International Journal of Clinical Endocrinology and Metabolism
The Role of Estradiol in Reproduction and Health: Insights from Molecular Signaling to Hormone Replacement Strategies
1College of Animal Science and Technology, Nanjing Agriculture University, Nanjing, 210095, China
2Laboratory of Experimental Animal Disease, Mosel, College of Veterinary Medicine, Sichuan Agriculture University, Chengdu 611130, China
3State key Laboratory of Agricultural Microbiology, College of veterinary Medicine, Huazhong Agricultural university, Wuhan, china
4Department of environmental science and engineering, Yangzhou university, China
Author and article information
Cite this as
Bo Z, et al. The Role of Estradiol in Reproduction and Health: Insights from Molecular Signaling to Hormone Replacement Strategies. Int J Clin Endocrinol Metab. 2026; 12(1): 10-26. Available from: 10.17352/ijcem.000068
Copyright License
© 2026 Bo Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Estrogen, particularly Estradiol (E2), plays a fundamental role in female reproductive health, yet the molecular mechanisms underlying its actions and the consequences of its deficiency remain incompletely understood, a critical research gap this review seeks to address. Using a systematic evaluation of peer-reviewed experimental and clinical literature as the primary data source, this study examines estradiol’s roles in reproductive physiology, molecular signaling, and therapeutic intervention strategies. Estradiol, predominantly synthesized in the ovaries, governs follicular development, uterine maintenance, and menstrual cycle regulation via the Hypothalamic-Pituitary-Gonadal (HPG) axis. The Ovariectomy (OVX) animal model is assessed as the principal experimental approach for simulating postmenopausal estrogen deficiency, revealing downstream effects including osteoporosis, cardiovascular disease, and neurodegeneration. The tissue-specific distribution and signaling functions of three estrogen receptor subtypes, ERα, ERβ, and GPER, are analyzed through both genomic and non-genomic pathways, including MAPK/ERK and PI3K/AKT cascades. The efficacy, mechanisms, and limitations of Hormone Replacement Therapy (HRT) and Selective Estrogen Receptor Modulators (SERMs) are also critically evaluated. These findings carry significant implications for reproductive medicine, endocrinology, and the development of targeted therapies for estrogen-related disorders.
Reproduction is an important aspect for species survival, with significant implications for understanding fertility, hormonal regulation, and the pathophysiology of reproductive disorders [1]. Mice (Mus musculus) are highly considered one of the optimal models for research in biomedical studies, reproductive biotechnology, and veterinary research due to their physiological, nutritional, and reproductive similarities to humans. These similarities make them ideal models for scientific explorations [2.3]. Central to reproductive health is the role of estrogen, a key steroid hormone that plays an important role in various aspects of the female reproductive tract and is involved in various physiological processes such as the menstrual cycle, follicular development, ovulation, and the actions on the reproductive organs [4]. Estrogen, particularly its most active form, Estradiol (E2), is an essential hormone of the menstrual cycle, acting in conjunction with FSH and LH for the development of follicles, ovulation, and endometrium preparation for implantation [5]. However, the molecular mechanisms underlying estrogen's actions, especially in the context of estrogen deficiency, remain incompletely understood. This study seeks to address this gap by investigating the impact of estradiol and Ovariectomy (OVX) on reproductive health in mice, with a focus on unraveling the molecular underpinnings and interactive effects of these factors. This review also provides a comprehensive overview of estrogen’s function and its physiological significance in reproductive health. It also summarizes current therapeutic approaches, including Hormone Replacement Therapy (HRT) and Selective Estrogen Receptor Modulators (SERMs), and outlines future research prospects.
The role of estrogen in reproductive health
Estrogen, a sex steroid hormone, plays a key role in various aspects of the female reproductive tract and is involved in various physiological processes such as the menstrual cycle, follicular development, ovulation, and the actions on the reproductive organs [6]. Estrogen is one of the key female hormones that plays a critical role in integrating hormonal communication within the Hypothalamic-Pituitary-Gonadal (HPG) axis to modulate the relationship between the brain, pituitary gland, and ovaries for the proper regulation of female reproduction. Out of all the forms of estrogen, E2 is the most concentrated and the active one, and it carries out its actions through complex signaling mechanisms. Estrogen deficiency causes a variety of physiological alterations, which can be approximated with high fidelity using ovariectomy, the most common model of estrogen depletion that is vital in studying conditions associated with estrogen deficiency, including osteoporosis, cardiovascular diseases, metabolic disorders, and neurodegenerative diseases [7]. Estrogen is a significant hormone in reproductive physiology and has various actions at the molecular level and significant outcomes during its deficiency, which are discussed in this study with reference to reproductive physiology only [8]. Estrogen is an essential hormone of the menstrual cycle, acting in conjunction with FSH and LH for the development of follicles, ovulation, and endometrium preparation for implantation. In the follicular phase, the estrogen level increases because the developing ovarian follicles secrete estradiol in response to FSH stimulus [9]. This rise of estradiol has a negative feedback on the hypothalamus and pituitary to inhibit FSH hypersecretion while enhancing follicular development. However, during ovulation, a positive feedback leading to a rise in LH levels is observed due to high estrogen levels [10]. This LH surge is necessary for the bursting of the largest follicle and the subsequent release of an ovum. During the late phase of the menstrual cycle, estrogen and progesterone co-regulate endometrial receptiveness in order to provide a favorable environment for implantation. If fertilization does not take place, then estrogen and progesterone concentrations drop, which causes the break-off of the endometrial lining and the beginning of a new menstrual cycle. Estrogen in the complex modulated hormonal system is of utmost importance for the reproductive success of a female organism, and estrogen abnormalities are known to impair anovulation, irregularity of the menstrual cycle, and fertility [11].
Estradiol has multiple impacts on the ovary that include stimulating follicular development, avoiding the process of atresia, and supporting granulosa cells. Estrogen also activates IGF-1, which works in cooperation with FSH to promote cell growth of the granulosa cell and estradiol production [12]. Lack of estrogen, as demonstrated by OVX models or postmenopausal females, is associated with poor follicle development, a low number of follicular growing cohorts, and high levels of granulosa cell death that are prone to enhanced rates of aging. Hormones such as estrogen are therefore important in the protection of the ovarian tissue, as they have an important role in sustaining ovarian function and fertility [13].
Ovariectomy (OVX), the surgical removal of ovaries, is a widely used model to study the effects of estrogen deficiency, mimicking conditions such as menopause. Ovariectomy led to the regression of ovarian tissue, cessation of folliculogenesis, and loss of estradiol production. In the uterus, ovariectomy decreased endometrial thickness, vascularity, and epithelial regeneration due to estrogen deficiency. Due to ovariectomy, estrogen deficiency causes desynchronization of GnRH pulses, which in turn increases FSH and LH secretion, which is characteristic of menopausal women. Behind reproduction, these hormonal changes lead to changes in lipid profile, increased central fat deposits, insulin resistance, and hypertension. Estrogen regulates neurotransmission and synaptic plasticity in the nerve cells, thus having a neuroprotective effect. Previous research has shown that OVX results in hormonal changes, and estrogen deficiency in the brain has been associated with loss of cognition and with neurodegenerative diseases like Alzheimer’s disease [14].
Overview of estrogen
Estrogen, a sex steroid hormone that is synthesized from cholesterol, plays an important role in regulating various bodily functions, including those of the neuroendocrine, vascular, skeletal, immune, central nervous system, and reproductive systems [8,15]. Basically, there are three naturally occurring estrogens that are produced in the bodies of rodents and humans: Estrone (E1), Estradiol (E2), and Estriol (E3). E1 and E2 are produced in the ovaries and adrenal glands, while E3, a metabolite of E1 and E2, is primarily produced by the placenta during pregnancy [16]. Estrogen synthesis is achieved through the aromatization of androgen (testosterone and androstenedione), which is catalyzed by the enzyme aromatase present in the ovary, adipose, and adrenal tissues. In addition to its traditional function in the reproductive system, estrogen plays a part in the bone, cardiovascular, neuro, and immune systems.
Molecular mechanisms of estradiol action
Estrogen exerts its influence through Estrogen Receptors (ERs) of which there are three types: ER-α, ER-β, and G-Protein coupled Estrogen Receptor (GPER). ERα is mainly located in reproductive organs (the uterus, ovaries, and mammary glands), while ERβ is located in the brain, the lungs, bones, and ovaries. GPER is a membrane receptor that plays a role in the non-genomic action of estrogen, which occurs at the membrane level [17]. When estrogen interacts with ERα or ERβ, it triggers the activation of two principal pathways: the genomic or classic pathway and the non-classical pathway. The genomic effects of estrogen include the hormone binding to intracellular ERs, which then move to the nucleus and interact with the EREs on the DNA and alter the transcription of the genes that are involved in cell proliferation, apoptosis, and metabolic activities. The non-genomic signaling, however, is evoked when estrogen binds to GPER, and this binding results in the activation of intracellular signaling, for instance, the MAPK/ERK and PI3K/AKT pathways, which are involved in rapid cellular responses like cell survival, cell migration, and metabolic regulation, respectively [18].
Estrogen is not only involved in the woman’s reproductive system but also in many other body systems. For this reason, it plays a vital role in the body’s bone metabolic process. It suppresses the activity of osteoclasts, thus preventing bone loss and osteoporosis. In the cardiovascular system, estrogen contributes to vasodilation due to stimulation of nitric oxide synthesis. It also reduces LDL cholesterol levels and has anti-inflammatory properties, therefore benefiting the heart in premenopausal women. Estrogen has also been shown to possess a neuroprotective role in the central nervous system. It strengthens the connections between brain cells and can work on engram memory, which is vital with regard to the neurodegenerative diseases, such as Alzheimer’s disease. Moreover, it is pertinent to discuss the impact of estrogen on the immune system since it plays an important role in it. Hence, it regulates inflammation and plays a role in the development and functioning of immune cells in autoimmune diseases and inflammation.
Since estrogen is known to impact most physiological systems of women, ovariectomy or natural causes such as postmenopausal status are associated with a plethora of effects, including osteoporosis, cardiovascular diseases, cognitive decline, and metabolic dysfunction. Such effects highlight the necessity for estrogen replacement therapy and further investigation of SERMs that would help to avoid the detrimental impacts of estrogen deficiency with fewer side effects. It is essential to comprehend the molecular properties of estrogen and the multiple systemic actions for developing new approaches in concepts of reproductive medicine, endocrinology, and gerontology.
Importance of estrogen in female reproductive physiology
Estrogen is a significant steroid hormone that plays a critical role in the female reproductive system, including its cyclic changes, the endometrium, follicular growth, cervical mucus, and mammary gland development. It is also during the menstrual cycle that estrogen plays a complex role in regulating the Hypothalamic-Pituitary-Gonadal (HPG) axis. During the follicular phase, the estradiol levels increase and thus increase the GnRH pulse frequency to release the LH and FSH, which is crucial for follicular growth and development. The mid-cycle increase of estradiol triggers the positive feedback mechanism that, in turn, stimulates the release of LH, which is responsible for ovulation. Thereafter, in the luteal phase, estrogen, with the help of progesterone, prepares the endometrium for the possible implantation of the embryo and its further nourishment [19].
In addition to the systemic hormonal roles, estrogen plays localized roles that are essential for proper reproductive processes. It helps in endometrial proliferation through the promotion of neovascularization and glandular development, thus increasing the receptivity of the uterus. In the ovarian microenvironment, estradiol enhances the FSH receptor on the granulosa cells, making them more responsive to FSH and hence promoting follicular growth and steroidogenesis. These processes are rather critical in ensuring that ovulation occurs. Furthermore, it changes the rheological properties of cervical mucus as a prerequisite for sperm to overcome, so as to facilitate its transport to the site of fertilization. It also supports the structural and functional aspects of the vaginal mucosa so that atrophic changes do not occur frequently and there is a reduced chance of infections [20]. In the case of mammogenesis, estrogen plays a very crucial role in ductal branching and elongation during puberty and pregnancy in order to support lactation.
Altogether, these diverse actions underscore the unique indispensability of estrogen in the female reproductive system, ranging from central-level control to peripheral changes that are critical for reproduction and female reproductive health.
Figure 1 shows how hypothalamic control the production of estrogen and progesterone in a female's body. The hypothalamus discharges GnRH, which then prompts the pituitary gland to release LH as well as FSH, so ovarian hormone production becomes possible. The hormone estrogen impacts several physiological functions, which involve brain cell protection, together with mood regulation, breast formation, ovulation control, and bone growth. Hormonal feedback processes maintain steady hormone levels for reproductive health as well as whole body wellness [21].
