Global Journal of Obesity, Diabetes and Metabolic Syndrome
Obesity and Type 2 Diabetes Mellitus: A Bidirectional Interplay, Pathogenic Mechanisms and Clinical Management
Department of Laboratory Medicine, Honghe Prefecture First People’s Hospital, Gejiu City, Yunnan Province, 661000, China
Author and article information
Cite this as
Cong-li G, et al. Obesity and Type 2 Diabetes Mellitus: A Bidirectional Interplay, Pathogenic Mechanisms and Clinical Management. Glob J Obes Diabetes Metab Syndr. 2026; 13(1): 5-16. Available from: 10.17352/gjodms.000070
Copyright License
© 2026 Cong-li G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Obesity, especially central obesity, is a well-recognized major modifiable risk factor for the development of type 2 diabetes mellitus (T2DM), and the comorbidity of obesity and T2DM has become a global public health crisis with rapidly increasing prevalence. The relationship between obesity and T2DM is not a simple causal association but a complex bidirectional interplay: excessive adiposity, particularly visceral adipose tissue accumulation, triggers a cascade of metabolic and inflammatory abnormalities that lead to insulin resistance (IR) and pancreatic β-cell dysfunction, the core pathological features of T2DM; conversely, T2DM-induced metabolic disorders further exacerbate adipose tissue dysfunction, abnormal fat distribution and weight gain, forming a vicious cycle. This review systematically elaborates the bidirectional pathological link between obesity and T2DM, clarifies the key underlying mechanisms including chronic low-grade inflammation, adipose tissue dysfunction, gut micro biota dysbiosis, mitochondrial dysfunction and genetic epigenetic regulation. We further summarize the current clinical strategies for the comprehensive management of obesity-related T2DM, covering lifestyle intervention, pharmacotherapy and bariatric surgery, and prospect the emerging research directions such as targeted therapy for adipose tissue inflammation, gut micro biota modulation and precision medicine. This review aims to provide a comprehensive understanding of the intimate association between obesity and T2DM, and offer a reference for the optimized clinical management and novel therapeutic development for patients with obesity and T2DM comorbidity.
The global epidemics of obesity and type 2 diabetes mellitus (T2DM) have become two of the most pressing public health challenges in the 21st century, with their prevalence increasing in a parallel and mutually reinforcing manner [1]. According to the World Health Organization (WHO) Global Status Report on Obesity, Overweight and Diet 2024, the global number of overweight adults reached 2.1 billion in 2023, among whom over 760 million were obese [2]. Meanwhile, the International Diabetes Federation (IDF) World Diabetes Atlas 2023 reported that the global number of adult T2DM patients reached 537 million in 2023 (projected to 579 million in 2024), accounting for approximately 90% of all diabetes cases, and more than 80% of T2DM patients are overweight or obese [3]. Notably, the comorbidity of obesity and T2DM is not only a simple superposition of two diseases but also leads to a significant increase in the risk of macro vascular and micro vascular complications, such as cardiovascular disease, diabetic nephropathy, retinopathy and neuropathy, as well as higher mortality rates [4].
Obesity and T2DM share common risk factors including unhealthy lifestyle (high-calorie diet, physical inactivity), genetic susceptibility, aging and environmental factors [5]. However, the relationship between them is far more complex than co-occurrence: epidemiological studies have confirmed that obesity, especially central obesity characterized by visceral adipose tissue (VAT) accumulation, is the strongest independent risk factor for T2DM; individuals with a body mass index (BMI) >30 kg/m² have a 5-10 times higher risk of developing T2DM than those with normal BMI [6]. Conversely, T2DM is also an independent risk factor for weight gain and adipose tissue dysfunction: insulin resistance and hyperinsulinemia in T2DM patients lead to abnormal fat synthesis and distribution, further exacerbating obesity [7]. This bidirectional interplay forms a vicious cycle that accelerates the progression of both diseases and increases the difficulty of clinical management.
