional information, on common small bowel loops. These data are in accordance with the conclusions of Spielmann and al that fetal MRI is an excellent complement to prenatal US, particularly in cases of complex fetal anomalies [9].
Post-mortem MRI (PMMRI) and post-mortem CT provided additional information as they identified the presence of a fusion of the lower part of both sternums as well as a common diaphragm. On the other hand, it could not determine the complete separation of the univentricular hearts. PMMRI has low diagnostic performance in diagnosing fetal cardiac malformation, especially in small gestational-age fetuses [10].
Therefore, both pre-and post-natal imaging is essential for mapping the abnormalities and thus allows clinicians and surgeons to establish a prognosis /outcome of the pregnancy and the possibility of postnatal surgery [11].
Conclusion
Both prenatal and postnatal imaging plays a key role in the assessment of abnormalities and common organs in conjoined twins and is valuable tools for prognostic assessment of pre- and neonatal management of CT fetuses. In our case, the complete and extensive assessment that was carried out allowed us to establish a precise mapping of the anomalies present in these Siamese in order to give a fairly clear prognosis to the parents.
We would like to thank all the gynecologists (especially Dr. Cayphas), neonatologists, and other caregivers who provided the best possible multidisciplinary management of this difficult case.
- Durier M, Vervaet H, Gabriel R. Multiple pregnancies. Anatomoclinical study and management. EMC – Consulte. 2010; 5(2):1-11.
- Tongsong T, Chanprapaph P, Pongsatha S. First-trimester diagnosis of conjoined twins: a report of three cases. Ultrasound Obstet Gynecol. 1999 Dec;14(6):434-7. doi: 10.1046/j.1469-0705.1999.14060434.x. PMID: 10658285.
- Winkler N, Kennedy A, Byrne J, Woodward P. The imaging spectrum of conjoined twins. Ultrasound Q. 2008 Dec;24(4):249-55. doi: 10.1097/RUQ.0b013e31818c8858. PMID: 19060714.
- Filler RM. Conjoined twins and their separation. Semin Perinatol. 1986;10(1):82–91
- Spencer R. Anatomic description of conjoined twins: a plea for standardized terminology. J Pediatr Surg. 1996 Jul;31(7):941-4. doi: 10.1016/s0022-3468(96)90417-0. PMID: 8811563.
- Wilson RL, Cetrulo CL, Shaub MS. The prepartum diagnosis of conjoined twins by the use of diagnostic ultrasound. Am J Obstet Gynecol. 1976 Nov 15;126(6):737. doi: 10.1016/0002-9378(76)90536-6. PMID: 984156.
- Spitz L, Kiely EM. Experience in the management of conjoined twins. Br J Surg. 2002 Sep;89(9):1188-92. doi: 10.1046/j.1365-2168.2002.02193.x. PMID: 12190687.
- Hill LM. The sonographic detection of early first-trimester conjoined twins. Prenat Diagn. 1997 Oct;17(10):961-3. PMID: 9358576.
- Spielmann AL, Freed KS, Spritzer CE. MRI of conjoined twins illustrating advances in fetal imaging. J Comput Assist Tomogr. 2001 Jan-Feb;25(1):88-90. doi: 10.1097/00004728-200101000-00015. PMID: 11176299.
- Thayyil S, Sebire NJ, Chitty LS, Wade A, Olsen O, Gunny RS, Offiah A, Saunders DE, Owens CM, Chong WK, Robertson NJ, Taylor AM. Post mortem magnetic resonance imaging in the fetus, infant and child: a comparative study with conventional autopsy (MaRIAS Protocol). BMC Pediatr. 2011 Dec 22;11:120. doi: 10.1186/1471-2431-11-120. PMID: 22192497; PMCID: PMC3259035.
- Mehollin-Ray AR. Prenatal and postnatal radiologic evaluation of conjoined twins. Semin Perinatol. 2018 Oct;42(6):369-380. doi: 10.1053/j.semperi.2018.07.015. Epub 2018 Jul 26. PMID: 30170825.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