The role of ovaries in estradiol production and overall reproductive function
Ovaries play a key role in the female reproductive system and thus are vital in the female body. It is known to be involved in oocyte maturation, corpus luteum formation, and hormonal regulation, therefore they are involved in estradiol production [22]. Estradiol biosynthesis in the ovaries is shown in a model referred to as the two-cell, two-gonadotropin model to demonstrate the roles of theca and granulosa cells in cooperation with LH and FSH. Theca cells, under the influence of LH, produce androgens like androstenedione and testosterone from cholesterol. They then diffuse into the adjacent granulosa cells; FSH enhances their conversion into estradiol by the aromatase enzyme. Apart from the synthesis of estradiol, the ovaries assist in oocyte development through folliculogenesis and ovulation of a mature oocyte. After ovulation, the follicular structure persists in the form of a corpus luteum, which is a temporary endocrine gland that is responsible for the secretion of progesterone, which is important for the early stages of pregnancy. Also, the ovaries release inhibitory hormones such as inhibin and activin that control the FSH hormone. These peptides play their role in the regulation of follicular development and selection.
Therefore, the ovaries are important in producing estradiol and regulating the reproductive system. This indicates that any complications in ovarian functions result in fertility problems, irregular menstrual cycles, and some other health complications like osteoporosis and cardiovascular diseases, making ovarian health very crucial for all women.
Ovariectomy is a relevant model for studying estrogen deficiency
Ovariectomy (OVX), or surgical removal of ovaries, is the most common model used to study the effects of estrogen deficiency, particularly those concerning post-menopausal status. The experiments with OVX produce the state of hormonal disruption, which correlates with the process of menopause [23]. It helps to reveal the multiple levels of the systemic impact of estrogen deficiency and contributes to the search for treatment strategies.
OVX causes several changes in the physiology of the body due to the sudden stoppage of estrogen secretion. The first effect, which is apparent, is hormonal dysregulation with a significant decrease in the level of estradiol [24]. This leads to such consequences as shrinkage of the reproductive organs, such as the uterus and the vagina, because of the lack of estrogen. In addition, estrogen deficiency also increases the activity of osteoclasts and hence the bone resorption, leading to osteoporosis. Cognitive functions are also impacted. Several studies have established that estrogen deficiency has negative effects on memory and neuroprotection [25].
Regarding human health, it has been useful in proving the mechanism of postmenopausal osteoporosis. Bone loss is significantly promoted after OVX since estrogen levels decrease abruptly in this model, thus making it fit for that purpose. This is because estrogen deficiency results in increased activation of osteoclasts, which in turn causes bone resorption to exceed bone formation. The present research demonstrates that OVX leads to a rapid reduction in trabecular bone mass in the first few months and increased bone fragility, which is similar to the condition in postmenopausal women. It is used to compare the effectiveness of anti-osteoporotic drugs such as bisphosphonates and selective estrogen receptor modulators for evaluating the efficacy of dietary and lifestyle interventions in the prevention of bone loss, in understanding the molecular mechanisms of osteoporosis, and, thus, the genes that may be targeted for therapy [26].
Furthermore, the OVX model has been used to elucidate estrogen’s neuroprotective effects to a great extent. Estrogen affects the structural remodeling of the synapses and the mechanisms related to memory. It modulates neurotransmitters, has antioxidant and anti-inflammatory properties, and has been shown to decrease oxidative stress in neurons. Ovariectomy-induced estrogen deficiency enhances the rate of cognitive dysfunction and neurodegeneration, which replicates the pathological changes in AD. These changes include elevated levels of β-amyloid and reduced estrogen levels, which lead to neurons’ death in the hippocampal or cortical regions and impaired mitochondria [27]. The OVX model is applied to study the neurochemical basis of estrogens on cognition in this study. It is also used for assessing estrogen-based neuroprotective interventions for the prevention or slowing down of AD and the modulation of estrogen effects on genetic risk factors of AD, such as the APOE4 allele [28]. The present study examined the association between HRT and dementia risk, but the results are inconclusive. When estrogen therapy is initiated within the first 10 years of menopause, it enhances cognitive function; however, if estrogen therapy is initiated after 10 years of menopause, then it raises the risk of dementia due to pre-existing brain disease. Other therapeutic possibilities exist since SERMs like raloxifene have been demonstrated to possess neuroprotective actions in OVX models.
Hence, OVX is an appropriate and useful model to use when investigating estrogen deficiency and its impact on the body. Since it mimics oestradiol levels in post-menopausal women, it assists in the understanding of diseases like osteoporosis, cardiovascular diseases, and neurodegenerative disorders, and the testing of drugs.
Figure 2 shows step-by-step information about how mice experience ovariectomy and receive estradiol treatment. Researchers begin by performing either sham surgery or ovariectomy on subject mice, followed by a period of essential post-operative recovery before administering two weeks of estradiol hormone treatment. Medical staff perform the surgical procedure by exposing the ovaries to make sutures at the uterus before closing the incision. The administration of estradiol injections requires a healing monitoring period as well as pain management to begin post-operatively [29].
Significance of the molecular mechanisms underlying estradiol’s actions and the effects of its absence
Estradiol, which is the most common type of estrogen, is known to be involved in the modulation of several physiological processes through complex signal transduction pathways. Studying these mechanisms and the effects of the lack of estradiol is crucial for the explanation of many disorders, with a main focus on those that are connected with the occurrence of menopause.
Estradiol has both genomic and non-genomic effects on cells and the different tissues of an organism. Estradiol is able to cross cell membranes and bind to intracellular receptors, ERa and ERβ These ligand-receptor complexes then move to the nucleus and act on specific sequences in DNA, which is called the estrogen response element, to control target genes. It has also been found to regulate the human genes that are involved in cell growth, development, and survival. For example, estradiol brings about an increase in the levels of the cell survival protein known as Bcl-2 and stimulates cell cycle progression by increasing the levels of cyclin D1 [30].
However, estradiol is not only involved in the classical genomic signaling but also in rapid non-genomic signaling. This includes the transmembrane receptors, for example, the G Protein-coupled Estrogen Receptor (GPER). Activation of GPER stimulates several intracellular signaling pathways, such as the MAPK/ERK pathway, which is involved in cell proliferation, and the PI3K/AKT pathway, which is pro-survival. These non-genomic effects are additional to the genomic actions of estradiol and enable it to exert rapid effects on cellular functions [17].
This is because the normal physiological changes that are observed following ovariectomy or during menopause are due to the reduction of estradiol levels. One of them is the intensification of oxidative stress, which negatively affects the stability of cell structures and their ability to function. Furthermore, estradiol deficiency is known to induce the production of pro-inflammatory cytokines, leading to chronic inflammation. All of these changes contribute to neuronal apoptosis, which may result in a decline of cognitive ability and an increase in the susceptibility to neurodegenerative diseases, including Alzheimer’s disease.
There are other effects of estradiol deficiency outside the neurological system. This is because the reduction in the levels of estradiol ensures that the rate of bone resorption by osteoclasts is higher than the formation of new bone by osteoblasts. This condition further increases the chances of osteoporosis and fractures in women after the menopausal age. However, since this hormone is crucial in determining the cardiovascular health of a woman, low levels of estradiol affect lipid profiles and make the blood vessels less elastic, thus rightfully making the woman prone to atherosclerosis and coronary artery disease [31].
These effects are due to the fact that estradiol is able to exert both genomic and non-genomic effects on various physiological processes. Lack of estrogen through the two pathways interferes with these pathways, leading to oxidative stress, inflammation, neuronal cell death, loss of bone density, and cardiovascular risk factors. Studying these mechanisms emphasizes the role of estradiol for organism survival and developing approaches to minimize the consequences of its deficiency.
Overview of estrogen signaling and its molecular mechanisms
Estrogen is involved in many physiological processes that include cell division, cell differentiation, cell death, and immune reactions due to a multitude of signaling pathways. These actions are through genomic as well as non-genomic effects by which the molecule elicits various cellular and systemic responses. The first one is the genomic, where estrogen binds to the nuclear receptors and gets involved in the process of gene transcription, and the second is the non-genomic, where estrogen binds to the membrane-bound receptors and initiates the intracellular signaling pathways. These signaling systems are, as such, very important in reproductive function, metabolism, the cardiovascular system, and the nervous system. Estrogen signaling disruption is closely associated with different pathological states such as hormone-sensitive cancers, bone disorders, cardiovascular diseases, and neurodegenerative diseases [32].
Description of different estrogen receptors (erα, erβ, gper) and their distribution in reproductive tissues
Despite being synthesized from the same gene, estrogen acts through three types of receptors, which include Estrogen Receptor alpha (ERa), Estrogen Receptor beta (ERβ), and G-Protein coupled Estrogen Receptor (GPER). These receptors are present in various tissues of the body, and they play various roles. ERa is most abundantly found in the uterus, breasts, liver, bones, and cardiovascular system, where its main focal areas of activity are in the commencement of reproductive tissues and reproductive organ functions. IUGR affects the growth of the uterus, development of the mammary glands, modeling of bones, lipid profile, and vascular integrity. ER is involved in the stimulation of cell division mainly in estrogen-responsive tissues, which makes it rational in conditions that are hormone-dependent, such as breast cancer and endometrial hyperplasia [33].
ERβ is mainly found in the ovaries, the prostate, the lungs, and the brain; it is involved in cell differentiation and has anti-proliferation effects. While ERa promotes cell growth in tissues, ERβ has been known to possess anti-tumor activity and is also involved in controlling the signal of estrogen regarding inflammation, immunological reaction, and neuronal signaling. Several studies have established that the activation of ERβ can have an inhibitory effect on ERa in the promotion of cell proliferation, thus providing the much-needed balance to prevent the progression of estrogen-induced carcinogenicity. Moreover, ERβ has been proven to be involved in the modulation of synaptic plasticity and age-related cognitive changes and thus might play a neuroprotective role in aging and neuropathological disorders, including Alzheimer’s disease [20].
Estrogen also binds to other nuclear receptors, but the most novel and well-known is the G-Protein-coupled Estrogen Receptor (GPER) that spans the membrane of cells and gives quick, non-genomic estrogen responses. GPER activates several pathways, such as the Mitogen-Activated Protein Kinase (MAPK) and Phosphatidylinositol-3-Kinase (PI3K/AKT) that involve various physiological aspects like metabolism, immunity, and the cardiovascular system. GPER is widely expressed in vascular endothelial cells, immune cells, and metabolic tissues and modulates vasodilation, swelling, and glucose metabolism. It has been shown that estrogen protection is mediated by GPER through an increase in NO production, a decrease in oxidative stress, and hence, the risk of atherosclerosis and hypertension [17].
The usage of ERa, ERb, and GPER in various tissues and their diverse types of functions emphasize the fact that the mechanisms of estrogen signaling are rather shades than black and white. Whereas ERa and ERb are mostly involved in the regulation of gene expressions in the target tissues, GPER mediates its rapid onset non-genomic effects on metabolism and signaling pathways [34]. This intertwined interaction makes sure that estrogen has a state and prolonged impact on numerous organ structures. Most noteworthy, ERa and ERb can regulate the biological actions of estrogen, and their ratio leads to either the promotion of cell proliferation or the inhibition and regulation of tissue homeostasis, respectively. This balance is of most significance in hormone-sensitive cancer being treated, where a selective targeting of the estrogen receptor is an approach to the management of breast and ovarian carcinomas [35].
Estrogen receptors are also involved in neuroendocrine activity, immune system function, and metabolism as well. In the brain, ERa and ERb b are implicated in processes such as synaptic plasticity, neuroprotection, and cognition, whereas GPER is involved in processes like neurotransmission and neuroinflammation [36]. Oxidative stress, neurodegeneration, and cognitive loss are the effects of low estrogen levels more evident in menopausal women, explaining why estrogenic signaling is critical in endorsing brain health. In the immune system and inflammation, estrogen interferes with cytokines and immune cells, involving both the ligand-binding domain ER and GPER, contributing to inflammation and/or immune sensitivity with autoimmune diseases. Furthermore, estrogen dependency of metabolic processes involves insulin sensitivity, adipogenesis, and energy balance through modulation of metabolic proteins and receptors, among them Peroxisome Proliferator-Activated Receptors (PPARs) and AMP-Activated Protein Kinase (AMPK) [37].