In recent years, with the in-depth research on adipose tissue biology, metabolic immunology and molecular genetics, the underlying mechanisms of the association between obesity and T2DM have been gradually elucidated. Chronic low-grade inflammation, adipose tissue dysfunction, gut micro biota symbiosis and mitochondrial dysfunction are recognized as the core links connecting obesity and T2DM [8]. At the same time, clinical management strategies for obesity-related T2DM have been continuously optimized, from traditional lifestyle intervention to novel anti-obesity and anti-diabetic drugs, and bariatric surgery with long-term efficacy, providing multiple options for clinical practice [9]. This review comprehensively sorts out the bidirectional pathological relationship between obesity and T2DM, clarifies the key pathogenic mechanisms, summarizes the current clinical management strategies and prospect the emerging therapeutic directions, in order to provide a theoretical basis and clinical reference for the prevention and treatment of obesity and T2DM comorbidity.
Bidirectional pathological interplay between obesity and T2DM
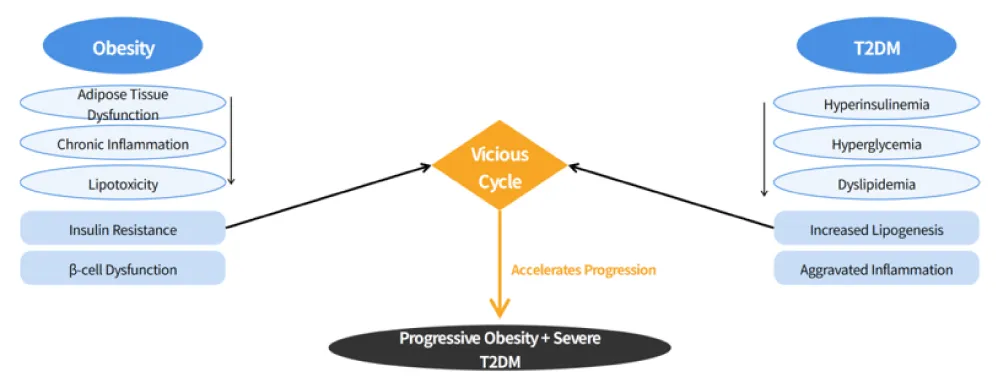
The association between obesity and T2DM is a typical bidirectional regulatory relationship: obesity is the primary driver of insulin resistance and pancreatic β-cell dysfunction, the two core pathological features of T2DM; while T2DM-induced metabolic disorders further aggravate adipose tissue dysfunction and abnormal fat metabolism, forming a vicious cycle that promotes the progression of both diseases (Figure 1).
Figure 1 illustrates the bidirectional pathological interplay between obesity and type 2 diabetes mellitus (T2DM). Obesity, particularly central obesity, induces insulin resistance and pancreatic β-cell dysfunction via adipose tissue dysfunction, chronic inflammation, and lipo toxicity, leading to T2DM onset. Conversely, T2DM-related hyperinsulinemia, hyperglycemia, and dyslipidemia exacerbate adipocyte hypertrophy and inflammation, forming a vicious cycle that accelerates the progression of both conditions.
Obesity induces the development and progression of T2DM
Excessive adiposity, especially the accumulation of VAT, is the initiating factor of T2DM pathogenesis. Adipose tissue is no longer considered a simple energy storage organ but a key endocrine and paracrine organ that secretes a variety of bioactive substances, including adipokines, cytokines, free fatty acids (FFAs) and hormones [10]. In obese individuals, the expansion of adipose tissue (especially VAT) leads to a series of structural and functional abnormalities, which trigger insulin resistance in peripheral tissues (liver, skeletal muscle, adipose tissue itself) and progressive pancreatic β-cell dysfunction, ultimately leading to the onset of T2DM [11].
Insulin resistance is the earliest and most core pathological change in obesity-induced T2DM. In obese individuals, excessive FFAs released by hypertrophic adipocytes cause lipid accumulation in the liver and skeletal muscle, leading to impaired insulin signal transduction and reduced glucose uptake and utilization [12]. Meanwhile, abnormal secretion of adipokines in obese individuals further exacerbates insulin resistance: the levels of anti-inflammatory and insulin-sensitizing adipokines (e.g., adiponectin) are significantly reduced, while the levels of pro-inflammatory and insulin-resistant adipokines (e.g., leptin, resistin, visfatin) are increased. Leptin resistance is a common phenomenon in obese individuals, which leads to impaired central appetite regulation and persistent weight gain, while also reducing insulin sensitivity in peripheral tissues [13,14].
