Global Journal of Obesity, Diabetes and Metabolic Syndrome
A Switch to the Duodenal Switch
Alcoy County Hospital and San Jorge Clinic, Alcoy, Alicante, Spain
Author and article information
Cite this as
Baltasar A, Pérez N, Bou R, Bengochea M, Serra C (2019) A Switch to the Duodenal Switch. Glob J Obes Diabetes Metab Syndr. 2019; 6(1): 1-9. Available from: 10.17352/2455-8583.000035
Copyright License
© 2019 Bahathiq AO. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Background: Duodenal Switch (DS) is a procedure that combines a Sleeve-Forming Gastrectomy (SFG) plus a biliopancreatic diversion (BPD) for the treatment of morbid obesity (MO) with a higher weight loss and resolution of comorbidities
Objectives: Report our experience with 950 consecutive DS operations performed from 1994 to 2016.
Setting: Mix of teaching and private institution in a county hospital of Spain.
Methods: We report an observational, retrospective and longitudinal study of 950 consecutive morbidly obese patients treated by DS surgery.
Results: We performed 518 open and 432 laparoscopic DS. Operative mortality was 0.84% (1.38% in DS and 0.38% in LDS), 4.84% had leaks, two had hepatic failures (0.2%) and malnutrition was present in 3.1%. At 5 years, the percentage of BMI lost was 80%, and percentage of expected BMI loss was more than 100%.
Conclusions: DS is the most aggressive bariatric surgery (BS) technique, but with the best long-term weight loss. We describe operative complications and long-term follow-up guidelines.
Duodenal Switch (DS) surgery consists of two operations, Sleeve-forming Gastrectomy (SFG) plus a Biliopancreatic diversion (BPD). It is the most complex technique in bariatric surgery (BS). DS combines restriction of food-intake and malabsorption in the small bowel.
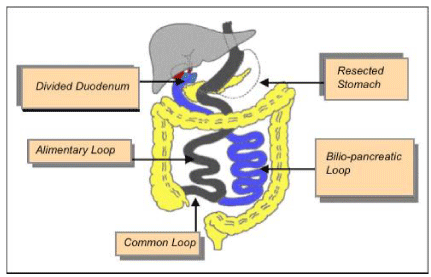
Doug Hess [1], initiated the DS in 1988 and described the three main components: 1) SFG to remove most of the stomach at the greater curvature, which reduces gastric volume but allows normal emptying; 2) Post pyloric division of the duodenum [D1] to allow emptying into the small bowel thru a Duodenal ileal anastomosis (DIA). 3) Diversion of 50% of proximal small bowel to cause malabsorption.
Hess [2], measures the entire small intestine, at low bowel tension, from ligament of Treitz to the ileocecal valve and uses 50% of its length as Biliopancreatic Loop (BPL), 40% as Alimentary Loop (AL) and 10% as Common Loop (CL).
Marceau [3], used the standard BPD until 1991 and one year later he changed to DS and he was the first to publish the DS [4], as first to publish and compared 252 BPD with distal gastrectomy and 465 DS with a 1.7% operative mortality. Lagacé [5], reported in 1995 the first good DS results in 61 patients with a new type of gastrectomy. Hess [6], uses a suture to invert the gastric serosa to cover the SFG staples and avoid leakage at the staple-line. Baltasar [7,8], describes the gastric part of the operation as SFG [7,8].
The CD [9-18], was considered a standard technique in the 90´s (Figure 1). Ren [19], made the first CDL in October 1999 and Baltasar [20-24], describes the first CDL in Europe in 2000 [25]. Paiva [26], in Brazil and Scopinaro [27], in Italy initiates in 2000 the standard laparoscopic DBP.
To measure weight loss results the Quetelet first to publish (BMI) is used. BMI = kg/m2, but… after reviewing 7,410 patients our mathematician developed the concept of predictive or expected BMI [28], to calculate BMI as Initial BMI (IBMI) any BMI superior to 25 and make the calculation not from IBMI but of IBMI in excess of 25. The % of excess weight lost (%EWL) are not equal in a MO patient grade 2 compared with the ones with triple obesity. Molina has reported that the expected BMI is useful [29].
Operative techniques
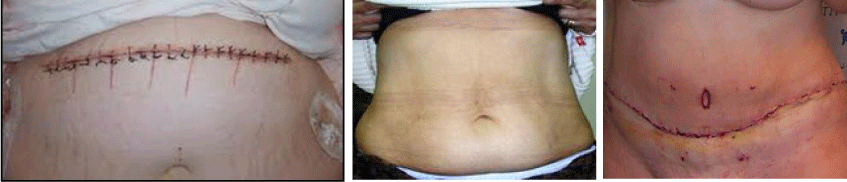
DS by Open Laparotomy (ODS): The patient is in Trendelenburg position. A supraumbilical transverse incision Is made between the two coastal margins (Figure 2 A-B). The round and triangular ligaments are sectioned. The gallbladder and appendix are removed. The open transverse approach gives better exposure and fewer hernias than the average laparotomy, but since laparoscopy its use today is restricted to re-operations.
The abdomen is closed in two layers of continuous Maxon. After weight loss, the scar shortens by one third in length (Figure 2b) and allows the superior wound to reach the pubic area in the abdominoplasty at the body contour surgery (Figure 2c). We started the ODS on 17.03.1994 with mean surgical times of 91 minutes.
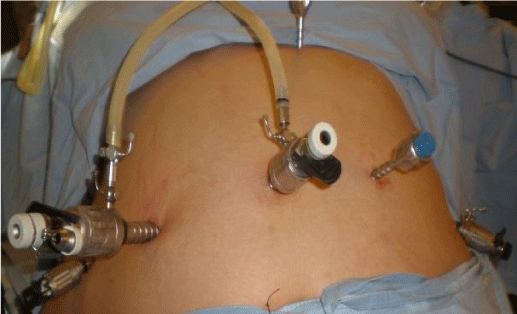
Laparoscopic CD (LDS): It’s done by three surgeons. Six ports are used (Figure 3). The Optical Trocar or “main port” is Ethicon # 12. It enters the abdomen under vision at the lateral edge of the right rectus, three fingers below the costal margin. A 10 mm central supraumbilical port is used for the mid-line camera.