Due to the role estrogen plays in estrogen-related disorders, it is very significant for one to have a clear understanding of estrogen signaling at the molecular level in order to develop appropriate therapeutic interventions. Tamoxifen and Raloxifene are two of the most important SERMs since they are used to produce tissue-selective activation of ERa and ERb to treat certain diseases, including breast cancer and osteoporosis. Likewise, GPER agonists and antagonists are still undergoing research for their capability to treat cardiovascular diseases, metabolic diseases, and neurodegenerative diseases. It is for these reasons and the fact that the estrogen signal transduction system is one of the most complicated known to man that more research is required to establish the various molecular mechanisms that regulate the physiological effects of estrogen and their relevance to health and disease [18].
Figure 3 shows the distribution of ERa and ERb receptors in different tissues, together with their corresponding effects on these tissues. The metabolic system shows divergent reactions between ERa and ERb, whereas these receptors control food ingestion levels alongside metabolic activities such as insulin responsiveness, together with the regulation of lipid substances. This schema presents two pathways where Estrogen (E2) affects tissues: one that works directly and another that operates through the autonomic nervous system. Various organs within the body receive estrogen signals to control their metabolic functions through physiological mechanisms at the brain, liver, pancreas, adipose tissue, and muscles [38].
Classical genomic and non-genomic signaling pathways of estradiol
Most of the biological functions of estradiol that have been established above occur through complex signaling processes that allow for the proper regulation of cellular processes in the microenvironment [39,40]. These activities occur through two main routes—genomic and non-genomic routes. The genomic pathway operates on a slower note and is used in maintaining long-term changes in expression of a particular gene, while the non-genomic pathway operates on a faster note and is used in eliciting intracellular signaling pathways [41]. The integration of these pathways unveils the intricate nature of estrogen and its significance to female reproductive, neurological, cardiovascular, and skeletal functions. Such knowledge of molecular changes offers key information on designing new treatments for conditions associated with estrogen, including osteoporosis, cardiovascular diseases, and neurodegenerative diseases [42].
The genomic pathway is the traditional or the first model through which estrogen elicits a response and involves intracellular nuclear estrogen receptors, Estrogen Receptor-a (ERa), and Estrogen Receptor-β (ERβ) [43]. These receptors are activated by the binding of ligands and act as transcription factors that control the activity of the genes after interaction with estradiol. It starts from the passive transport of estradiol across the plasma membrane, where it binds to ERs, which could be soluble in the cytoplasm or the nuclear forms [43]. This ligand-receptor interaction brings about an alteration of conformation, which eventually leads to dimerization of receptors. The dimerized receptor complex is then transported to the nucleus, where it binds to estrogen response elements, or EREs, that can be located in the promoter region of target genes. This leads to the formation of co-regulators such as co-activators or co-repressors, which control the chromatin fabric and the transcription of genes. The genomic pathway leads to new protein synthesis, and as such, the physiological effects resulting from the activation of this pathway take between hours and days.
Some of the genes that are directly affected by the effects of estradiol, depending on the locations and the genomic signaling, include Cyclin D1, which enables cell division in the G1/s phase of the cell cycle, thus ensuring proper tissue remodeling and repair in the body. The second target is Bcl-2, being one of the members of the family of proteins that oppose apoptosis and help to regulate cell balance. In the vascular system, Vascular Endothelial Growth Factor (VEGF) is stimulated by estradiol, playing a major role in angiogenesis and making tissues such as the reproductive organs better supplied with blood [43]. Many of the effects of the estrogen receptor also go beyond the genome binding processes; the estrogen receptor can interact with other transcription factors like AP-1 and Sp1 and regulate gene expression regardless of the presence of EREs. This transfer increases the versatility of estrogen signaling and extends its cooperation into an impact on various tissues, including the brain and heart, as well as bones [44].
In this context, while the genomic action plan takes hours to exert its effects, the non-genomic form impacts several cellular reactions that happen within seconds to minutes. It is done through the use of G protein-coupled estrogen receptors, and the membrane-bound estrogen receptors include ERa as well as ERβ. These receptors trigger intracellular signaling mechanisms once they bind to the mentioned factors involved in cell survival, growth, metabolism, and neurotrophic factors. The signaling that is initiated through non-genomic estrogen signaling involves the MAPK/ERK, which is a signaling condition that is associated with actions such as cell growth, differentiation, and survival. This pathway is particularly relevant in the case of changes in reproductive tissue, synapse formation, and the function of the cardiovascular system. The activation of ERK by estradiol augments long-term potentiation and delays long-term depression, which enhances cognitive function and has neuroprotective effects that may be beneficial in lessening the development of neurodegenerative diseases, including Alzheimer’s disease [45].
Another possible non-genomic signaling pathway is a phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) one that is important for cell survival, glycolysis, and cardioprotection. Estradiol-related signaling with PI3K/AKT enhances endothelium functionality, reduces apoptosis, and displays anti-inflammatory potentialities. Here, it plays a crucial role in the cardiovascular system, stabilizing the blood vessels, reducing the effect of oxidants, and serving as protection against atherosclerosis or the thickening of the arterial walls. Another non-genomic signaling effector by estrogen is cyclic AMP/Protein Kinase A (PKA). Estrogen binding activates adenylate cyclase and leads to a rise in intracellular cAMP levels, resulting in PKA activation. This signaling cascade affects neurotransmitters, the immune system, and metabolism, especially in the liver and the brain. Thus, it now appears that estrogen is specifically involved in these fast signal transduction processes and serves as an essential regulator of the homeostasis of entire organ systems.
Though it is important to understand that the actions of estrogen and the benefits on the body are different in the genomic and non-genomic pathways, they do not work in isolation. Rather, they are tightly tied so that estrogenic signaling can be organized and targeted to particular tissues. Non-genomic signaling can also regulate gene expression at the transcription level using essential proteins like ERK, AKT, and Nuclear Factor-kappa B (NF-κB). On the other hand, genomic signaling is capable of modulating proteins that are involved in non-genomic signaling, thereby forming negative feedback [45]. It has been evidenced in reproductive organs for both synergistically coordinated actions in regulating ovarian follicle, uterine, and mammary gland development. Estrogen also has protective effects on brain cells via two different processes: through the activation of the genes concerned with neurotrophins on a genomic level and through an influence on the neuronal plasticity on a non-genomic level. Likewise, in the cardiovascular system, estrogen has its immediate effects on NO production via direct activation of eNOS through non-genomic actions, while the genomic effects include vasomodification and lipid profile regulation in blood vessels [46].
Hence, the right ratios between genomic and non-genomic estrogen signaling are vital for maintaining the body’s normal function. These pathways can be disrupted to result in several pathophysiological states; certain illnesses may be acquired due to the interruption of these pathways. For example, estrogen deficiency during the postmenopausal period is associated with the disruption of both pathways, leading to a rise in oxidative stress, inflammation, and apoptosis in different tissues. This leads to osteoporosis, cardiovascular diseases, and the decline in cognitive ability in people. Hormonal Replacement Therapy (HRT) has the objective of replacing the estrogen signaling, but it must be used according to the dosage and the time of use. SERMs are a class of drugs that help to stimulate the favorable effects of estrogen while reducing their drawback of HRT, like breast cancer and thromboembolism. The relationship between these pathways is important in the future strategy for the treatment of estrogen-related disease [47].
All physiological activities of estradiol are mediated by both the genomic and the non-genomic signaling pathways. Genomic estrogen action involves nuclear receptors and takes a longer time to change the gene expression and long-term cellular response, while the non-genomic mode of action works through membrane receptors and gives rapid responses to intracellular events [47]. They are tightly coordinated so that estrogenic regulation is precise and selective by various tissues. Thus, genomic and non-genomic actions of estrogen are crucial for ovarian function, neuronal survival, cardiovascular health, and metabolism. These disruptions can lead to various diseases and describe a need for specific therapeutic treatments. Molecular studies of these pathways to enhance the understanding of the hormonal processes will help in creating safer and more efficient statement therapies for the diseases resulting from estrogen imbalance.
Figure 4 shows two main activation routes for estrogen receptors. ER activates gene expression through DNA binding in the nucleus when using the ligand-dependent genomic pathway, and activates ER through phosphorylation with signaling interactions in the ligand-independent non-genomic pathway. The two regulatory mechanisms unite to function together for controlling gene transcription, which alters target cell operations. Membrane-bound ER plays a vital part in activating independent signaling pathways according to the figure representation [39] (Table 1).
Overview of key genes and molecular pathways regulated by estradiol in reproductive tissues
It has been proposed that estradiol is involved in the mechanisms of different molecular processes and gene interactions in reproductive tissues. These mechanisms play critical roles in proper developmental processes, cellular functions, regulation of cell death and living tissues, formation of new blood vessels, and changes in tissue organization of reproductive organs. Estrogen’s effects are exerted predominantly through Estrogen Receptors (ERα, ERβ, and G protein-coupled receptor, GPER), which, on activation, stimulate various signal transduction pathways that are vital for maintaining the health of the reproductive system. Disruptions in these pathways because of estrogen deficiency and hormonal alterations may enhance reproductive issues, for example, infertility issues, polycystic ovary syndrome, endometriosis, and ovarian failure [38].
GnRH signaling and calcium signaling pathway
As a molecular parameter the Gonadotropin-Releasing Hormone (GnRH signaling pathway is responsible for the regulation of reproductive hormone release and an appropriate working of the Hypothalamic-Pituitary-Gonadal (HPG) axis. GnRH is a decapeptide hormone produced and secreted from the neurons in the hypothalamus, and its release in burst frequency is critical for gonadotropin release from the anterior pituitary gland [48]. GnRH hence executes its function by binding to its specific receptor, the GnRH Receptor (GnRHR), which is a G Protein-Coupled Receptor (GPCR) that is located on the gonadotroph cells. This interaction leads to intracellular signaling pathways activation, particularly the Gq/11 proteins that cause the release of intracellular calcium and activation of PKC, which then activate the MAPK pathway. These intracellular pathways lead to the production and secretion of Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH), which in turn control steroidogenesis and gametogenesis of the gonads.
One of the main issues with the GnRH signal is that it is secreted in a pulsatile manner to allow for differential gonadotropin synthesis and secretion. In the secreting rate of the GnRH, there are rhythms and intensities that will lead to the selective release of LH or FSH. High frequency pulses are known to stimulate the secretion of LH, and low frequency pulses, on the other hand, stimulate the secretion of FSH. This interactive pulsatility of GnRH is very important in the process of follicular growth and ovulation. Over time and in the absence of pulsatile release, GnRH brings about down-regulation of GnRHR and GnRH secretion and forms the principle of therapy where GnRH agonists are used to treat endometriosis, PCOS, and hormonally-responsive cancers.
The calcium signaling pathway represents a core cellular signaling system that controls multiple bodily functions in reproductive tissues from the start of follicle formation until the end of implantation. The regulatory mechanism for cellular Ca2+ components relies upon tightly controlled channels, pumps, and exchangers that sit on cell membranes, endoplasmic reticulum, and mitochondria. When gonadotropins stimulate the ovary through granulosa and theca cell G protein-coupled receptors, they initiate a receptor signaling process that activates Phospholipase C (PLC). Through hydrolysis of PIP2, phospholipase C generates IP3 and DAG. IP3 reaches endoplasmic reticulum receptors to initiate the release of Ca2+storage and thereby raise cytoplasmic calcium concentrations temporarily. The elevated Ca2+ levels activate calmodulin and diverse calcium-dependent kinases and phosphatases, leading to the regulation of critical cellular processes for oocyte maturation and cumulus expansion as well as the ovulatory follicle rupture [49].
When GnRH attaches to gonadotropin receptor molecules, it sparks multiple cellular signals that involve calcium regulation to control both gonadotropin protein genes and hormone production. GnRH stimulation activates the phospholipase C pathway, which yields Inositol 1,4,5-trisphosphate (IP3) and Diacylglycerol (DAG) as products. Within the cytoplasm, IP3 triggers the release of Ca2+ from endoplasmic reticulum spaces to grow calcium levels. The increase of intracellular calcium levels triggers calcium/Calmodulin-dependent protein Kinases (CaMKs) and additional downstream components that activate the transcription of Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH) subunit genes that lead to their production and secretion. The calcium signaling pathway brings extracellular hormonal signals to nuclear events that drive gene expression for regulating reproductive processes [50].