Pancreatic β-cell dysfunction is the necessary condition for the onset of T2DM in obese individuals with insulin resistance. In the early stage of obesity, the pancreas compensatorily increases insulin secretion to overcome insulin resistance, resulting in hyperinsulinemia [15]. However, long-term hyperinsulinemia, chronic inflammation and high glucose and lipid levels cause oxidative stress and endoplasmic reticulum stress in pancreatic β-cells, leading to impaired insulin secretion, reduced β-cell mass and even β-cell apoptosis [16]. When the compensatory secretion of insulin by β-cells can no longer compensate for insulin resistance, fasting hyperglycemia and postprandial hyperglycemia occur successively, marking the onset of T2DM [17].
T2DM exacerbates obesity and adipose tissue dysfunction
T2DM is not only the consequence of obesity but also an important factor that exacerbates adipose tissue dysfunction and abnormal fat metabolism, forming a vicious cycle between the two diseases. The core metabolic abnormalities of T2DM, including insulin resistance, hyperinsulinemia, hyperglycemia and dyslipidemia, directly or indirectly lead to abnormal fat synthesis, distribution and breakdown, further aggravating obesity and adipose tissue inflammation [6,18].
Hyperinsulinemia is the key link in T2DM-induced weight gain. Insulin is a potent anabolic hormone that promotes glucose uptake and lipogenesis in adipose tissue, and inhibits lipolysis [19]. In T2DM patients with insulin resistance, the pancreas secretes a large amount of insulin to compensate, resulting in chronic hyperinsulinemia. Although the insulin sensitivity of peripheral tissues is reduced, the lipogenic effect of insulin on adipose tissue is still preserved, leading to excessive fat synthesis and accumulation, especially the further expansion of VAT [20]. Meanwhile, hyperglycemia in T2DM patients leads to increased glucose uptake in adipose tissue through the glucose transporter 4 (GLUT4) independent of insulin, which is converted into triglycerides and stored, further promoting adipocyte hypertrophy [21].
T2DM also exacerbates adipose tissue dysfunction and chronic inflammation. Hyperglycemia and dyslipidemia in T2DM patients cause increased oxidative stress and endoplasmic reticulum stress in adipocytes, leading to further impairment of adipokine secretion: adiponectin levels are further reduced, while pro-inflammatory cytokines (e.g., TNF-α, IL-6) are further increased. In addition, T2DM-induced metabolic disorders lead to the infiltration of more immune cells (e.g., M1-type macrophages) into adipose tissue, forming crown-like structures and aggravating chronic low-grade inflammation, which in turn further exacerbates insulin resistance and β-cell dysfunction [22,23]. This vicious cycle between T2DM and obesity continuously accelerates the progression of both diseases and increases the risk of complications.
Key pathogenic mechanisms linking obesity and T2DM
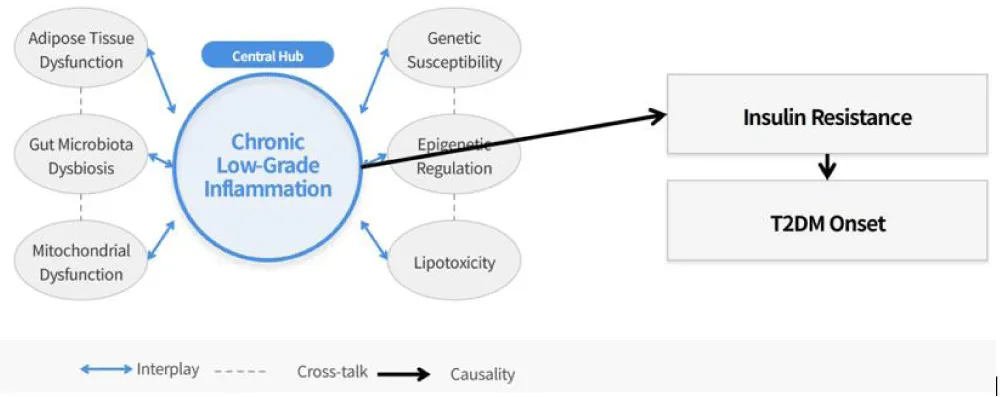
The complex bidirectional interplay between obesity and T2DM is mediated by multiple interrelated pathogenic mechanisms, among which chronic low-grade inflammation, adipose tissue dysfunction, gut microbiota dysbiosis, mitochondrial dysfunction and genetic epigenetic regulation are the most critical and well-studied (Figure 2). These mechanisms are not independent but interact with each other, forming a complex regulatory network that connects obesity and T2DM.