There are four 5 mm ports, two sub-costal located on the right and left, one in the left hypochondrium and the other in the epigastrium used to retract the liver (Figure 3). To avoid slippage, we use Termanian trocars.
The entire small intestine is measured from the ileocecal valve to Treitz ligament with forceps at 5 cm steps to avoid lesions of the intestinal serosa. The CL is marked with a clip.
The upper AL is divided with linear stapler. The BPL starts at the first part of the duodenum (D1) and joins at the union of AL-CL as end--lateral Roux-Y (RY) jejunal-ileal anastomosis (JIA) with a continuous monoplane resorbable suture. The mesenteric defect is closed with a non-absorbable suture.
The greater gastric curvature vascular supply is divided starting 3 cm distal to the pylorus and up to the angle of His. A 12 mm nasogastric probe is passed along the inner curvature of the stomach and used as a guide to divide the stomach sequentially with linear staplers starting at the pylorus. Then both gastric walls, posterior and anterior of the staple-line and the omentum are sutured together with a continuous inverting suture to avoid torsion and leakage of the gastric tube.
A retro-duodenal tunnel is created in D1, distal to the right gastric artery, which allows the duodenal division with a linear stapler before the Oddi sphincter. The distal duodenal stump is also reinforced with continuous inverting suture.
The proximal AL passes retro-colic, on the right, and a Duodenum-Ileal Anastomosis (DIA) is performed. The operation has four sutures-lines (gastric staple-line reinforcement, distal duodenal stump, DIA, and the jejunum-ileal RY anastomosis) and drainages are placed, one next to the gastric tube and the other in DIA.
All anastomoses are hand-sutured with a monolayer continuous suture. All sutures start with the sliding and self-blocking knot of Serra-Baltasar [30-31] and end with Cuschieri one [32]. The stomach is removed without a protective bag. A Maxon suture closes the 12 mm port fascia to prevent hernias. We started the LDS on 10.5.2000 [20]. The average operative time was 155 ‘ after the first 50 cases.
At discharge, patients received prescriptions with vitamin A complex 20.000 UI, vitamin D 50.000 IU, calcium carbonate 1000 mg and ferrous sulfate 300, vitamins B1 and B2.
Material and Methods
950 consecutive MO patients (518 open and 432 laparoscopic) have been operated from 1994 to 2011, after a complete multidisciplinary preoperative evaluation and legal informed consent. 782 were women (82.3%) and 168 men (17.7%). The average age was 35 years (24-63). The 474 foreigners (361 US citizens, 17 from Canada, 73 from Norway and 25 from England) were intervened by the same team at the private center.
The average Initial BMI (IBMI) (Kg/m2) was 49.23 (Women-49.26 and Men-49.07). The obesity range was: a) Obesity, Grade 2 with comorbidities (IMCI < 40), 110 patients (mean 37.66); b) Morbidly Obese (MO) (IMCI 40-50), 464 patients (mean 45.11); c) Super/Super Obesity (SO) (BMI 50-60), 272 patients (mean 54.32) and D) Super/Super Obesity (SSO) IBMI > 60, 104 patients (mean > 66.50) and a patient had IBMI-100.
Comorbidities: 115 patients suffered diabetes mellitus Type 2 (DM2), 103 hypertension, 5 heart disease, 62 dyslipidemia, 19 Obstructive pulmonary disease, 16 osteoarthritis, 1 pseudo brain tumor.
Results
Main intraoperative complications: Three patients needed tracheostomy due to a failure of oral intubation and severe desaturation, without incidents.
In three patients, the 12 mm gastric tube did not pass past the esophageal-gastric junction and stapling of the stomach was done under visual control.
Surgical mortality: At 30 days occurred in six ODS patients (1.38%). The causes were: a) Leak in ADI - 1; b) Leak in RY, rhabdomyolysis and multiorgan failure - 1; c) Pulmonary embolism - 2; d) Leak in duodenal stump - 1; e) Leak in His angle - 1. Two LDS patients died (0.38%) due to pulmonary emboli. The average mortality of both groups was 0.84%.
Postoperative morbidity
1. Leaks. There were 46 Leaks for a total leak rate of 4.84%.
a) Leakage in his angle of His: 21 cases (2.3% incidence). 10 were treated with endoprosthesis, drainage or laparotomy and 3 with RY shunt. One of them died.
b) Leakage of the duodenal stump. One patient suffered a leak in the duodenal stump, it was repaired but died of sepsis. Since then, we have protected all staple-lines with an inverting suture and there were no more leaks.
c) DIA Leakage: 24 cases (2.5% incidence). This the most difficult anastomosis. 19 of them suffered early leaks, were successfully treated with drainage or re-do anastomosis, five cases had late leaks (up to 2-14 years later) and at re-operation the anastomosis was redone. In one case, the leak occurred 3 years after the intervention, as a gastro-pleural fistula and was treated with total gastrectomy.
d) RY leak. A patient had a small intestine diverticulum 110 cm from the ileocecal valve, and it was removed, and an open RY performed at the site without incident. There was a leakage and the diagnostic radiological tests did not clarify the cause, and with late diagnosis, he was re-operated and suffered rhabdomyolysis and multiple organ failure and died.
2. Pulmonary embolism. Two patients with IBMI-70 and IBMI-65 had embolism despite prophylactic therapy and died. Deep vein thrombosis in another case was successfully treated.
Top of Form:
3. Liver: a) Hepatic Disorders. Twelve patients underwent early liver function alterations with significant elevations of bilirubin (up to 15 and 29) that resolved with medical treatment.
b) Hepatic Failure. Two patients suffered liver failure (0.2%). The first occurred 6 months after surgery, included in the urgent list of liver transplantation but died due to lack of a donor. The second patient suffered liver failure three years after and she was treated successfully with a liver transplant plus BPD reversal and is healthy four years later. A patient died 13 years after ODS due to alcoholism.