The signaling network controlling gonadotropins develops through an integration of calcium signaling with Protein Kinase C (PKC) and cyclic AMP (cAMP) pathways to achieve precise activity control. Estradiol controls GnRH pulse dynamics through feedback systems that affect calcium signals within gonadotropins, along with their hormonal production. The important function of calcium signaling involves supporting the developmental maturation of gametes, together with the growth of follicles and the process of ovulation. The normal functioning of cumulus expansion, together with meiotic resumption, depends on changing calcium concentration levels found in granulosa cells and oocytes. Impaired folliculogenesis and luteal dysfunction, together with infertility, occur because of dysregulated calcium signaling patterns, which underscore its vital role in reproductive success. The precise knowledge of GnRH and calcium signaling relationships remains essential for creating targeted methods to protect fertility, as well as hormone treatments and reproductive disorder clinical interventions.
Figure 5 shows the sequence of events that start when Gonadotropin Releasing Hormone (GnRH) binds to its receptor. Activation of GnRH triggers G protein stimulation and creates two second messengers by activating Adenylyl Cyclase (AC) and Phospholipase C (PLCβ) that result in cyclic AMP (cAMP) and Inositol Triphosphate (IP3) generation. Activated second messengers trigger signaling pathways that engage Protein Kinase A (PKA) and Protein Kinase C (PKC), which function to control transcription factors that include ERK1/2 and the calcium signaling pathway. Gonadotropin synthesis, together with its secretion from the pituitary gland, becomes possible following this activation step. Reproductive functions become regulated through this signaling process because GnRH performs an essential role in the hypothalamic-pituitary axis functions (Table 2).
Factors and cofactors that influence the estrogen response
In fact, the changes of estradiol on the human body are not only based on the presence or absence of the hormone but also depend on cofactors and epigenetic modifications that control the activity of ER. These regulatory mechanisms further modulate the action of estrogen and keep the gene activation or repression within the right bounds in the right organs and at the right time [51]. The stimulation of estradiol and related cofactors ensures an influence on such processes as cell division, cell determination and differentiation, metabolic regulation, and even occurrences of specific diseases. This way, the scientists will be able to get a better view of estrogen’s roles and come up with effective treatment for estrogen-related diseases such as infertility, osteoporosis, and hormone-dependent cancers [52].
This is because some proteins can either enhance or suppress the activity of estrogen receptors through so-called co-activator or co-repressor proteins. These co-activators include Steroid Receptor Co-activator 1 (SRC-1) and the p300 protein complex, which have been implicated in the process of mediation of the effects of estrogens by enhancing gene transcription. SRC-1 binds directly to ERα and ERβ and cooperates with them by participating in chromatin remodeling with attractive coactivators for the transcription process. All these are crucial roles for tissue repair, cell division, and the control of reproduction. Another coactivator known as CBP/p300 is also considered the HAT through altering the chromatin structure to enhance the transcription rate. These coactivators are relevant particularly in reproductive tissues, which require estrogen-promoted cell division that is essential in developing the uterus and mammary glands [53].
On the other hand, the estrogen receptor is negatively controlled by co-repressors such as the nuclear receptor co-Repressor (NcoR) and the silencing mediator of the retinoic acid and thyroid hormone receptor (SMRt). These co-Repressors also bring in Histone Deacetylases (HDACs) to add to the chromatin and reduce gene transcription [54]. Here, NCoR and SMRT participate in the specifically important task of repressing the estrogen receptor in order to prevent hormonal overstimulation due to estrogen. In reproductive tissue, these corepressors play a role in regulating endometrial cell division, thereby preventing estrogen reinforcement of cell growth by the introduction of progesterone. Abnormalities in corepressors or their activity result in conditions like endometriosis, estrogen-sensitive carcinomas, and cancers where estrogenic activity is not controlled and contributes to the development of endometriosis [55]. This way, the coactivators and corepressors play an intricate balance that defines the degree and duration of estrogen signaling that is crucial for the reproductive and overall well-being of a woman.
It is important to comprehend that the estrogen signaling pathway involves numerous cofactors and epigenetic modifications to be able to design a particular treatment for estrogen-associated diseases. Coactivator and co-Repressor regulation, as well as epigenetic markers, have direct potential for therapeutic intervention in disorders such as infertility, bone loss, and hormone-dependent cancers [56]. For instance, Tamoxifen and Raloxifene are selective estrogen receptor modulators; they bind to estrogen receptors and change the co-activator/corepressor binding destinations, which partially or fully activate estrogen in the tissues. The usage of HDACi and DNMTi is promising for the manipulation of the process, which tries to restore the normal estrogen receptor gene function in cancer treatment. Using these molecular details, it is possible to treat cases of estrogen-related disorders with enhanced effectiveness while reducing side effects [57].
It is therefore important to consider that the response to estrogen, such as gene regulation, is not predetermined by the amount of hormone that is present in the body but depends on factors such as coactivators, co-Repressors, and epigenetic modifications of the estrogen receptor factor. Coactivators and co-Repressors regulate the ability of estrogen signaling, while epigenetic changes modify gene expression for physiological regulation.
There is also the challenge of external factors, which also play a role in altering the activity of the estrogen receptors by epigenetic features. Thus, a better understanding of such regulatory factors is critical for improving the treatment of hormone-associated diseases, including reproductive issues and estrogen-responsive malignancies.
Impact of ovariectomy on reproductive health
Ovariectomy is a common experimental technique used for the study of estrogen deficiency and its overall impact in the body. This model is more important in the biomedical research to study the postmenopausal physiology, hormonal replacement therapy, and related pathophysiologies of postmenopausal conditions such as osteoporosis, metabolic syndrome, and cardiovascular disease. Ovariectomy in mice helps the researchers to study the initial and potential effects of estrogen deficiency and therefore gives options to investigate such aspects of the reproductive system as physiology and signaling, as well as the disease process. The surgical removal of the ovaries results in hormonal and metabolic disturbances characteristic of the postmenopausal state in female animals, making this model an indispensable tool in studies of aging and endocrinology [58].
Immediate and long-term effects of ovariectomy on the physiology and reproductive organs of mice
Ovariectomy in mice provides a broad spectrum of physiological models to study a number of effects related to estrogen deficiency. It offers a controlled environment to analyze constant changes that occur soon after ovariectomy and sequential changes that occur with time. Some of the alterations include changes in the form and function of the reproductive organs, metabolism, skeletal structure, and the heart. Ovariectomy is another model that continues to expand the knowledge concerning the multifunctional roles of estrogen; it is also constructive for the production of treatment programs for estrogen-deficient diseases [59].
Short-term effect of ovariectomy: Firstly, ovariectomy leads to a rapid reduction in the concentrations of estrogen in the circulatory system, thus significant alterations in the morphology and physiology of the reproductive organs ensue. Estrogen plays a central role in the maintenance of uterine and vaginal epithelial tissues, and their sudden depletion leads to the squamous epithelium of the uterus and vagina to wither shortly after its depletion. In the uterus, total weight loss, tubal and uterine gland atrophy, and epithelial thinning are all observed due to the absence of estrogen stimulation. It becomes thinner, gets less blood supply, and produces less mucosal secretions, which is quite similar to atrophic vaginitis in women after menopausal years [60]. These changes have an impact on fertility studies, and for investigation must be considered in relation to hormone replacement therapy as well.
Apart from the reproductive system, ovariectomy induces significant metabolic disturbances almost immediately after the procedure. Estrogen is consequently proven to be very crucial in adipose tissue distribution, insulin sensitivity, and energy balance. Ovariectomized mice lack estrogen, store more fat in the visceral depots, expend less energy, and are negatively affected in glucose handling; thus are prone to obesity and insulin resistance [61]. Some of them include changes in leptin and adiponectin signaling that occur during the disruption of estrogen’s metabolic protectiveness, leading to increases in adiposity as well as glucose intolerance. These metabolic changes are in line with the observations of metabolic changes in postmenopausal women who normally develop increased abdominal obesity and other complications, including the development of Type 2 diabetes after the onset of ovarian senescence.
Long-term effect of ovariectomy: The long-term sequestration of estrogen in ovariectomy results in the breakdown of various physiological systems in the body. One of the most documented consequences in the long run is a deterioration of the mass and density of bones due to an increased mass of bone marrow fat fraction [62]. Estrogen plays an important role in regulating bone remodeling since it inhibits osteoclast activity and provides support to the osteoblasts. Instead, bone resorption occurs at a higher rate than bone formation, which in turn results in low bone mineral density, high cortical porosity, and high risk of fractures [63]. A recent postoperative study in ovariectomized mice has revealed that there is a decrease in bone mass by 30-50% within a few months, which is almost the same as woman after menopause [64].
In addition to skeletal deterioration, ovariectomy induces significant neuroendocrine and cardiovascular changes. It results in increased FSH level, and this has been attributed to the causes of bone loss, fat deposition, and neurodegeneration. In addition, the effects of estrogen loss cause chronic stress levels that lead to suppression of the HPA axis and stress reactivity [28]. Ovariectomized mice also have altered responses to stress and increased sympathetic activity since stress causes hypertension and impairs endothelium-dependent vasodilation. Estrogens have been proven to have direct vasculoprotective effects, such as regulation of NO synthesis and anti-inflammatory activity. Its deficiency causes vascular senescence and accelerates arterial wall hardening, atherosclerosis, and cardiovascular morbidity and mortality. These results are supported by data of epidemiological nature in a postmenopausal female when estrogen deficit relates to increased risk of hypertension, dyslipidemia, and ischemic heart disease.
Some surgical models, such as the ovariectomy model, have also served to give insights into cognitive aging and neurodegenerative diseases. Estrogen is an effective anti-oxidative hormone that is involved with synaptic plasticity, neurotransmitter release, and oxidative stress. Ovariectomy leads to enhanced cognitive impairment, decreased hippocampal performance, and increased β-amyloid deposition, which are characteristics of Alzheimer’s disease [65]. It has been established that ovariectomy in mice causes the effects mentioned earlier, and estrogen replacement can prevent these by improving synaptic density, decreasing neuroinflammation, and encouraging neurogeneration. However, the time of starting estrogen therapy seems to be important, since postponing the beginning of estrogen treatment may either be ineffective for preventing the deterioration of cognitive function, or even worsen it (Table 3).
Overview of molecular changes after ovariectomy
Some of the effects of estrogen deficiency on OVX mice are a result of multiple molecular changes throughout the physiologic signaling pathways. Estrogens are known to modulate neurogenic expression with respect to vasopressin, a neurohypophysial hormone that is also involved in the stress responses and regulation of feminine sexually selected behaviors, such as oxytocin. The OVX altered the level of AVP and oxytocin signaling, leading to enhanced anxiety, such as seen in ovariectomized females, and fine-tuning of emotion [65].
This is because estrogen deficiency at the cellular level affects multiple signaling pathways that are pertinent to maintaining neuronal viability and plasticity. This has been noted to have a significant impact on cognitive resilience and primarily affects a specific signaling pathway, which is the BDNF-TrkB signaling pathway. This impairs the expression of BDNF and downregulates the receptor TrkB due to decreased estrogen levels that cause neurotrophic and cognitive deficits. Intracellular signaling includes the estrogen modulation of the protein that is encoded by the PH domain leucine-rich repeat-containing protein phosphatase that activates the Phosphoinositide 3-Kinase (PI3K)/Akt pathway to stimulate the exonuclease-3 gene that inhibits the pro-apoptotic factors in the cell. OVX eliminates the level of estrogen and causes alteration in PI3K/Akt signaling and increased neuronal death, together with reduced synapses [66].
It has also been well studied that estrogen has its critical impact on the metabolic gene regulation that relates to glucose and lipid balances. The OVX-induced estrogen deprivation affects the insulin signaling pathway, which is associated with glomerular glucose uptake abnormalities and increased hepatic gluconeogenesis, contributing to hyperglycemia and a higher predisposition to develop the metabolic syndrome. Furthermore, estrogen plays an important role in lipid metabolism since it influences the activity of enzymes that are responsible for fatty acid elimination and storage. However, OVX mice have significantly higher levels of hepatic lipid content and dyslipidemia profile, which puts the OVX mice at a higher risk for metabolic disorders. These results indicate that estrogen plays a critical role in metabolic regulation and suggest that the OVX model may be a useful model to study metabolic dysfunction postmenopausally [67].