Key Pathogenic Mechanisms Linking Obesity and Type 2 Diabetes Mellitus. Multiple interrelated pathogenic mechanisms mediate the association between obesity and T2DM, with chronic low-grade inflammation as the central hub, and adipose tissue dysfunction, gut microbiota dysbiosis, mitochondrial dysfunction and genetic/epigenetic regulation interacting with each other to form a complex regulatory network that promotes insulin resistance and T2DM onset.
Chronic low-grade inflammation
Chronic low-grade inflammation is the central hub linking obesity and T2DM, and is the common pathological basis of both diseases [24]. In obese individuals, the expansion of adipose tissue (especially VAT) leads to the infiltration of a large number of immune cells, mainly M1-type macrophages, as well as T lymphocytes, neutrophils and mast cells [24,25]. These immune cells and hypertrophic adipocytes secrete a variety of pro-inflammatory cytokines, including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), monocyte chemoattractant protein-1 (MCP-1) and C-reactive protein (CRP), leading to a state of systemic chronic low-grade inflammation [26].
Pro-inflammatory cytokines exacerbate insulin resistance by impairing insulin signal transduction. TNF-α and IL-6 can inhibit the phosphorylation of insulin receptor substrate-1 (IRS-1) and insulin receptor (IR), block the downstream PI3K/Akt signal pathway, and reduce the translocation of GLUT4 to the cell membrane, thereby reducing glucose uptake and utilization in liver, skeletal muscle and adipose tissue [27]. Meanwhile, chronic inflammation also induces pancreatic β-cell dysfunction: pro-inflammatory cytokines activate the nuclear factor-κB (NF-κB) pathway in β-cells, leading to oxidative stress and endoplasmic reticulum stress, which in turn cause impaired insulin secretion and β-cell apoptosis [27,28]. In T2DM patients, chronic inflammation is further aggravated by hyperglycemia and dyslipidemia, forming a pro-inflammatory cycle that continuously promotes the progression of obesity and T2DM.
Adipose tissue dysfunction
Adipose tissue dysfunction is the initiating factor of the pathological link between obesity and T2DM, and is mainly manifested as abnormal adipose tissue expansion, impaired adipokine secretion and abnormal lipid metabolism. Adipose tissue expansion in obesity is divided into two forms: hyperplastic expansion (increase in adipocyte number) and hypertrophic expansion (increase in adipocyte size) [9,29]. Hyperplastic expansion is a healthy expansion mode with small adipocytes, normal lipid metabolism and low inflammatory level; while hypertrophic expansion is an unhealthy mode with large adipocytes, impaired lipid storage capacity, increased FFAs release and high inflammatory level [30]. Obese individuals, especially those with central obesity, are mainly characterized by hypertrophic expansion of VAT, which is the core cause of adipose tissue dysfunction.
Abnormal secretion of adipokines is the main manifestation of adipose tissue dysfunction and a key link in regulating insulin sensitivity and glucose metabolism. Adiponectin, the most abundant adipokine secreted by adipocytes, has insulin-sensitizing, anti-inflammatory and anti-atherosclerotic effects. Its levels are significantly reduced In patients with obesity and T2DM, and the reduction degree is positively correlated with the severity of insulin resistance and β-cell dysfunction [10,31]. Leptin, a hormone that regulates appetite and energy balance, is overexpressed in obese individuals, but leptin resistance occurs due to the downregulation of leptin receptors in the hypothalamus, leading to impaired appetite suppression and energy expenditure promotion, as well as reduced insulin sensitivity. Other adipokines such as resistin, visfatin and chemerin are increased in patients with obesity and T2DM, and they promote insulin resistance and chronic inflammation through different mechanisms [14,32].
Abnormal lipid metabolism in adipose tissue is another important manifestation of dysfunction. Hypertrophic adipocytes have impaired lipid storage capacity, leading to increased lipolysis and excessive release of FFAs into the circulation [33]. Elevated circulating FFAs cause lipid accumulation in non-adipose tissues (liver, skeletal muscle, pancreas), a phenomenon known as lipotoxicity. Lipotoxicity leads to impaired insulin signal transduction in peripheral tissues, and also causes oxidative stress and endoplasmic reticulum stress in pancreatic β-cells, leading to β-cell dysfunction [13,34].