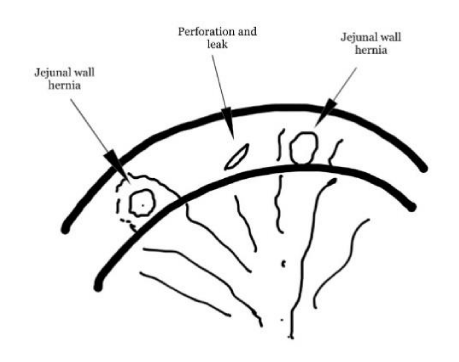
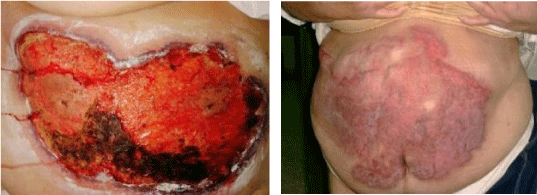
4) Caloric-protein malnutrition (CPM). Thirty-three patients (3.3%) developed CPM and 24 required CL elongation. 13 of them were open without complications. In 11 cases, the CL was laparoscopically lengthened and in two of them the small intestine was injured by the dissecting forceps that easily perforated the weaken wall (Figure 4). Both cases were diagnosed intra-operatively and repaired, but they died later due to re-leakage. Multiple hernias were found in the weak muscular wall between the mesentery vessels. These types of hernias have not been previously reported. Therefore, we recommend laparotomy for intestinal lengthening.
5) Pancreatic-cutaneous fistula. Both lesions of the skin healed spontaneously (Figure 5).
6) Hypoglycemia: Two patients had recurrent episodes of hypoglycemia that required BPD reversal.
7) Evisceration in four cases without consequences after proper repair.
8) Delayed bowel obstruction: 7 cases (incidence of 0.73%). We treated two in our unit and the others in other units with small bowel resections.
9) Beriberi. Three patients had vitamin B1 deficiency with neurological symptoms, changes in gait and spontaneous fall, all successfully corrected. This serious complication needs urgent of intravenous B1 administration.
10) Fractures due to the bad absorption of Ca that required Vitamin D25 plus Ca. Two cases presented that are now asymptomatic after proper care.
11) Toxic megacolon 16 years after surgery due to pseudo membranous colitis. The patient required a subtotal colectomy at 22 cm from the anus with terminal ileostomy and then later the ileum was re-anastomosed to the rectum.
12) Miscellaneous: Pneumonia-4. Seroma-4, wound infection-15. Digestive bleeding-5 (3 requiring laparotomy). Catheter-related sepsis-3.
Top of Form:
Long-term Mortality: An acute appendicitis not diagnosed at two years. Intestinal necrosis by internal hernia at 3 years. There were other causes of death not related to the CD (cancer, melanoma, myocardial infarction, etc.).
Weight loss results
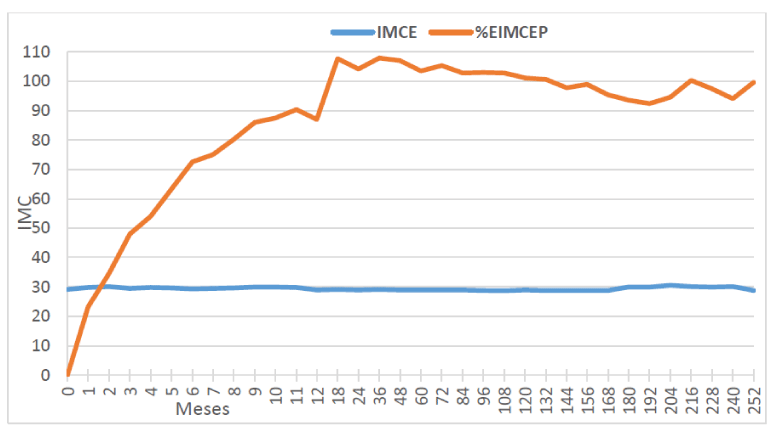
The figure 6 graph shows in blue the average expected BMI that was around 30 and in red the % expected excess BMI lost and that at the end was around 100%.
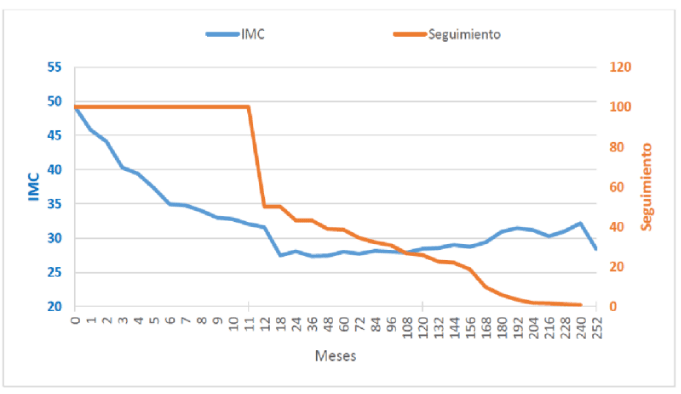
The figure 7 graph shows in blue the evolution of the average BMI over the months (it starts at 49 and ends at about 30) and in red the% of patients followed (starts at 100, continues with 38.5 at age 5, 25.8 at age 10 and ends in 0.9 at 20 years.
Therefore, the final %EWL was excellent at 90% in these series and is probably better than with any other obesity operation.
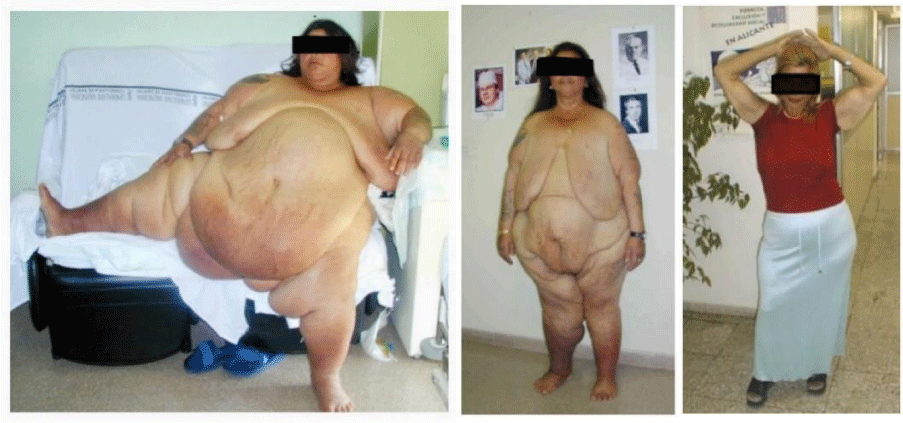
The DS is also very effective in the SSO. A patient with IBMI-100 fell to FBMI-34 (Figure 8).