The role of estradiol replacement therapy and its effects
ERT is an important step taken to prevent and control the other systemic effects of estrogen deficiency that result from ovariectomy. Ovarian failure results in large amounts of low levels of circulating estrogen, and this deficiency triggers changes in various physiological systems in the body, such as the skeletal, cardiovascular, metabolic, and nervous systems. ERT is a way to restore the impact of estrogen, which will help some peri- or postmenopausal symptoms after ovariectomy and minimize the risk of the dangerous consequences for the woman’s health, such as osteoporosis, decreased cognition, and cardiovascular diseases. Nonetheless, the efficacy of ERT is not without limitations and complications; this means that the initiation of the therapy has to take cognizance of the patient’s status and other surrounding factors. This has brought emphasis on customized intervention plans and schedules, proper evaluation of the risks/benefits ratio in treatment, and toxicity profiles for enhancing therapeutic efficacy and minimizing harm in the patients.
Effects of exogenous estradiol administration after ovariectomy
Ovariectomy in currently used experimental animals, primarily rodents, causes the complete elimination of endogenous estrogen biosynthesis and provokes multisystem metabolic, cardiovascular, bone, and immune changes. The use of exogenous estrogen, namely estradiol, has been investigated extensively because of its effect in alleviating the above-mentioned post- ovariectomy effects [68].
Estrogen deficiency is the leading cause of metabolic change leading to insulin resistance and dyslipidemia. It is known that the administration of estradiol increases insulin effectiveness and the effectiveness of glucose uptake by the peripheral tissues, as well as reducing the rate of gluconeogenesis in the liver. Further, it also decreases ectopic lipid accumulation, especially in the liver and skeletal muscle, thereby reducing the high propensity of ailments like type 2 diabetes and non-alcoholic fatty liver diseases [37].
This is because estradiol replacement plays a very important role in the maintenance of the vascular system. Deficiency leads to endothelial dysfunction, the biomarkers of which include decreased NO production and impaired ACh-mediated relaxation. Thus, NO is a vasodilator, being involved in the regulation of vascular homeostasis, and its deficiency leads to increased vascular resistance and hypertension. Thus, estradiol seems to have cardio–protective effects by enhancing NO levels, improving endothelial function, and increasing vascular compliance [32].
Other significant effects of estrogen loss up to 6 months post-ovariectomy are related to body composition and muscle physiology. It has been reported that ovariectomy leads to changes in FFM, especially for body fat because of anabolic and catabolic hormone imbalance. These changes have, however, been observed to be prevented, among others, by estradiol replacement, which maintains the lean muscle mass and body composition of the animals to levels similar to those of non-ovariectomized animals.
The immune system is also regulated by estrogen, and estrogen depletion may have an impact on the immune system and actions involving inflammation. However, certain investigations of the impact of ERT on the immunocompetent have evidenced that exogenous estrogens do not impact neutrophil phagocytic ability or raise the risk of acquiring systemic infections. These observations indicate that although estrogen affects immunity, it is not necessarily detrimental to the innate immunity following its replacement therapy [69].
Potential side effects and limitations of estradiol replacement therapy
This is not to say that there are no drawbacks to using ERT; on the contrary, like with any clinical tool, ERT has its risks and limitations as discussed below. Another critical issue that has concerned people is the effect of HRT on cardiovascular outcomes. Essentially, estradiol has beneficial effects on the cardiovascular system, but some types of HRT, including combined oral estrogen-progestin treatment, increase the risk of thromboembolic events, which include deep vein thrombosis and stroke. Nevertheless, transdermal and bioidentical estrogen formulations have been reported to be safer, which means that patients should be treated differently depending on their cardiovascular risk factors.
Another important factor is that long-term usage of ERT can somehow be related to cancer. Estrogens for a long time have been associated with a higher risk of cancers that are sensitive to this hormone, such as breast and endometrial cancers. However, in research, participants pointed out that the risk seems to vary with the doses and the women taking estrogen alone or along with progestins. In order to minimize this side effect, the patient should be closely monitored, as well as have the minimal amount of medication and frequency possible involved [70].
Further possible side effects of ERT include abnormal uterine bleeding, breast discomfort, emotional sensitivity, edema, and headache. These side effects are usually not serious and can be eliminated by adjustments of the dosage or by the use of other forms of administration. However, there are marked differences in the patients’ response to ERT, due to which further analysis needs to be carried out based on the age factor, initial health state, and the onset of the therapy. These studies indicate that higher benefit is achieved when ERT is started as early as possible, at the time of onset of menopause or ovariectomy, and that initiation of therapy may be useful at a later stage or may be detrimental [71].
Interactive effects of estradiol and ovariectomy and their complex interactions
As has already been demonstrated, estrogen—the principal subtype being 17 β-estradiol—is involved in managing reproductive health through a network of signaling mechanisms. Ligation and ovariectomy of estrogen have been found to have various direct and indirect opposing actions and interactions through the estrogen signaling pathway. These contribute to tissue-selective estrogen action by factors that include the type of estrogen receptors, availability of co-activators, the extent to which specific DNA sites are opened up for binding, and the nature of membrane signal transduction. Knowledge of these complex processes is subsequently necessary to find ways to treat reproductive health problems and estrogen-related diseases and conditions [72].
Interactions between estrogen signaling and other signaling pathways relevant to reproductive health
Estrogen, being a hormone, acts through Estrogen Receptors (ER), mostly ERα and ER(12 pt), which are intracellular proteins that function as the DNA-bound receptors only when bound to the ligand. On ligand binding, these receptors change their conformation, dimerize, and attach to particular DNA sequences referred to as Estrogen Response Elements (EREs) so as to control the processes of gene transcription [18]. Besides this classical genomic pathway, estrogen also affects several other pivotal signaling pathways that are involved in reproduction.
Another important connection of estrogen is with the growth factor signaling pathways, such as IGF-1 and EGF. Such processes can then alter the functions of the ER through phosphorylation, downstream gene expression, and further cell evaluation. This is particularly plausible in view of the finding that estrogens’ effects are regulated by the activation of the IGF-1 and EGF receptors that can phosphorylate ERs, which in turn dictate gene expression and cellular responses [46].
The estrogen signaling pathway influences inflammatory signaling components with respect to women’s fertility. Estrogen may affect the cytokine and chemokine production, so it can affect inflammation in reproductive tissues. Parturition, ovulation, and implantation processes otherwise require controlled inflammation since it plays a crucial role in these processes [33].
It’s known that the estrogen signal is regulated by epigenetic changes, that is, DNA methylation and changes in histone proteins. Estrogen receptors can bring to the chromatin/DNA complex co-regulators that possess chromatin-modifying activity, implying changes in chromatin conformation and gene regulation [33]. It plays an important role in the tissue-selective estrogenic actions and is considered crucial for the non-cancerous roles of estrogen.
How these interactions contribute to the overall response to estradiol and ovariectomy
Overall, estrogen signaling interacts with other signaling pathways to affect the consequences of estradiol treatment as well as ovariectomy.
Modulation of reproductive tissue function: In reproductive tissues, estrogen interacts with the growth factor signaling pathways to perform normal function and growth. For instance, estrogen in combination with IGF stimulates the endometrium as well as the mammary gland during female development [73]. Ovariectomy means the removal of the ovaries, and this results in a deficiency of estrogen and therefore causes shrinkage of these tissues. This shows that exogenous estradiol can effectively replenish these interactions; hence, the tissue architecture and functionality are regained (Table 4).
Influence on cellular proliferation and apoptosis: Estrogen acts in the regulation of cell growth and death of reproductive cells by interacting with growth factor signaling. This regulation is essential when it comes to processes like endometrial regeneration during the menstrual cycle. As observed, estrogen deficiency following ovariectomy can alter this balance and lead to endometrial thinning, for example. There is a possibility of replacing the proliferative signals with estradiol so that the endometrial thickness returns to normal [74].
Impact on inflammatory responses: Estrogen Signaling and inflammatory pathways are in close relation to each other to facilitate reproductive events that entail inflammation. Ovariectomy leads to estrogen deficiency and disrupts the balance of pro- and anti-inflammatory cytokines, thus having an impact on fertility and enhancing the risk of infections. These studies have shown that the administration of estradiol influences these inflammatory pathways to ensure that the tissue remains healthy and functional.
Epigenetic influences: Estrogen signaling has been shown to regulate the epigenome changes and therefore the tissue- specific effects of estradiol treatment and ovariectomy [75]. In this case, modifications of DNA molecules, such as DNA methylation and histones, affect the gene expression patterns, which in turn affect the general physiology of the body. These epigenetic modifications may have definite impacts on reproductive health and some other processes like folliculogenesis and embryo implantation [76].
Figure 6 shows multiple ways through which estrogen operates to protect vascular wellness. The combination of protective mechanisms exercised by estrogen against oxidative stress, inflammation, and termination processes promotes anti-vascular aging effects. Through its mechanism, the hormone regulates blood pressure while binding homocysteine and preventing both lipid irregularities and glycemic issues. Blood vessels stay protected through several mechanisms enabled by estrogen, which demonstrates its crucial role in cardiovascular well-being [77].
Molecular mechanisms for tissue-specific responses: It has been realized that tissue-specific response to both estradiol and ovariectomy involves receptor isoform availability, co-regulator availability, and chromatin environment.
Estrogen receptor isoforms and distribution: The fact is that the body tissue can be differentially sensitive to the action of these particular estrogen receptor subtypes, ERα and ERβ. For example, ER-α is largely associated with the uterus and the mammary gland and is involved in the proliferative processes, while ER-β is associated with the ovary and the prostate, and it usually controls differentiation and anti-proliferative activities. This distribution determines how the tissues become receptive to the signals from the estrogen hormone.
Co-regulator proteins: Experimental studies show that co-activators and co-repressors have an impact on the transcription mediated through estrogen receptors. The tissues will have an increased response to estradiol for those cells with high co-activator content and a minimal response for tissues with high co-repressor signals. This balance is essential when it comes to adjusting the quantity of a specific protein expressed by the gene in accordance with hormonal signals.
Chromatin accessibility: Chromatin structure and composition, such as the position of nucleosomes and hence the state of histone proteins, influence the binding of the estrogen receptor to DNA. This is because genes that have chromatin in the open conformation are the most likely to be affected in response to the action of estradiol. This is because ovariectomy leads to the withdrawal of estrogen that elicits chromatin remodeling, which otherwise changes the accessibility of genes. These changes can be reversed by estradiol replacement and bring back the transcriptional program [78].
Membrane-Initiated Steroid Signaling (MISS): Apart from the nuclear membrane-bound receptor model, estrogen also must bind to membrane-anchored receptors to produce non-genomic responses. These MISS pathways include cell signaling through activation of kinases and various second messengers that produce tissue-selective effects. For instance, estrogen can cause the quick activation of membrane receptors within hours, promoting vasodilation or cardioprotection [79].
Selective Estrogen Receptor Modulators (SERMs): Selective estrogen receptor modulators (SERMs) are compounds that act as estrogen receptor agonists or antagonists depending on the target tissue. This selective activity is influenced by the differential expression of ER subtypes and co-regulators in various tissues (Arnal et al., 2017) [79]. For instance, tamoxifen acts as an antagonist in breast tissue but as an agonist in bone and uterine tissues, highlighting the complexity of estrogen signaling and its tissue-specific effects. This selective modulation of estrogen receptors allows for the development of targeted therapies in conditions such as breast cancer, osteoporosis, and cardiovascular diseases, where estrogen plays a crucial regulatory role [80].
Conclusion
This review shows that estradiol is not limited to a reproductive hormone but acts as a system-wide regulator linking the hypothalamic–pituitary–gonadal axis with ovarian activity, uterine receptivity, bone remodeling, vascular protection, metabolic homeostasis, and neuroprotection. These effects are mediated through ERα, ERβ, and GPER via coordinated genomic and non-genomic pathways, including MAPK/ERK, PI3K/AKT, GnRH, and calcium signaling. Evidence discussed across OVX models demonstrates that estrogen loss disrupts reproductive tissues and also accelerates bone loss, metabolic imbalance, vascular dysfunction, inflammation, and neural vulnerability. Estradiol replacement can reverse several of these abnormalities, but its benefits remain dependent on dose, timing, formulation, and tissue-specific receptor responses, which explains why clinical translation requires caution. Overall, an integrated understanding of estradiol signaling is essential for improving menopause-related care and for developing safer, more selective therapies that preserve beneficial estrogenic actions while minimizing adverse effects. This synthesis also supports the continued use of OVX models as a valuable platform for mechanistic and translational endocrine research.