Gut microbiota dysbiosis
Gut micro biota, the complex microbial community residing in the human gastrointestinal tract, plays an important role in regulating host energy metabolism, immune function and glucose homeostasis, and its dysbiosis is an important environmental factor linking obesity and T2DM [35]. The composition and abundance of gut micro biota in patients with obesity and T2DM are significantly different from those in healthy individuals: the ratio of Firmicutes to Bacteroidetes (F/B ratio) is significantly increased, the abundance of short-chain fatty acid (SCFA)-producing bacteria (e.g., Bifidobacterium, Lactobacillus) is reduced, and the abundance of pro-inflammatory bacteria (e.g., Desulfovibrio, Enterobacter) is increased [35,36].
Gut microbiota dysbiosis promotes the development of obesity and T2DM through multiple mechanisms. First, Firmicutes have a stronger ability to decompose indigestible carbohydrates and absorb energy, leading to increased energy harvest and storage in the host [37,38]. Second, reduced SCFA production impairs the regulation of gut barrier function, appetite and insulin sensitivity: SCFAs can strengthen the intestinal tight junctions to prevent the entry of lipopolysaccharide (LPS) and other endotoxins into the circulation, and also activate G protein-coupled receptors (GPR41, GPR43) to regulate glucose and lipid metabolism. Third, gut microbiota dysbiosis leads to increased intestinal permeability, and LPS enters the circulation to trigger metabolic endotoxemia, which activates the TLR4/NF-κB pathway and induces systemic chronic low-grade inflammation. Fourth, gut microbiota can regulate the secretion of gut hormones (e.g., glucagon-like peptide-1 (GLP-1), ghrelin) that regulate glucose and energy metabolism: reduced GLP-1 secretion and increased ghrelin secretion lead to impaired insulin secretion, increased appetite and weight gain [39-41].
Mitochondrial dysfunction
Mitochondria are the core organelles of cellular energy metabolism, and their dysfunction is an important cellular and molecular mechanism linking obesity and T2DM. In patients with obesity and T2DM, mitochondrial dysfunction is mainly manifested in reduced mitochondrial biogenesis, impaired oxidative phosphorylation, increased reactive oxygen species (ROS) production and abnormal mitochondrial dynamics (fission and fusion) [42,43], which is most obvious in adipose tissue, skeletal muscle and liver.
Mitochondrial dysfunction in skeletal muscle and liver directly leads to insulin resistance. Reduced mitochondrial biogenesis and impaired oxidative phosphorylation lead to decreased cellular energy production, and impaired fatty acid oxidation leads to intramyocellular and hepatic lipid accumulation, which in turn impairs insulin signal transduction. Increased ROS production by dysfunctional mitochondria causes oxidative stress, which inhibits the phosphorylation of IRS-1 and IR, further exacerbating insulin resistance [44,45]. Mitochondrial dysfunction in adipose tissue impairs lipid oxidation and energy expenditure, leading to increased fat accumulation and adipocyte hypertrophy, and also promotes the secretion of pro-inflammatory cytokines, aggravating chronic inflammation. In pancreatic β-cells, mitochondrial dysfunction impairs glucose-stimulated insulin secretion (GSIS), as mitochondria are the core organelles that sense glucose and produce ATP to trigger insulin exocytosis [46,47].
Genetic and epigenetic regulation
Genetic susceptibility and epigenetic regulation are the important intrinsic factors that determine the individual difference in the susceptibility to obesity and T2DM, and form the genetic basis of the bidirectional interplay between the two diseases [48]. Genome-wide association studies (GWAS) have identified more than 100 genetic loci associated with obesity and more than 400 genetic loci associated with T2DM, among which many loci are shared by both diseases, such as TCF7L2, FTO, PPARG and KCNJ11 [48], indicating the common genetic basis of obesity and T2DM.
The FTO gene is the first identified obesity susceptibility gene, and its variants are associated with increased BMI and risk of T2DM [49]. The FTO gene regulates energy balance and fat metabolism by affecting the expression of genes related to appetite regulation (e.g., MC4R) [49,50]. The TCF7L2 gene is the most significant T2DM susceptibility gene, and its variants are also associated with abnormal fat distribution and insulin resistance. The PPARG gene encodes the peroxisome proliferator-activated receptor γ, a key regulator of adipocyte differentiation and insulin sensitivity, and its variants lead to impaired adipocyte differentiation, reduced adiponectin secretion and increased risk of obesity and T2DM [50-52].