Correction of comorbidities
Diabetes type II (DM2): DS is a very effective operation to treat diabetes. 98% of our patients are normoglycemic with normal glycosylated hemoglobin. Two non-diabetic patients suffered severe hypoglycemia and the BPD was reversed.
Hypertension was corrected in 73% of cases and sleep apnea in 100%.
Quality of life
We use the Horia-Ardelt [33], Classification of the BAROS scale to evaluate the changes in the quality of life of the patients. Changes after surgery included: self-esteem, physical activity, social activity, work activity plus sexual activity on a scale of-1 to 1. The average score was 2.03 (maximums of 3 points) in 348 patients, which means a significant improvement in their quality of life.
The gastrointestinal symptoms were evaluated from a minimum of 1 as excellent to a maximum of 5 as very bad. In the 558 patients evaluated, food intake of all types was 1.4, vomiting -1.3, appetite -1.96, type of stool (from pasty to liquid) - 2.2, frequency (without problem intolerable) -1.8, odor stool - 3.35, abdominal swelling- 2.26. Therefore, the sum of all measures was 12.14, for a total rating of 5 (excellent) to 35 (poor). The worst side effect was the bad odor of feces with an average of 3.35.
Discussion
DS has never been a popular therapy among surgeons; Hess [6], describes how after seeing a video of ours in Seatle-1996 at the ASBS Meeting, modified the procedure with an invagination suture of greater curvature and only had a leak in 188 cases. Very few surgeons continued making the DS and in fact a subdivision was created in the ASBS called “The switchers” with its own logo.
The DS remained unpopular, and we had to meet, for years, outside the congress venues as a separate group of 25-30 surgeons (Figure 9).
The unpopular “Switchers” have continued doing the intervention and many patients, even extra-national ones, knew about its advantages and looked for this therapy. We have not hidden the difficulties of the operation and especially its complications. We trained 72 bariatric surgeons and we have interacted live in several national and international congresses. An LDS video was second prize at IFSO 2002 in Sao-Paulo [34].
Three patients required an emergency tracheotomy [35,36]. In 2000, we used non-removable rigid stent [37], and then removable ones [38], to treat leaks. Nine patients required total gastrectomy [39]. In three patients we used a Y-Roux derivation [40,41] and this technique of 2007 is today the therapy of choice when the removable endoprosthesis fails [42].
We reported liver disorders [43] and failures [44]. Caloric-protein malnutrition [45,46]. Leaks [47], in laparoscopic lengthening. One patient suffered pancreas-cutaneous fistula [48].
Buchwald [49], at the 2004 consensus conference stated that in OM, surgery should be considered for patients with obesity greater than class IBMI 30-34.9 kg/m2 with associated comorbidities. Morbi-mortality should be low and obtain an optimal and sustained %EWL with minimal side effects. No BS technique is 100% successful or durable in all patients and there is no single standard procedure and probably never will be. In addition, BS cannot be the solution for the 1.7 billion with OM.
SFG leaks are a cause of significant morbidity manifested in the specific SFG meetings of Deitel [50]. The DS is a long and difficult procedure that requires experienced and experienced surgeons. Operative mortality should be <1% and morbidity <5%. Our 0.38% mortality for LDS has been quite low. Since patients with DS have four suture lines, early detection of leaks is essential.
Mason [51] drew attention to tachycardia as the first warning sign of leakage and no patient should be discharged with tachycardia.
Duncan [52], gives early discharges as outpatient surgery. Although our stay after LDS is 2-3 days, we instruct patients to enter to a telematic database [53,54], the pulse and temperature digitally, and notify us these parameters every four hours, for two weeks. Patients with significant change in these parameters need immediate and urgent consultation.
DeMaria [55], reported that 450 institutions and 800 surgeons participated in the 2009 BSCOE program of two years (2007-9). Only 0.89% of the 57,918 patients had the LDS done.
English [56], in ASMBS-2016 reports that obesity has increased alarmingly in the last 5 decades in the US, from 13.4% to 36.4%. In 2014, 215,666 operations were carried out in 795 accredited centers, SFG - 58.1%. And only 1187 BPD and 26% of them were LDS. The indirect costs of obesity and the overall economic impact are estimated at $ 1.42 trillion, 8.2% of the gross domestic product and more than double the defense spending. Obesity is the fifth most important risk factor for mortality in the world. And the reoperation rate was 14%. Revisions, including conversions, are increasing and suggest the need to develop better evidence-based algorithms to minimize the use of new operations. More effective initial BS operations are needed.
Nelson [57], using BOLD data from 2007-2010, identified 78,951 patients undergoing gastric bypass (GBP) or DS. Of these patients, 98% had GBP with IMCI-52 and only 2% had DS with IMCI-48. The DS was associated with longer surgical operation times, blood loss and longer hospital stays. The rates of early reoperation were higher in the DS group (3.3% vs. 1.5%). The drop in BMI was significantly higher in DS cases in all follow-up intervals (P> 0.05). In OS (BMI> 50) there was also a greater fall at 2 years, DS 79% compared to 67% in the GBP. The improvement of comorbidities (diabetes, hypertension and sleep apnea) was higher with DS (all P <0.05).
Iannelli [58], in 110 patients with BMI >50 found a reduction in the rate of postoperative complications when performing two-stage DS. Only 39 patients (35.5%) required the SFG and DBP avoided in 74.5% of the patients.
Biertho [59], did performed the DS in 1000 patients in 2006-2010. The conversion rate in the laparoscopy group was 2.6%. A postoperative death (0.1%) due to embolism. The mean hospital stay was shorter in the LDS group (6 to 6 days versus 7 to 9 days, P <0.01). The complication rates were 7.5%, without significant differences. No differences were found in abscesses or abdominal leaks.
Biertho [60], also treated 566 patients between 2011-2015 with LDS with BMI >49 and no mortality at 90 days. The average stay in the hospital was 4.5 days. Main complications greater than 30 days occurred in 3% of patients and minor ones 2.5%. The %EWL was 81% at 12, 88% at 24 and 83% at 36 months. Patients with HbA1C above 6% decreased from 38% to 1.4%. The readmission was 3.5% and 0.5% of the patients needed a new operation. The rate of complications in the short and medium term of LDS is like other mixed bariatric procedures and with excellent metabolic results.