Limitations and future Research directions
Despite providing a comprehensive overview of estradiol signaling, ovariectomy-induced estrogen deficiency, and estradiol replacement strategies, the available literature is highly heterogeneous with respect to animal model, species, age, experimental duration, ovariectomy timing, estradiol dose, route of administration, and measured endpoints, making direct comparison among studies difficult and limiting the ability to draw fully uniform conclusions. Although the Ovariectomy (OVX) model is widely used to mimic postmenopausal estrogen deficiency, it does not completely reproduce the gradual endocrine and physiological transition that occurs during natural menopause in women. Therefore, findings from OVX-based studies should be interpreted cautiously when considering their translational and clinical relevance. This review mainly emphasizes estradiol-centered mechanisms, whereas female reproductive physiology is regulated through a broader hormonal and molecular network that includes progesterone, gonadotropins, inflammatory mediators, metabolic regulators, and other signaling molecules. As a result, some of the biological responses discussed here cannot be attributed to estradiol alone and need further research. Greater standardization of experimental design, including OVX protocols, estradiol dosing regimens, treatment duration, and molecular endpoints, is needed to improve comparability across studies.
In future investigations, researchers should also adopt more integrative approaches, including transcriptomics, proteomics, epigenomics, and metabolomics, to better define the molecular basis of estradiol action across reproductive and extra-reproductive tissues. Such approaches may help identify novel biomarkers of estrogen deficiency, treatment response, and tissue-selective estrogen activity. Further research is needed to clarify the distinct and overlapping roles of ERα, ERβ, and GPER in reproductive tissues as well as in the brain, bone, cardiovascular system, and metabolic organs. A better understanding of receptor-specific signaling may support the development of safer and more selective therapeutic strategies, including improved hormone replacement approaches and selective estrogen receptor modulators. From a clinical perspective, future research should place greater emphasis on the timing, dose, formulation, and route of estradiol administration, particularly in relation to long-term outcomes involving fertility, cognition, bone health, cardiovascular function, and metabolism. Well-designed longitudinal studies and comparative clinical trials are essential to determine how estrogen-based interventions can be optimized for different physiological and pathological conditions.
- Mei L, Osakada T, Lin D. Hypothalamic control of innate social behaviors. Science. 2023;382(6669):399-404. Available from: https://doi.org/10.1126/science.adh8489
- Shi M, Sirard MA. Transcriptome and epigenome analysis of porcine embryos from non-esterified fatty acid-exposed oocytes. Domest Anim Endocrinol. 2021;76:106605. Available from: https://doi.org/10.1016/j.domaniend.2021.106605
- Yang H, Wu Z. Genome editing of pigs for agriculture and biomedicine. Front Genet. 2018;9:360. Available from: https://doi.org/10.3389/fgene.2018.00360
- Al-Suhaimi EA, Khan FA, Homeida A. Regulation of male and female reproductive functions. In: Emerging concepts in endocrine structure and functions. Springer; 2022. Available from: https://doi.org/10.1007/978-981-16-9016-7_9
- Murray CM, Orr CJ. Hormonal regulation of the menstrual cycle and ovulation. In: Maternal-fetal and neonatal endocrinology. Elsevier; 2020:159-167. Available from: https://doi.org/10.1016/B978-0-12-814823-5.00012-X
- Babaniyi GG, Ajao BH, Akor UJ, Babaniyi E. Reproductive endocrinology drug development: hormones, metabolism, and fertility in female reproductive health. In: Perspectives of quorum quenching in new drug development. CRC Press; 2023. Available from: https://www.taylorfrancis.com/chapters/edit/10.1201/9781003297826-10/reproductive-endocrinology-drug-development-gabriel-gbenga-babaniyi-babatunde-hadiyatullahi-ajao-ulelu-jessica-akor-elizabeth-babaniyi
- Zecchi-Orlandini S, Formigli L, Tani A, Benvenuti S, Fiorelli G, Papucci L, et al. 17β-estradiol induces apoptosis in the preosteoclastic FLG 29.1 cell line. Biochem Biophys Res Commun. 1999;255(3):680-685. Available from: https://doi.org/10.1006/bbrc.1999.0215
- Hamilton KJ, Hewitt SC, Arao Y, Korach KS. Estrogen hormone biology. Curr Top Dev Biol. 2017;125:109-146. Available from: https://doi.org/10.1016/bs.ctdb.2016.12.005
- Barbieri RL. The endocrinology of the menstrual cycle. In: Humana Press; 2014:145-169. Available from: https://doi.org/10.1007/978-1-4939-0659-8_7
- Jeon H, Lee WS, Kim JW. Elevated luteinizing hormone levels during the artificial endometrial preparation cycle do not impact pregnancy outcomes in patients undergoing single vitrified-warmed blastocyst transfer. Hum Fertil (Camb). 2024;27(1):2424336. Available from: https://doi.org/10.1080/14647273.2024.2424336
- Chandel S, Das S, Ojha S, Pandey M. Hormonal imbalances and genetic factors in menstrual cycle irregularities. Bentham Sci Publ; 2024;28:101-128. Available from: https://doi.org/10.2174/9789815256291124010008
- Lim CED, Ng RWC, Cheng NCL, Zhang GS, Chen H. Acupuncture for polycystic ovarian syndrome. Cochrane Database Syst Rev. 2019;(7). Available from: https://doi.org/10.1002/14651858.cd007689.pub4
- Zhang WY, Guo YJ, Wang KY, Chen LM, Jiang P. Neuroprotective effects of vitamin D and 17β-estradiol against ovariectomy-induced neuroinflammation and depressive-like state: role of the AMPK/NF-κB pathway. Int Immunopharmacol. 2020;86:106734. Available from: https://doi.org/10.1016/j.intimp.2020.106734
- Osse AML. Investigations into the relationship between menopause and Alzheimer’s disease pathology in a mouse model with ovarian follicle depletion [thesis]. 2024. Available from: https://oasis.library.unlv.edu/thesesdissertations/5058/
- Zamani MR, Desmond NL, Levy WB. Estradiol modulates long-term synaptic depression in female rat hippocampus. J Neurophysiol. 2000;84(4):1800-1808. Available from: https://doi.org/10.1152/jn.2000.84.4.1800
- Diller M, Schüler S, Buchholz S, Lattrich C, Treeck O, Ortmann O. Effects of estriol on growth, gene expression and estrogen response element activation in human breast cancer cell lines. Maturitas. 2014;77(4):336-343. Available from: https://doi.org/10.1016/j.maturitas.2014.01.004
- Prossnitz ER, Barton M. Estrogen biology: new insights into GPER function and clinical opportunities. Mol Cell Endocrinol. 2014;389(1-2):71-83. Available from: https://doi.org/10.1016/j.mce.2014.02.002
- Levin ER. Integration of the extranuclear and nuclear actions of estrogen. Mol Endocrinol. 2005;19(8):1951-1959. Available from: https://doi.org/10.1210/me.2004-0390