Epigenetic regulation, including DNA methylation, histone modification and non-coding RNA regulation, mediates the interaction between environmental factors (e.g., high-calorie diet, physical inactivity) and the genome, and plays an important role in the development of obesity and T2DM. High-calorie diet and physical inactivity can cause DNA methylation changes in genes related to glucose and lipid metabolism (e.g., PPARG, IRS-1) and inflammatory response, leading to persistent changes in gene expression and long-term impairment of metabolic function [53,54]. MicroRNAs (miRNAs), such as miR-146a, miR-21 and miR-126, are abnormally expressed in patients with obesity and T2DM, and they regulate insulin signal transduction, adipose tissue differentiation and inflammatory response by targeting key genes, participating in the pathogenesis of both diseases [55]. Epigenetic modifications can be passed on to offspring, leading to the intergenerational inheritance of metabolic disorders, which is an important reason for the increasing prevalence of obesity and T2DM in children and adolescents [56].
Clinical management of obesity-related T2DM
The core goal of clinical management for obesity-related T2DM is to break the vicious cycle between obesity and T2DM by reducing body weight, improving insulin resistance, protecting pancreatic β-cell function, and ultimately achieving good glycemic control and reducing the risk of complications [57,58]. The current clinical management strategies follow a stepwise approach, including lifestyle intervention (the foundation), pharmacotherapy (the mainstay) and bariatric surgery (for severe obesity), and the individualized comprehensive management plan should be formulated according to the patient’s BMI, glycemic status, age and comorbidities (Table 1).
Lifestyle intervention: The fundamental strategy
Lifestyle intervention is the first-line and fundamental treatment for all patients with obesity-related T2DM, and its core measures include medical nutrition therapy, increased physical activity and behavioral intervention. The goal of lifestyle intervention is to achieve a moderate and sustained weight loss (5-10% of initial body weight in the first 6 months), which can significantly improve insulin resistance, restore β-cell function and achieve good glycemic control, and even induce T2DM remission in some patients [59].
Medical nutrition therapy is the core of weight loss and glycemic control. The principle is to reduce total calorie intake (300-500 kcal/day for mild obesity, 500-800 kcal/day for moderate to severe obesity) on the basis of ensuring balanced nutrition, and adjust the dietary structure [59,60]. Priority is given to a low-calorie, high-fiber, low-glycemic index (LGI) diet, including increasing the intake of vegetables, fruits, whole grains, legumes and lean protein, and reducing the intake of refined carbohydrates, added sugars, saturated fats and trans fats. Intermittent fasting, as a novel dietary pattern (e.g., 16:8 fasting, 5:2 fasting), has been proven to effectively reduce body weight, improve insulin sensitivity and glycemic control in obese T2DM patients, and is a safe and effective supplementary nutrition therapy [61,62]. In addition, limiting alcohol intake and reducing salt intake are also important components of medical nutrition therapy, which can reduce the risk of cardiovascular complications.
Increased physical activity can increase energy expenditure, improve insulin sensitivity, reduce body fat (especially VAT) and protect β-cell function. The clinical guidelines recommend that obese T2DM patients should perform at least 150 minutes of moderate-intensity aerobic exercise per week (e.g., brisk walking, jogging, swimming, cycling) and 2-3 times of resistance training per week (e.g., weight lifting, yoga, Pilates) [62-64]. Aerobic exercise mainly reduces total body fat and improves insulin sensitivity in skeletal muscle and liver, while resistance training increases muscle mass, which is beneficial to long-term weight maintenance and metabolic improvement. Even a small amount of physical activity (e.g., 10 minutes of walking after meals) can effectively reduce postprandial blood glucose and improve insulin sensitivity [65]. For patients with poor mobility, low-intensity physical activity and daily living activities (e.g., housework, climbing stairs) are also recommended to avoid sedentary behavior [66].
Behavioral intervention is the guarantee for the long-term adherence of lifestyle intervention, including weight loss education, dietary and exercise behavior monitoring, psychological counseling and social support. Regular follow-up by medical staff can help patients monitor weight and glycemic changes, adjust intervention plans in a timely manner, and solve the problems encountered in the process of weight loss and glycemic control. Psychological counseling can help patients relieve anxiety, depression and other negative emotions caused by obesity and T2DM, and improve treatment compliance. Social support from family and friends is an important factor for patients to maintain long-term lifestyle changes [67-69].