Biron [61], studied the quality of life of 112 patients at 8.8 years. He observed some decrease in the quality of life in the long term after the initial changes that occurred 1-2 years after the surgery, during the so-called “honeymoon period”. The DS improves the specific quality of life of the disease in the short and long term.
Prachand [62], observed %EWL at 2 years in 350 patients of 54% in 152 GBP patients versus 68% in 198 patients with DS with only one deceased (P = not significant). %EWL at 3 years were DS = 68.9% was much higher than with GB = 54.9% and showed that the DS was more effective.
Strain [63], also had greater weight loss DS than GBP. The average weight decreased by 31.2% after the GBP and 44.8% after the DS.
Topart [64 in 2002-2009 had 83 DS and 97 GBP, with IMCI >55. After 3 years of follow-up, the average %EWL was 63.7% after the GBP and 84.0% after the DS (P> 0.0001). The results were significantly better with DS.
Våge [65], 2001-2008 treated 182 consecutive patients with DS without 30-day mortality. A patient needed surgery due to a leak; three patients due to bleeding and one due to leakage of bile. Six patients (3.2%) underwent surgical revision due to PCM, data similar to ours (3.3%).
In summary, patients undergoing DS constantly reduce BMI more than patients with GBP. So, why are there so few patients had DS?
Angrisani [66], reports that in 2018 there were 685,874 global bariatric operations; 92.6% primary and 7.4% revision. 96% of them surgical and 4% endoluminal. SFG-53.6%, GBP-30.1% and OAGBP-4.8% and only 1.3% LDS. Is the DS a too complex operation or the results are not so good? At the DS the SFG is irreversible but intestinal continuity can be restored to normal. The LDS can be performed in two stages, SFG as initial operation, in high-risk patients with IMCI >60.
Rabkin [67], reports that the DS is not associated with extensive nutritional deficiencies. Annual laboratory studies, which are required after any type of bariatric operation, appear to be sufficient to identify unfavorable trends. In selected patients, additional iron and calcium supplements are necessary.
Keshishian [68], performed a preoperative needle biopsy of the liver in 697 patients with DS. There was transient worsening of the AST (13%, P <.02) and ALT (130-160% of the reference levels, P <.0001) up to 6 months after the DS. And he observed a progressive improvement of 3 degrees in the severity of NASH and 60% in hepatic steatosis at 3 years after DS.
Type 2 diabetes
Buchwald [69], reports that DS and BPD have diabetes resolution rates that exceed 90%. In comparison, the GBP rate is approximately 70%. Tsoli [70], showed that SFG was comparable to DBP in the resolution of T2D but lower in dyslipidemia and blood pressure. In our first diabetes surgery [71], a low IBMI-35 patient had a and successfully performed a LDS without SFG to treat diabetes with excellent results.
Våge [72], thinks that DS is effective in T2D, hypertension and hyperlipidemia. And that the duration of diabetes and the age of the patients are the most important preoperative predictors.
DS done by stages
What part of the operation should be done first? The DBP or the SFG? Most surgeons recommend the SFG. However, Marceau [73], from 2001 to 2009 treated 1,762 patients all scheduled for DS. The procedures of the first stage were 48 BPD isolated without SFG and 53 cases only SFG. The conclusions were that SFG and BPD contribute independently to the beneficial metabolic results. Long-term results in terms of weight loss and resolution of metabolic abnormalities were better with isolated DBP than with isolated SFG. The results of one stage DS were better than those of two stages.
Moustarah [74 treated 49 OS patients with BPD without SFG. The initial weight was 144 Kg and IMCI-52.54 and the drop in the BMI of 14.5 Kg/m2 was very significant (p <0.001).
DBP without SFG is still rare as the only weight loss procedure; but in patients whose clinical indications justify the omission of SFG, the isolated DBP has better weight loss results. In this series, weight loss at 2 years compares favorably with other commonly performed bariatric operations.
We believe that BPD without SFG is totally reversible and this is an advantage, since the SFG can be added at any later time. The bowel surgery of the BPD is easier in the SO patient than the infra diaphragmatic of the SFG.
The mayor problem with DS patient follow-up is that other doctors and / or surgeons may not understand how to prevent or treat their long-term complications. Follow-up of DS patients is very important. At discharge a detailed technical explanation of the operation, each possible complication and their correction, and an extensive explanatory sheet of laboratory analyzes necessary for life.
Ankle swelling and serum albumin are the most important finding to detect long-term CPM. PTH and vitamin D25 (to detect calcium absorption and deficits and protect bone pathology), blood count to correct Fe deficits with intravenous therapy and deficits in all trace elements such as Mg, Cu.
In addition to leakage, the most serious long-term complication of is CPM. Surgical correction is simple when using the “Kiss-X” technique” as a jejunal-jejunal anastomosis to lengthen the CL, preferably by laparotomy.
Today we are privileged with endocrinology units, compared with 15 years ago when they did not believe in surgery and their support was very limited. So is the support of bariatric nursing and nutritionists.
As we do record all LDS, we provide patients with the digitized videos of the entire operation, because if a new operation is necessary, the surgeon should know all the details of the original technique.
Spanish surgeons made significant contributions on BPD such as Larrad [75,76], Solano [77], Ballesteros [78], Hoyuela [79], on the 90s. Recently Sánchez-Pernaute [80], proposed the now popular LDS of a single anastomosis (SADI).
A switch to the Duodenal Switch? How to change to the DS. A call!
Halawani [81], says that one third (34.9%) states that the DS has not yet said its last word since as of American adults are more obese. The LDS number in the US is less than 1% for the years 2011-2015. The DS should be added to the practice of the “Center of Excellence in Obesity” (CEO).
The DS is better than the GBP since it gives higher %EWL and has a lower rate of weight recovery. It conserves the pylorus and produces a slower gastric emptying. With adjustments to the length of the CL and the size of the gastric tube, any obese patient may be a candidate for DS.
Low BMI <50 patients may also be candidates. The DS is a viable option due to its flexibility. The surgeon can adjust the size of the SFG and alter the impact of the restriction. The length of CL can be variable.