- Ramírez-González JA, Vaamonde-Lemos R, Cunha-Filho JS, Varghese A. Overview of the female reproductive system. In: Exercise and human reproduction: induced fertility disorders and possible therapies. 2016:19-46. Available from: https://doi.org/10.1007/978-1-4939-3402-7_2
- Deroo BJ, Korach KS. Estrogen receptors and human disease. J Clin Invest. 2006;116(3):561-570. Available from: https://doi.org/10.1172/JCI27987
- Wesley AL, Zein M, Olgun M, Catapano LA. The reproductive life cycle. In: Textbook of women’s reproductive mental health. 2021:1-30. Available from: https://doi.org/10.1176/appi.books.9781615379545.lg01
- Zhang J, Bao Y, Zhou X, Zheng L. Polycystic ovary syndrome and mitochondrial dysfunction. Reprod Biol Endocrinol. 2019;17(1):67. Available from: https://doi.org/10.1186/s12958-019-0509-4
- Lorenzo J. From the gut to bone: connecting the gut microbiota with Th17 T lymphocytes and postmenopausal osteoporosis. J Clin Invest. 2021;131(5). Available from: https://doi.org/10.1172/jci146619
- Żabińska M, Wiśniewska K, Węgrzyn G, Pierzynowska K. Exploring the physiological role of the G protein-coupled estrogen receptor (GPER) and its associations with human diseases. Psychoneuroendocrinology. 2024:107070. Available from: https://doi.org/10.1016/j.psyneuen.2024.107070
- Yaffe K, Sawaya G, Lieberburg I, Grady D. Estrogen therapy in postmenopausal women: effects on cognitive function and dementia. JAMA. 1998;279(9):688-695. Available from: https://doi.org/10.1001/jama.279.9.688
- Kalu DN. The ovariectomized rat model of postmenopausal bone loss. Bone Miner. 1991;15(3):175-191. Available from: https://doi.org/10.1016/0169-6009(91)90124-i
- Clarke IJ, Smith JT, Caraty A, Goodman RL, Lehman MN. Kisspeptin and seasonality in sheep. Peptides. 2009;30(1):154-163. Available from: https://doi.org/10.1016/j.peptides.2008.08.022
- Brinton RD, Yao J, Yin F, Mack WJ, Cadenas E. Perimenopause as a neurological transition state. Nat Rev Endocrinol. 2015;11(7):393-405. Available from: https://doi.org/10.1038/nrendo.2015.82
- Bouxsein ML, Myers KS, Shultz KL, Donahue LR, Rosen CJ, Beamer WG. Ovariectomy-induced bone loss varies among inbred strains of mice. J Bone Miner Res. 2005;20(7):1085-1092. Available from: https://doi.org/10.1359/jbmr.050307
- Ucar EH, Peker C, Hitit M, Kose M, Tatar M, Bozkaya F, et al. Altered luteal expression patterns of genomic and non-genomic progesterone receptors in bitches at different reproductive states. Theriogenology. 2024;218:153-162. Available from: https://doi.org/10.1016/j.theriogenology.2024.02.001
- Khosla S, Melton LJ 3rd, Riggs BL. The unitary model for estrogen deficiency and the pathogenesis of osteoporosis: is a revision needed? J Bone Miner Res. 2011;26(3):441-451. Available from: https://doi.org/10.1002/jbmr.262
- Mendelsohn ME. Protective effects of estrogen on the cardiovascular system. Am J Cardiol. 2002;89(12):12-17. Available from: https://doi.org/10.1016/s0002-9149(02)02405-0
- Heldring N, Pike A, Andersson S, Matthews J, Cheng G, Hartman J, et al. Estrogen receptors: how do they signal and what are their targets. Physiol Rev. 2007;87(3):905-931. Available from: https://doi.org/10.1152/physrev.00026.2006
- Chen P, Li B, Ou-Yang L. Role of estrogen receptors in health and disease. Front Endocrinol. 2022;13:839005. Available from: https://doi.org/10.3389/fendo.2022.839005
- Thomas C, Gustafsson JA. The different roles of ER subtypes in cancer biology and therapy. Nat Rev Cancer. 2011;11(8):597-608. Available from: https://doi.org/10.1038/nrc3093
- Xu H, Qing T, Shen Y, Huang J, Liu Y, Li J, et al. RNA-seq analyses the effect of high-salt diet in hypertension. Gene. 2018;677:245-250. Available from: https://doi.org/10.1016/j.gene.2018.07.069
- Mauvais-Jarvis F. Estrogen and androgen receptors: regulators of fuel homeostasis and emerging targets for diabetes and obesity. Trends Endocrinol Metab. 2011;22(1):24-33. Available from: https://doi.org/10.1016/j.tem.2010.10.002
- Hewitt SC, Korach KS. Estrogen receptors: new directions in the new millennium. Endocr Rev. 2018;39(5):664-675. Available from: https://doi.org/10.1210/er.2018-00087
- Silva E, Kabil A, Kortenkamp A. Cross-talk between non-genomic and genomic signalling pathways—distinct effect profiles of environmental estrogens. Toxicol Appl Pharmacol. 2010;245(2):160-170. Available from: https://doi.org/10.1016/j.taap.2010.02.015
- Zhang W, Wang L, Hu B, Jin M, Zhou J. Changes in ovarian tissue structure and distribution of oestrogen receptors in Huanghuai goats at different ages. Anat Histol Embryol. 2024;53(3):e13042. Available from: https://doi.org/10.1111/ahe.13042
- Simoncini T, Genazzani AR. Non-genomic actions of sex steroid hormones. Eur J Endocrinol. 2003;148(3):281-292. Available from: https://doi.org/10.1530/eje.0.1480281
- Azuma K, Inoue S. Genomic and non-genomic actions of estrogen: recent developments. Biomol Concepts. 2012;3(4):365-370. Available from: https://doi.org/10.1515/bmc-2012-0002
- Simoncini T, Mannella P, Fornari L, Caruso A, Varone G, Genazzani AR. Genomic and non-genomic effects of estrogens on endothelial cells. Steroids. 2004;69(8-9):537-542. Available from: https://doi.org/10.1016/j.steroids.2004.05.009
- Dahlman-Wright K, Cavailles V, Fuqua SA, Jordan VC, Katzenellenbogen JA, Korach KS, et al. International union of pharmacology. LXIV. Estrogen receptors. Pharmacol Rev. 2006;58(4):773-781. Available from: https://doi.org/10.1124/pr.58.4.8
- Eyster KM. The estrogen receptors: an overview from different perspectives. Methods Mol Biol. 2016;1366:1-10. Available from: https://doi.org/10.1007/978-1-4939-3127-9_1
- Nilsson S, Makela S, Treuter E, Tujague M, Thomsen J, Andersson G, et al. Mechanisms of estrogen action. Physiol Rev. 2001;81(4):1535-1565. Available from: https://doi.org/10.1152/physrev.2001.81.4.1535
- Srivastava DP, Waters EM, Mermelstein PG, Kramár EA, Shors TJ, Liu F. Rapid estrogen signaling in the brain: implications for the fine-tuning of neuronal circuitry. J Neurosci. 2011;31(45):16056-16063. Available from: https://doi.org/10.1523/jneurosci.4097-11.2011
- Bliss SP, Navratil AM, Xie J, Roberson MS. GnRH signaling, the gonadotrope and endocrine control of fertility. Front Neuroendocrinol. 2010;31(3):322-340. Available from: https://doi.org/10.1016/j.yfrne.2010.04.002
- Fan HY, Liu Z, Johnson PF, Richards JS. CCAAT/enhancer-binding proteins (C/EBP)-α and -β are essential for ovulation, luteinization, and the expression of key target genes. Mol Endocrinol. 2011;25(2):253-268. Available from: https://doi.org/10.1210/me.2010-0318
- Dupont J, Scaramuzzi RJ. Insulin signalling and glucose transport in the ovary and ovarian function during the ovarian cycle. Biochem J. 2016;473(11):1483-1501. Available from: https://doi.org/10.1042/BCJ20160124
- Sarwar S, Alamro A, Huq F, Alghamdi A. Insights into the role of epigenetic factors determining the estrogen response in estrogen-positive ovarian cancer and prospects of combining epi-drugs with endocrine therapy. Front Genet. 2022;13:812077. Available from: https://doi.org/10.3389/fgene.2022.812077