Pharmacotherapy: Combined anti-obesity and anti-diabetic treatment
For obese T2DM patients who fail to achieve the weight loss and glycemic control goals through lifestyle intervention alone, pharmacotherapy should be initiated in a timely manner. The ideal drugs for obesity-related T2DM should have both anti-obesity and anti-diabetic effects, can improve insulin resistance, protect β-cell function, and have a good safety profile [70]. The currently used drugs are mainly divided into two categories: anti-diabetic drugs with weight loss effects and anti-obesity drugs with glycemic improvement effects, and combined drug therapy can be considered for patients with poor single drug efficacy.
Anti-diabetic drugs with weight loss effects: Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are the first-line pharmacotherapy for obesity-related T2DM, and are the most widely used drugs with both excellent anti-diabetic and anti-obesity effects. GLP-1 RAs mimic the action of endogenous GLP-1, which can enhance glucose-stimulated insulin secretion, inhibit glucagon secretion, slow gastric emptying, and reduce appetite and food intake by acting on the hypothalamus [71,72]. In addition, GLP-1 RAs can reduce VAT accumulation, improve adipose tissue function, and reduce chronic inflammation, thereby improving insulin resistance. Currently, long-acting GLP-1 RAs (e.g., semaglutide, liraglutide, dulaglutide) are the preferred drugs, with once-weekly or once-daily administration, good patient compliance, and significant weight loss (5-15% of initial body weight) and glycemic control effects. Semaglutide (2.4 mg weekly) has been approved for the treatment of obesity and T2DM, and is the most effective GLP-1 RA for weight loss [73-75].
Sodium-glucose cotransporter 2 inhibitors (SGLT2is) are another class of anti-diabetic drugs with weight loss effects, and are especially suitable for obese T2DM patients with cardiovascular and renal comorbidities. SGLT2is reduce blood glucose by inhibiting glucose reabsorption in the renal proximal convoluted tubule and increasing urinary glucose excretion, and the calorie loss caused by urinary glucose excretion leads to moderate weight loss (3% - 5% of initial body weight) [76,77]. SGLT2is can also reduce VAT accumulation, lower blood pressure, and have cardioprotective and renoprotective effects, which can reduce the risk of major adverse cardiovascular events (MACE) and diabetic kidney disease progression. Common SGLT2is include dapagliflozin, empagliflozin and canagliflozin, with good safety and tolerability [78,79].
Anti-obesity drugs with glycemic improvement effects: For obese T2DM patients with severe obesity (BMI ≥35 kg/m²) or moderate obesity (BMI ≥30 kg/m²) with poor glycemic control, anti-obesity drugs can be added on the basis of anti-diabetic drugs. Orlistat is a lipase inhibitor that reduces fat absorption by inhibiting pancreatic lipase, leading to moderate weight loss (3% - 7% of initial body weight), and can also improve glycemic control and reduce the risk of T2DM in obese individuals [80-82]. Its main adverse reactions are gastrointestinal symptoms (e.g., oily stool, flatulence), which can be alleviated by reducing fat intake. Phentermine-topiramate extended-release is a combined anti-obesity drug with central appetite suppression effects, which can achieve significant weight loss (8% - 10% of initial body weight) and improve glycemic control, but it is contraindicated in patients with hypertension, glaucoma and mental disorders [83].
Principles of combined pharmacotherapy: For obese T2DM patients with poor single drug efficacy, combined pharmacotherapy with complementary mechanisms can be considered, such as GLP-1 RAs combined with SGLT2is. This combination has a synergistic effect on weight loss and glycemic control: GLP-1 RAs mainly reduce food intake and improve β-cell function, while SGLT2is mainly increase calorie loss and reduce insulin resistance, and the combination can also produce additional cardioprotective and renoprotective effects [84,85]. When combined with drugs, attention should be paid to avoiding the combination of drugs that cause weight gain (e.g., sulfonylureas, insulin), and the dose should be adjusted according to the patient’s weight and glycemic status to avoid hypoglycemia [86].