The DS is good for chronic patients, using non-steroidal anti-inflammatories and steroids. The highest rate of early mortality compared to the SFG (0.28%) is slightly higher (0.43%). although the DS it is still considered to be a high risk procedure and the results should be analyzed with caution.
Conclusions
DS techniques are not common in MO management. The DS is the most complex technique and its learning curve is longer than other operations. It takes at least LDS 50 cases to standardize the technique. The LDS is safe and the most effective in terms of long-term weight loss results.
Ethical approval
All procedures involving human participants were conducted in the studies cited in this document in accordance with the ethical standards of national and institutional research committees and with the Declaration of Helsinki and amendments.
Informed consent
Informed consent was obtained from all individual participants included in the studies cited.
- Hess DS, Hess DW (1998) Biliopancreatic Diversion with a Duodenal Switch. Obes Surg 8: 267-282. Link: https://goo.gl/SXFP63
- Hess D (2003) Limb Measurements in Duodenal Switch. Obes Surg 13: 966. Link: https://goo.gl/y9RYyr
- Marceau P, Biron S, Bourque RA (1993) Biliopancreatic Diversion with a New Type of Gastrectomy. Obes Surg 3: 29-35. Link: https://goo.gl/LcXw1c
- Marceau P, Hould FS, Simard S (1998) Biliopancreatic Diversion with Duodenal Switch. World J. Surg 22: 947–954. Link: https://goo.gl/N21TBp
- Lagacé M, Marceau P, Marceau S (1995) Biliopancreatic Diversion with a New Type of Gastrectomy: Some Previous Conclusions Revisited. Obes Surg 5: 411-418. Link: https://goo.gl/xzp5u5
- Hess DS, Hess DW, Oakley RS (2005) The Biliopancreatic Diversion with the Duodenal Switch: Results beyond 10 Years. Obes Surg 15: 408-416. Link: https://goo.gl/t3rn9B
- Baltasar A (2012) Laparoscopic sleeve gastrectomy is a misnomer. SOARD 8: 127-131. Link: https://goo.gl/jXFQoY
- BaltasarA (2015) Sleeve-forming gastrectomy is the right terminology. Obes Surg 25: 935-932. Link: https://goo.gl/jH7CHr
- Baltasar A, Bou R, Bengochea M (1996) Hybrid bariatric Surgery: Duodenal Switch and bilio-pancreatic diversion. Video -Revista de Cirugía. VCR 12: 16-41. Link: https://goo.gl/nry3U7
- Baltasar M, Bou R, Cipagauta LA (1995) Hybrid Bariatric Surgery: Bilio-pancreatic Diversion and Duodenal Switch - Preliminary Experience. Obes Surg 5: 419-423. Link: https://goo.gl/4BnriA
- Baltasar A, del Río J, Bengochea M (1996) Cirugía híbrida bariátrica: Cruce duodenal en la derivación bilio-pancreática. Cir. Esp 59: 483-486.
- Baltasar A, del Río J, Escrivá C (1997) Preliminary results of the duodenal switch. Obes Surg 7: 500-504. Link: https://goo.gl/kbELSt
- Baltasar A, Bou R, Bengochea M (2001) Duodenal switch: an effective therapy for morbid obesity-intermediate results. Obes Surg 11: 54-58.
- Link: https://goo.gl/6AxHeE
- Baltasar A (2007) Hand-sewn Laparoscopic duodenal Switch. Surg Obes Relat Dis 3: 94-96.
- Biron S, Hould FS, Lebel L (2004) Twenty Years of Biliopancreatic Diversion: What is the Goal of the Surgery? Obes Surg 14: 160-164. Link: https://goo.gl/S2Pg1g
- Marceau P, Biron S, Hould FS (2007) Duodenal Switch: Long-Term Results. Obes Surg 17: 1421-1430. Link: https://goo.gl/LftmHb
- Marceau P, Biron S, Hould FS (2009) Duodenal switch improved standard biliopancreatic diversion: a retrospective study. Surg Obes Relat Dis 5: 43-47. Link: https://goo.gl/JJKeay
- Biertho L, Biron S, Hould FS (2010) Is biliopancreatic diversion with duodenal switch indicated for patients with body mass index <50 kg/m2? Surg Obes Relat Dis 6: 508–515. Link: https://goo.gl/tThzTA
- Ren CJ, Patterson E, Gagner M (2000) Early results of laparoscopic biliopancreatic diversion with duodenal switch: a case series of 40 consecutive patients. Obes Surg 10: 514-523. Link: https://goo.gl/oqWRCS
- Baltasar A, Bou R, Miró J (2001) Avances en técnica quirúrgica. Cruce duodenal por laparoscopia en el tratamiento de la obesidad mórbida: técnica y estudio preliminar. Cir. Esp 70: 102-104. Link: https://goo.gl/TusHgS
- Baltasar A, Bou R, Miró J (2002) Laparoscopic Biliopancreatic Diversion with Duodenal Switch: Technique and Initial Experience. Obes Surg 12: 245-248. Link: https://goo.gl/GK6A6q
- Baltasar A, Bou R, J Miró M (2003) Laparoscopic Duodenal Switch: Technique and Initial experience. Chir Gastroenterol 2003; 19:54-56. Link: https://goo.gl/Juzyjw
- Baltasar A (2007) Video case report. Hand-sewn laparoscopic duodenal switch. Surg Obes Relat Dis 3: 94–96.
- Baltasar A, Bou R, Bengochea M (2012) Laparoscopic hand-sewn Duodenal Switch. Video. BMI-Latina 2: 11-13.