- Nevola R, Tortorella G, Rosato V, Rinaldi L, Imbriani S, Perillo P, et al. Gender differences in the pathogenesis and risk factors of hepatocellular carcinoma. Biology (Basel). 2023;12(7):984. Available from: https://doi.org/10.3390/biology12070984
- Iwase H. Molecular action of the estrogen receptor and hormone dependency in breast cancer. Breast Cancer. 2003;10:89-96. Available from: https://doi.org/10.1007/bf02967632
- McKenna NJ, Lanz RB, O’Malley BW. Nuclear receptor coregulators: cellular and molecular biology. Endocr Rev. 1999;20(3):321-344. Available from: https://doi.org/10.1210/edrv.20.3.0366
- Parisi F, Sonderegger B, Wirapati P, Delorenzi M, Naef F. Relationship between estrogen receptor α location and gene induction reveals the importance of downstream sites and cofactors. BMC Genomics. 2009;10:1-13. Available from: https://doi.org/10.1186/1471-2164-10-381
- Raimondo D, Raffone A, Zakhari A, Maletta M, Vizzielli G, Restaino S, et al. The impact of hysterectomy on oncological outcomes in patients with borderline ovarian tumors: a systematic review and meta-analysis. Gynecol Oncol. 2022;165(1):184-191. Available from: https://doi.org/10.1016/j.ygyno.2022.01.019
- Arriola SIA, Lin A, Brinkman JA, Meyer E, Morrison M, Tomasiewicz JL, et al. Ovariectomy uncouples lifespan from metabolic health and reveals a sex-hormone-dependent role of hepatic mTORC2 in aging. Elife. 2020;9:e56177. Available from: https://doi.org/10.7554/elife.56177
- Labrie F, Martel C, Pelletier G. Is vulvovaginal atrophy due to a lack of both estrogens and androgens? Menopause. 2017;24(4):452-461. Available from: https://doi.org/10.1097/gme.0000000000000768
- Hickey M, Fraser IS. Clinical implications of disturbances of uterine vascular morphology and function. Best Pract Res Clin Obstet Gynaecol. 2000;14(6):937-951. Available from: https://doi.org/10.1053/beog.2000.0136
- Vigil P, Melendez J, Petkovic G, Del Río JP. The importance of estradiol for body weight regulation in women. Front Endocrinol. 2022;13:951186. Available from: https://doi.org/10.3389/fendo.2022.951186
- Wong SK, Chin KY, Suhaimi FH, Ahmad F, Ima-Nirwana S. The relationship between metabolic syndrome and osteoporosis: a review. Nutrients. 2016;8(6):347. Available from: https://doi.org/10.3390/nu8060347
- Imai Y, Youn MY, Kondoh S, Nakamura T, Kouzmenko A, Matsumoto T, et al. Estrogens maintain bone mass by regulating expression of genes controlling function and life span in mature osteoclasts. Ann N Y Acad Sci. 2009;1173:E31-E39. Available from: https://doi.org/10.1111/j.1749-6632.2009.04954.x
- Wang X, Zhang C, Zhao G, Yang K, Tao L. Obesity and lipid metabolism in the development of osteoporosis. Int J Mol Med. 2024;54(1):61. Available from: https://doi.org/10.3892/ijmm.2024.5385
- Genazzani AR, Pluchino N, Luisi S. Estrogen, cognition and female ageing. Hum Reprod Update. 2007;13(2):175-187. Available from: https://doi.org/10.1093/humupd/dml042
- Farkas S, Szabó A, Török B, Sólyomvári C, Fazekas CL, Bánrévi K, et al. Ovariectomy-induced hormone deprivation aggravates Aβ1-42 deposition in the basolateral amygdala and cholinergic fiber loss in the cortex but not cognitive behavioral symptoms in a triple transgenic mouse model of Alzheimer’s disease. Front Endocrinol. 2022;13:985424. Available from: https://doi.org/10.3389/fendo.2022.985424
- Spencer JL, Waters EM, Romeo RD, Wood GE, Milner TA, McEwen BS. Uncovering the mechanisms of estrogen effects on hippocampal function. Front Neuroendocrinol. 2008;29(2):219-237. Available from: https://doi.org/10.1016/j.yfrne.2007.08.006
- do Nascimento AL, Furtado GC, Vilhena VM, Carvalho HO, Sales PF, Barcellos AON, et al. Osteoprotective effect of the phytonutraceutical Ormona® on ovariectomy-induced osteoporosis in Wistar rats. Nutraceuticals. 2024;4(2):147-164. Available from: https://doi.org/10.3390/nutraceuticals4020010
- Souza MF, Couto-Pereira N, Freese L, Costa PA, Caletti G, Bisognin KM, et al. Behavioral effects of endogenous or exogenous estradiol and progesterone on cocaine sensitization in female rats. Braz J Med Biol Res. 2014;47(6):505-514. Available from: https://doi.org/10.1590/1414-431x20143627
- Bouman A, Heineman MJ, Faas MM. Sex hormones and the immune response in humans. Hum Reprod Update. 2005;11(4):411-423. Available from: https://doi.org/10.1093/humupd/dmi008
- Beral V, Reeves G, Bull D, Green J; Million Women Study Collaborators. Breast cancer risk in relation to the interval between menopause and starting hormone therapy. J Natl Cancer Inst. 2011;103(4):296-305. Available from: https://doi.org/10.1093/jnci/djq527
- Manson JE, Kaunitz AM. Menopause management—getting clinical care back on track. N Engl J Med. 2016;374(9):803-806. Available from: https://doi.org/10.1056/nejmp1514242
- Clarke CL, Sutherland RL. Progestin regulation of cellular proliferation. Endocr Rev. 1990;11(2):266-301. Available from: https://doi.org/10.1210/edrv-11-2-266
- Katzenellenbogen BS, Katzenellenbogen JA. Estrogen receptor transcription and transactivation: estrogen receptor alpha and estrogen receptor beta—regulation by selective estrogen receptor modulators and importance in breast cancer. Breast Cancer Res. 2000;2:1-10. Available from: https://doi.org/10.1186/bcr78
- Critchley HO, Maybin JA, Armstrong GM, Williams ARW. Physiology of the endometrium and regulation of menstruation. Physiol Rev. 2020;100(3):1149-1179. Available from: https://doi.org/10.1152/physrev.00031.2019
- Naimi S, Viennois E, Gewirtz AT, Chassaing B. Direct impact of commonly used dietary emulsifiers on human gut microbiota. Microbiome. 2021;9:1-19. Available from: https://doi.org/10.1186/s40168-020-00996-6
- Recillas-Targa F. Cancer epigenetics: an overview. Arch Med Res. 2022;53(8):732-740. Available from: https://doi.org/10.1016/j.arcmed.2022.11.003
- El-Gendy AA, Elsaed WM, Abdallah HI. Potential role of estradiol in ovariectomy-induced derangement of renal endocrine functions. Ren Fail. 2019;41(1):507-520. Available from: https://doi.org/10.1080/0886022x.2019.1625787
- Klemm SL, Shipony Z, Greenleaf WJ. Chromatin accessibility and the regulatory epigenome. Nat Rev Genet. 2019;20(4):207-220. Available from: https://doi.org/10.1038/s41576-018-0089-8
- Arnal JF, Lenfant F, Metivier R, Flouriot G, Henrion D, Adlanmerini MM, et al. Membrane and nuclear estrogen receptor alpha actions: from tissue specificity to medical implications. Physiol Rev. 2017;97(3):1045-1087. Available from: https://doi.org/10.1152/physrev.00024.2016
- Garg A, Singh B, Sharma R, Singh A, Kumar A. Selective estrogen receptor modulators (SERMs): mechanistic insights against microbial infections. Curr Mol Med. 2020;20(2):102-115. Available from: https://doi.org/10.2174/1566524019666191014112133
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
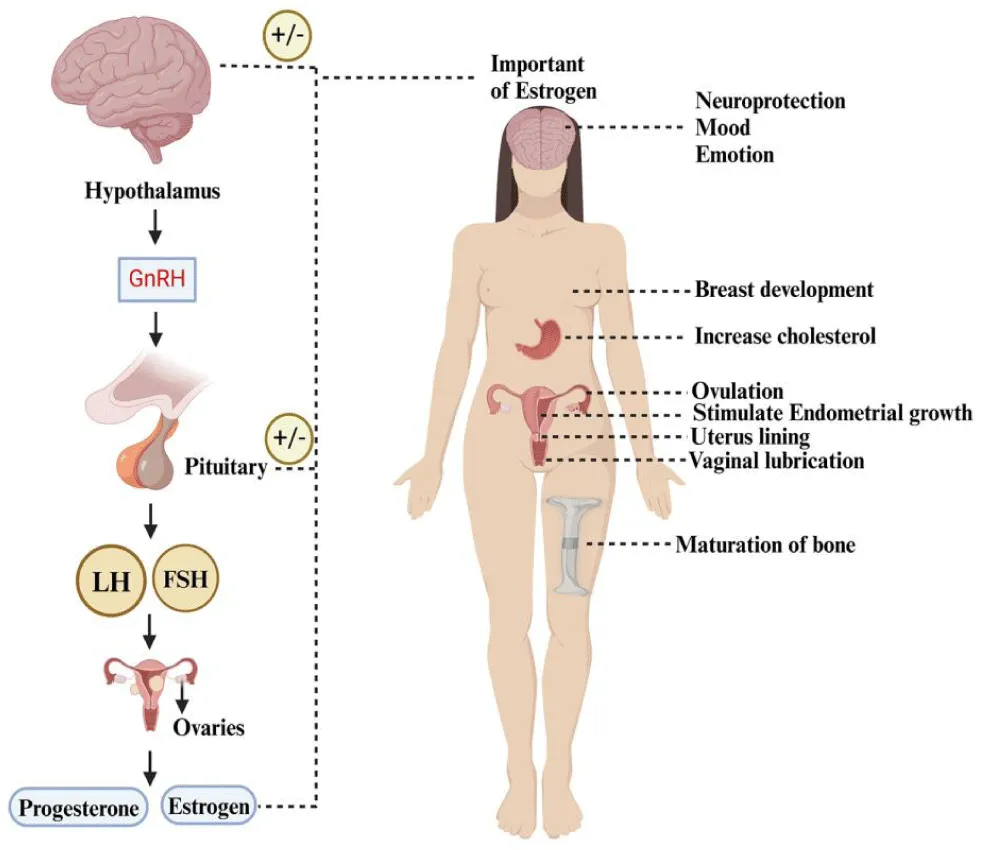
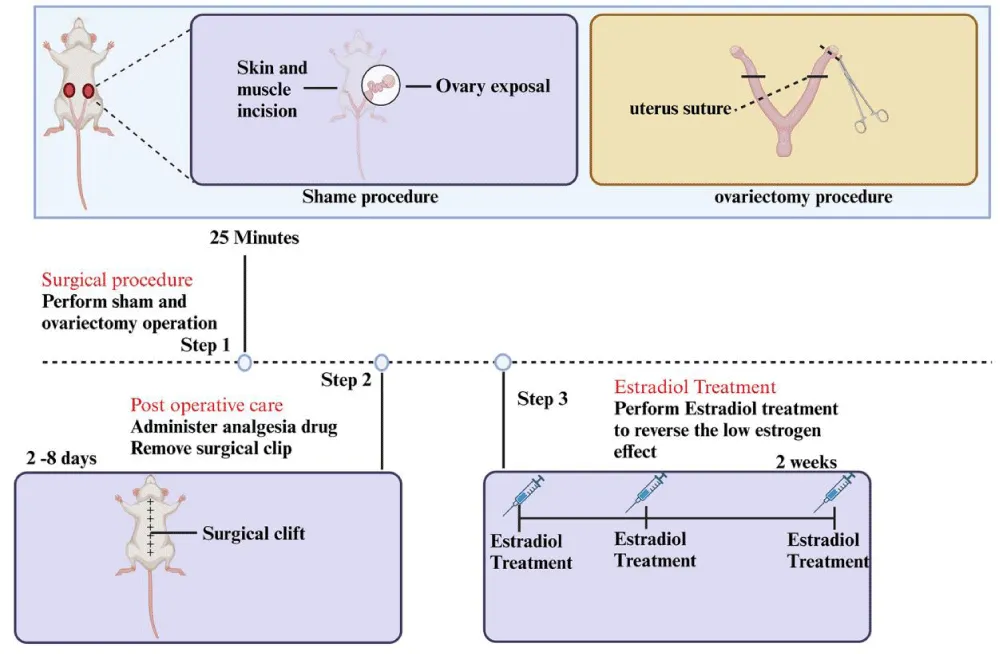
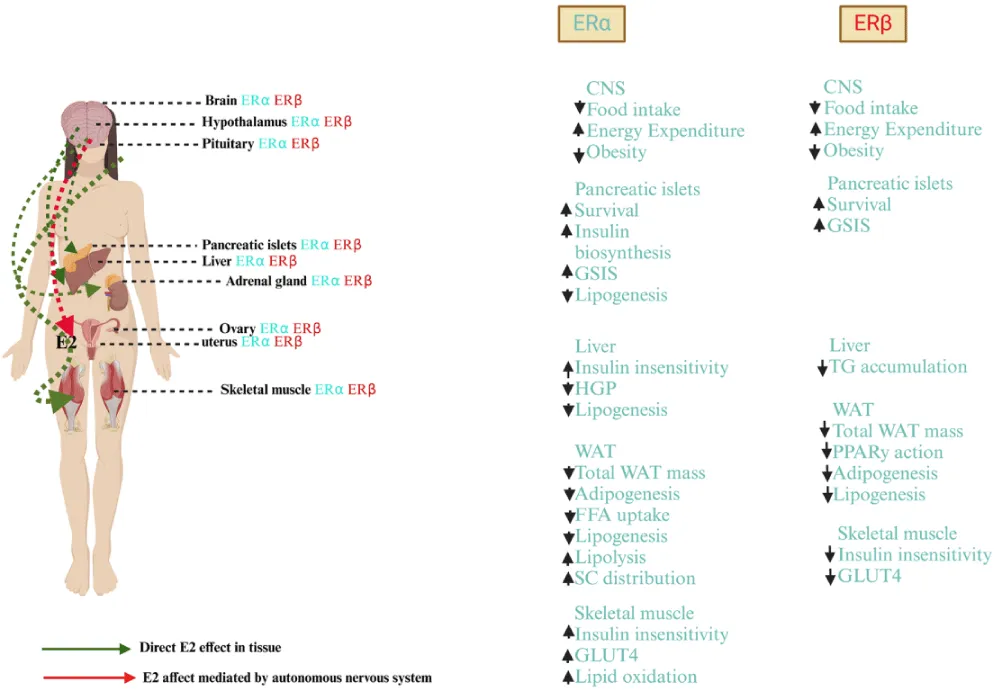
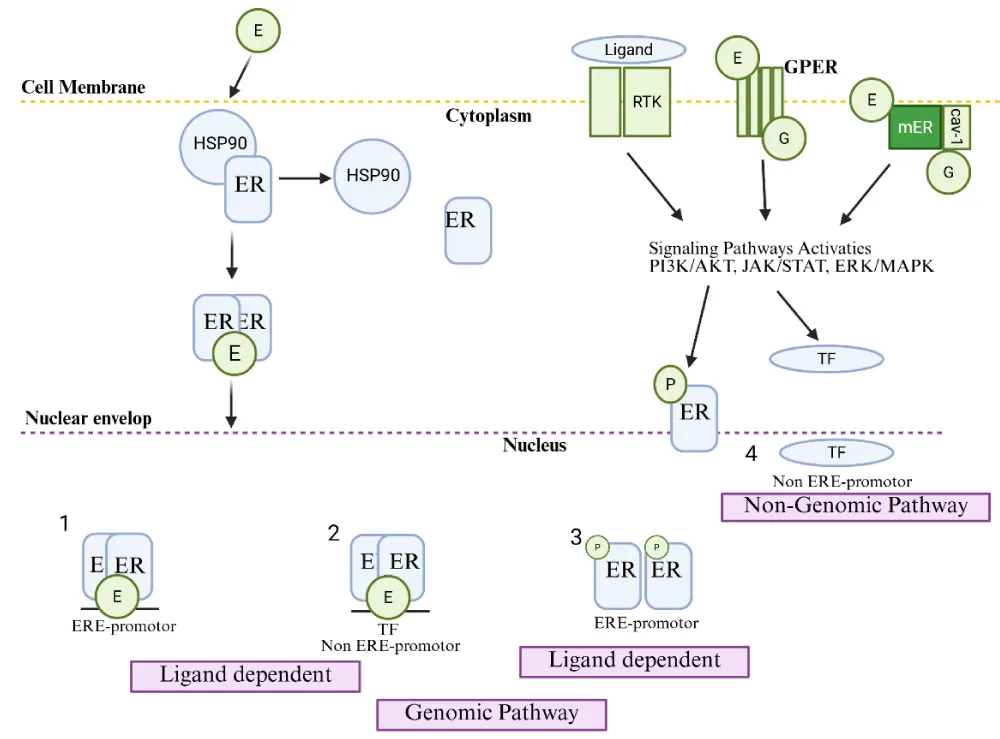
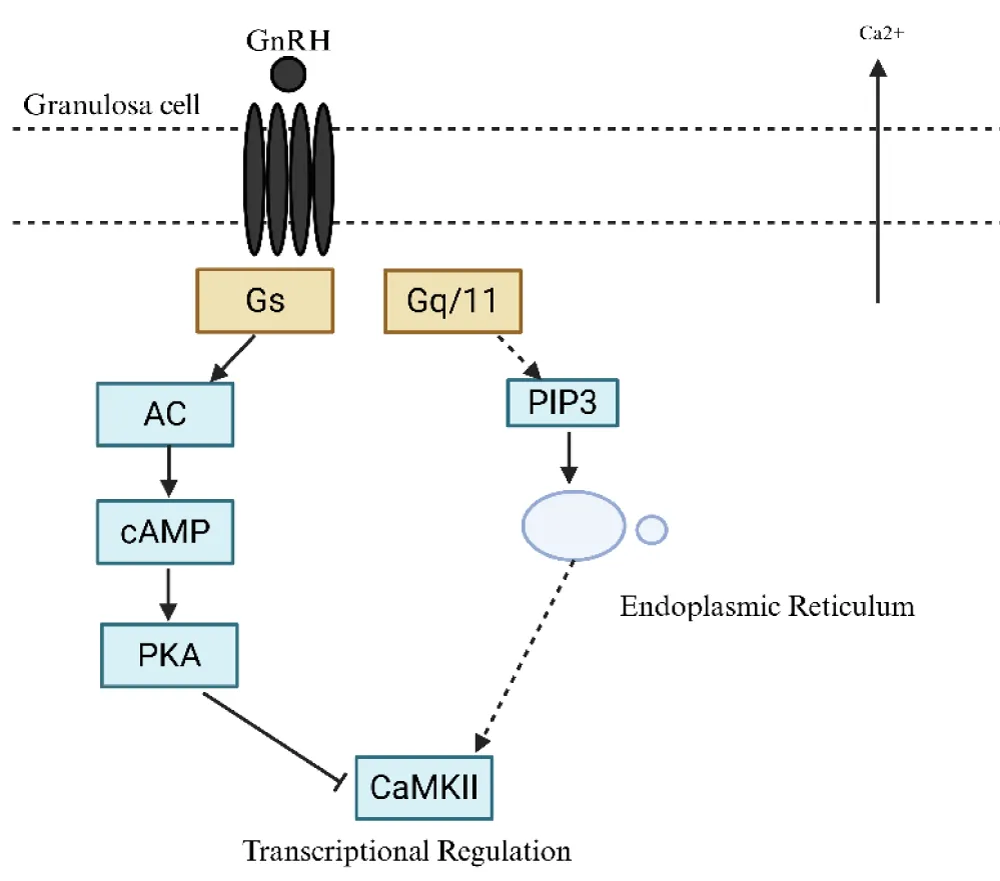
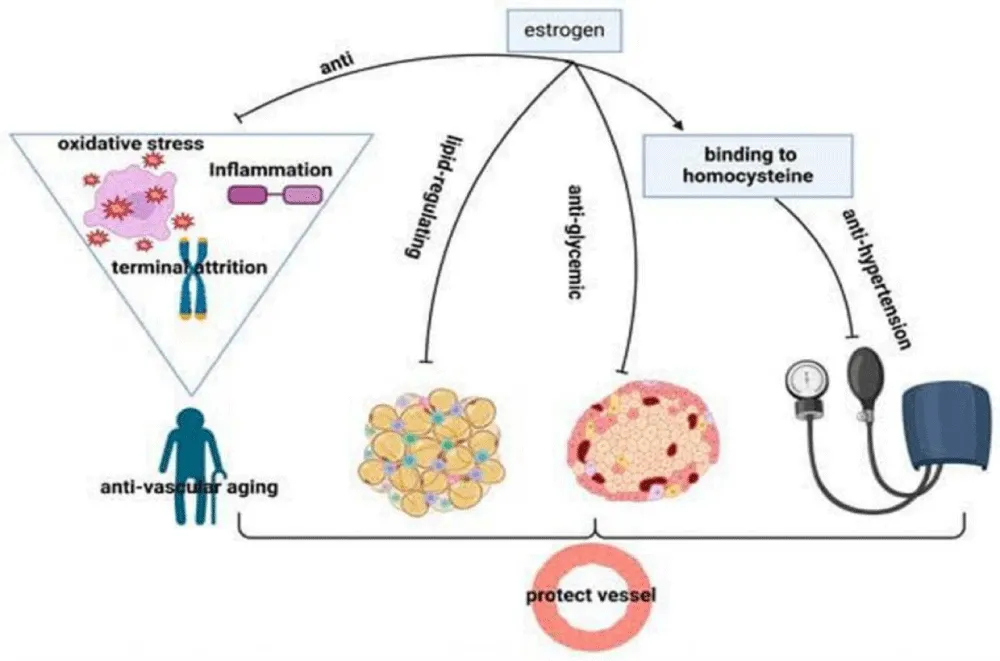


 Save to Mendeley
Save to Mendeley