- Weiner RA, Blanco-Engert R, Weiner S (2004) Laparoscopic biliopancreatic diversion with duodenal switch: Three different duodeno-ileal anastomotic techniques and initial experience. Obes Surg 14: 334-340. Link: https://goo.gl/fVYexZ
- Paiva D, Bernardes L, Suretti L (2001) Laparoscopic Biliopancreatic Diversion for the Treatment of Morbid Obesity: Initial Experience. Obes Surg 11: 619-622. Link: https://goo.gl/KbpAv9
- Scopinaro N, Marinari G, Camerini G (2002) Laparoscopic Standard Biliopancreatic Diversion: Technique and Preliminary Results. Obes Surg 12: 241-244. Link: https://goo.gl/g4sknK
- Baltasar A, Perez N, Serra C, Marceau P (2011) Weight Loss Reporting: Predicted BMI after Bariatric Surgery. Obes Surg 21: 367–372. Link: https://goo.gl/K7KNPg
- Molina A, Fàtima F, Vives M, Sanjaume AB, Blasco SB, et al. (2016) Usefulness of Baltasar’s expected body mass index as an indicator of bariatric weight loss surgery. Obes Surg 26: 2712-2717. Link: https://goo.gl/aNjKu4
- Serra C, Pérez N, Bou R, Baltasar A (2014) Sliding Self-Locking First Stitch and Aberdeen Knots in Suture Reinforcement with Omentoplasty of the Laparoscopic Gastric Sleeve Staple line. Obes Surg 24: 1739–1740. Link: https://goo.gl/kmDP6e
- Baltasar A, Bou R, Serra R, Bengochea M, Pérez N (2015) Use of self-locking knots in running intestinal bariatric sutures. Glob Surg 2: 100-101. Link: https://goo.gl/zxVKst
- Cuschieri A. Szabo Z, West D (1995) Tissue Approximation in Endoscopic Surgery: Suturing & Knotting. Informa Healthcare. Link: https://goo.gl/BqD28T
- Oria HE, Moorhead H (2009) Updated bariatric Analysis and Reporting Outcome System (BAROS). Surg Obes Relat Dis 5: 60-66. Link: https://goo.gl/DLTJL5
- Baltasar R, Bou J, Miró M, Bengochea N, Pérez (2012) Hand-sutured laparoscopic duodenal switch. Technical aspects. Obes Surg 12: 402.
- Baltasar A, Bou R, Bengochea M. Emergency Tracheotomy in Morbid Obesity. Sci Forschen. Obes Open Access 3. Link: https://goo.gl/hBwuP8
- Baltasar A, Bou R, Bengochea M, Serra C, Pérez N (2013) Difficult intubation and emergency tracheotomy in morbid obesity. BMI-Latina 3: 4-7. Link: https://goo.gl/kvZawc
- Baltasar A (2000) Wall-stent prosthesis for severe leak and obstruction of the duodenal switch. Obes Surg 10: 29.
- Serra C, Baltasar A, Andreo L (2007) Treatment of Gastric Leaks with Coated Self-expanding Stents after Sleeve Gastrectomy. Obes Surg 17: 866-872. Link: https://goo.gl/MkXLUL
- Serra C, Baltasar A, Pérez N (2006) Total Gastrectomy for Complications of the Duodenal Switch, with Reversal. Obes. Surg 16: 1082-1086. Link: https://goo.gl/uFbcyu
- Baltasar A, Bou R, Bengochea M (2007) Use of a Roux limb to correct esophago-gastric junction fistulas after sleeve gastrectomy. Obes Surg 17: 1408–1410. Link: https://goo.gl/wDZ2Uf
- Baltasar A, Serra C, Bengochea M (2008) Use of Roux limb as remedial surgery for sleeve gastrectomy fistulas. Surg Obes Relat Dis 4: 759–763. Link: https://goo.gl/cvK41u
- Mcheimeche H, Dbouk S, Saheli R (2018) Double Baltazar Procedure for Repair of Gastric Leakage Post-Sleeve Gastrectomy from Two Sites: Case Report of New Surgical Technique. Obes Surg 28: 2092–2095. Link: https://goo.gl/GkAQNM
- Baltasar A, Serra C, Pérez N (2014) Clinical Hepatic Impairment after the Duodenal Switch. Obes Surg 14: 77-78. Link: https://goo.gl/4R3Lui
- Baltasar A (2014) Liver failure and transplantation after duodenal switch. Surg Obes Relat Dis 10: c93-c96. Link: https://goo.gl/fXaSDy
- Baltasar A, Serra C (2010) Treatment of complications of duodenal switch and sleeve gastrectomy. In: Deitel M, Gagner M, Dixon JB, Himpens J, Madan AK, editors. Handbook of obesity surgery. Toronto: FD Communications; 156–161.
- Baltasar A, Bou R, Bengochea M, Serra C (2011) Malnutrición calórico-proteica. Tres tipos de alargamiento de asa común. BMI-Latina 5: 96-97.
- Baltasar A, Bou R, Bengochea M (2010) Fatal perforations in laparoscopic bowel lengthening operations for malnutrition. Surg Obes Relat Dis 6: 572–574. Link: https://goo.gl/X6KYu2
- Bueno J, Pérez N, Serra C (2004) Fistula pancreato-cutánea secundaria a pancreatitis postoperatoria tras cruce duodenal laparoscópico. Cir. Esp 76: 184-186. Link: https://goo.gl/Y2xzPa
- Buchwald H (2005) 2004 ASBS. Consensus Conference Statement Bariatric surgery for morbid obesity: Health implications for patients, health professionals, and third-party payers. Surg Obes Relat Dis 1: 371-381. Link: https://goo.gl/nX5pqQ
- Deitel M, Gagner M, Erickson AL (2011) Third International Summit. Current Status of sleeve gastrectomy. Surg Obes Relat Dis 7: 749–759. Link: https://goo.gl/44vBPs
- Mason R (1995) Diagnosis and treatment of rapid pulse. Obes Surg 3: 341. Link: https://goo.gl/8b4TXP
- Duncan T, Tuggle K, Larry Hobson L (2011) PL-107. Feasibility of laparoscopic gastric bypass performed on an outpatient basis. Surg Obes Relat Dis 7: 341–342. Link: https://goo.gl/tXr7jv
- Baltasar A (2015) Simple bariatric tele-medicine. BMI-Latina 5: 901-902. Link: https://goo.gl/SAUnYR
- Baltasar A (2017) Assistance in Bariatric Surgery Journal of Obesity & Eating Disorders 3: 28.
- DeMaria E, Pate V, Warthen M (2010) Original article Baseline data from American Society for Metabolic and Bariatric Surgery-designated Bariatric Surgery Centers of Excellence using the Bariatric Outcomes Longitudinal Database. Surg Obes Relat Dis 6: 347-355. Link: https://goo.gl/EpZ44R
- English W, DeMaria M, Brethauer SA (2016) American Society for Metabolic and Bariatric Surgery estimation of metabolic and bariatric procedures performed in the United States in 2016. Surg Obes Relat Dis 14: 259-263. Link: https://goo.gl/owz977
- Nelson D, Blair KS, MD; Martin M (2012) Analysis of Obesity-Related Outcomes and Bariatric Failure Rates with the Duodenal Switch vs Gastric Bypass for Morbid Obesity. Arch Surg 147: 847-854. Link: https://goo.gl/28E7t8
- Iannelli A, Schneck AS, Topart P (2013) Laparoscopic sleeve gastrectomy followed by duodenal switch, in selected patients versus single-stage duodenal switch for super-obesity: case– control study. Surg Obes Relat Dis 9: 531-538. Link: https://goo.gl/KJzVDt
- Biertho L, Lebel S, Marceau S (2013) Perioperative complications in a consecutive series of 1000 duodenal switches. Surg Obes Relat Dis 9: 63-68. Link: https://goo.gl/j83ooj
- Biertho L, Simon‑Hould F, Marceau S (2016) Current Outcomes of Laparoscopic Duodenal Switch. Ann Surg Innov. Res 10: 1. Link: https://goo.gl/U16xaS
- Biron S, Biertho L, Marceau S (2018) Long-term follow-up of disease-specific quality of life after bariatric surgery. Surg Obes Relat Dis 14: 658-664. Link: https://goo.gl/FL9HWQ
- Prachand V, DaVee R, Alberdy JA (2006) Duodenal switch provides superior weight loss in the super-obese (BMI>50) compared with gastric bypass. Annals of Surg 244: 611-619. Link: https://goo.gl/k1r7Fo
- Strain CW, Gagner M, Inabnet WB (2007) Comparison of effects of gastric bypass and biliopancreatic diversion with duodenal switch on weight loss and body composition 1-2 years after surgery. Surg Obes Relat Dis 3: 31-36. Link: https://goo.gl/bV5TXs
- Topart P, Becouarn G, Salle A (2011) Five-year follow-up after biliopancreatic diversion with duodenal Switch. Surg Obes Relat Dis 7: 199-205. Link: https://goo.gl/uezBo1
- Våge V, Gåsdal R, Lakeland C (2011) The Biliopancreatic Diversion with a Duodenal Switch (BPDDS): How Is It Optimally Performed? Obes Surg 21: 1864-1869. Link: https://goo.gl/Egb2Q6
- Angrisani L, Santonicola A, Iovino P (2018) IFSO Worldwide Survey 2016: Primary, Endoluminal, and Revisional Procedures. Obes Surg 28: 3783-3794. Link: https://goo.gl/ESLVxY
- Rabkin R, Rabkin JM, Metcalf B (2004) Nutritional Markers following Duodenal Switch for Morbid Obesity. Obes Surg 14: 84-90. Link: https://goo.gl/sVPGxZ
- Keshishian A, Zahriya K, Willes EB (2005) Duodenal Switch Has No Detrimental Effects on hepatic Function and Improves Hepatic Steatohepatitis after 6 Months. Obes Surg 15: 1418-1423. Link: https://goo.gl/B7r7j5
- Buchwald H, Estok R, Fahrbach K (2009) Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med 122: 248-256. Link: https://goo.gl/PHGCZk
- Tsoli M, Chronaiou A, Kehagias I (2013) Hormone changes and diabetes resolution after biliopancreatic diversion and laparoscopic sleeve gastrectomy: a comparative prospective study. Surg Obes Relat Dis 9: 667–678. Link: https://goo.gl/JkNDo8
- Baltasar A (2016) Primera operación metabólica de diabetes en España. BMI-Latina 6: 996-999. Link: https://goo.gl/Zk91pS
- Våge V, Roy M, Nilsen M, Berstad M (2013) Predictors for Remission of Major Components of the Metabolic Syndrome after Biliopancreatic Diversion with Duodenal Switch. Obes Surg 23: 80-86. Link: https://goo.gl/qbsbFU
- Marceau P, Biron S, Marceau SK (2014) Biliopancreatic Diversion-Duodenal Switch: Independent Contributions of Sleeve Resection and Duodenal Exclusion. Obes Surg 24: 1843-1849. Link: https://goo.gl/J4jFVJ
- Moustarah F, Marceau S, Lebel S (2010) Weight Loss after Duodenal Switch without Gastrectomy for the treatment of Severe Obesity: Review of a single institution Case Series of Duodeno-Ileal Intestinal Bypass. Can J Surg 53: 51.
- Larrad A, Moreno B, Camacho A (1995) Resultados a los dos años de la técnica de Scopinaro en el tratamiento de la obesidad mórbida. Cir. Esp 58: 23.
- Larrad A, Sánchez-Cabezudo C, Borrajo (2007) Short-, Mid- and Long-Term Results of Larrad Biliopancreatic Diversion. Obes Surg 17, 202-210. Link: https://goo.gl/yV9G8H
- Solano J, Resa JJ, Fatas JA (2003) Derivación bilio-pancreática laparoscópica para el tratamiento de la obesidad mórbida. Aspectos técnicos y análisis de los resultados preliminares. Cir. Es p 7: 347-350. Link: https://goo.gl/De8fe2
- Ballesteros M, González T, Urioste A (2016) Biliopancreatic Diversion for Severe Obesity: Long-Term Effectiveness and Nutritional Complications. Obes Surg 26: 38-44. Link: https://goo.gl/sbN8wc
- Hoyuela C, Veloso E, Marco C (2006) Derivación bilio-pancreática con cruce duodenal y su impacto en las complicaciones posoperatorias. Cir. Esp 80: 1-250-175.
- Sánchez-Pernaute A, Rubio M, Torres A (2007) Proximal Duodenal–Ileal End-to-Side Bypass with Sleeve Gastrectomy: Proposed Technique. Modern surgery: Technical innovation Obes Surg 17: 1614-1618. Link: https://goo.gl/YXCo9S
- Halawani HM, Antanavicius G, Bonanni F (2017) How to Switch to the Switch: Implementation of Biliopancreatic Diversion with Duodenal Switch into Practice. Obes. Surg 27: 2506-2509. Link: https://goo.gl/CQdXNC
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