Bariatric surgery: For severe obesity-related T2DM
Bariatric surgery is the most effective treatment for severe obesity-related T2DM, and is recommended for patients with T2DM and severe obesity (BMI ≥35 kg/m² for Asians) who fail to achieve the treatment goals through lifestyle intervention and pharmacotherapy, or moderate obesity (BMI ≥30 kg/m²) with serious comorbidities. Bariatric surgery not only achieves significant and sustained weight loss (20% - 30% of initial body weight) but also can induce long-term remission of T2DM in more than 50% of patients, and significantly reduce the risk of cardiovascular complications and all-cause mortality [87,88].
The commonly used bariatric surgery procedures for obesity-related T2DM include sleeve gastrectomy (SG), Roux-en-Y gastric bypass (RYGB) and one anastomosis gastric bypass (OAGB). SG is a restrictive surgery that reduces stomach volume by resecting most of the gastric fundus, leading to reduced food intake and appetite, and is the most commonly used procedure due to its simple operation, low complication rate and good weight loss and anti-diabetic effects [89,90]. RYGB is a combined restrictive and malabsorptive surgery that not only reduces stomach volume but also changes the digestive tract anatomy, leading to early satiety and reduced nutrient absorption, and its T2DM remission rate is higher than SG, but the operation is more complex and has a higher risk of nutritional deficiencies. OAGB is a modified RYGB with a simpler operation and lower complication rate, and its weight loss and anti-diabetic effects are similar to RYGB, which is increasingly used in clinical practice [91,92].
The anti-diabetic mechanism of bariatric surgery is not only due to weight loss but also includes the direct metabolic effects of intestinal rearrangement. Intestinal rearrangement leads to early contact between food and the distal small intestine, which increases the secretion of GLP-1 and other incretins, thereby enhancing insulin secretion, inhibiting glucagon secretion and improving insulin sensitivity [93,94]. In addition, bariatric surgery can improve adipose tissue function, reduce chronic inflammation, correct gut microbiota dysbiosis and improve mitochondrial function, which all contribute to the remission of T2DM. Long-term follow-up after surgery is crucial, including nutritional monitoring, supplement of vitamins and minerals, and lifestyle intervention, to maintain weight loss and T2DM remission, and reduce the risk of long-term complications [95].
Emerging research directions and future prospects
With the in-depth research on the pathogenic mechanisms of obesity and T2DM, novel therapeutic targets and strategies are continuously emerging, which bring new hopes for the precise prevention and treatment of obesity-related T2DM. The future research directions mainly focus on targeted therapy for core pathogenic mechanisms, gut microbiota modulation, precision medicine and early intervention, which are expected to break through the limitations of current clinical treatment and achieve more effective and personalized management of obesity and T2DM.
Targeted therapy for core pathogenic mechanisms
Targeted therapy for the core pathogenic mechanisms linking obesity and T2DM (e.g., chronic inflammation, adipose tissue dysfunction, mitochondrial dysfunction) is an important development direction of novel drugs. Anti-inflammatory targeted therapy is a research hotspot: monoclonal antibodies against pro-inflammatory cytokines (e.g., anti-TNF-α, anti-IL-6) have been proven to improve insulin resistance in clinical trials, and small molecule inhibitors of the NF-κB pathway and JAK/STAT pathway are also in preclinical and clinical research [96,97]. Targeted therapy for adipose tissue dysfunction: drugs that increase adiponectin secretion or activate adiponectin receptors, as well as leptin receptor agonists, are being developed to improve insulin sensitivity and regulate energy balance. Mitochondrial function modulators: drugs that promote mitochondrial biogenesis (e.g., PPARγ coactivator-1α (PGC-1α) agonists) and reduce ROS production (e.g., mitochondrial antioxidants) are expected to improve mitochondrial dysfunction in patients with obesity and T2DM [97-99].
Gut microbiota modulation: A novel therapeutic approach
Gut microbiota modulation has become a promising novel therapeutic approach for obesity and T2DM, with the advantages of non-invasiveness and good safety. Probiotics, prebiotics and fecal microbiota transplantation (FMT) are the main strategies for gut microbiota modulation. Probiotics (e.g., Bifidobacterium, Lactobacillus) can increase the abundance of SCFA-producing bacteria, reduce intestinal permeability and chronic inflammation, and improve insulin sensitivity and glycemic control [100,101]. Prebiotics (e.g., inulin, fructooligosaccharides) can promote the growth of beneficial bacteria in the gut and increase SCFA production [102]. FMT can reconstruct the gut microbiota of obese and T2DM patients by transplanting fecal microbiota from healthy individuals, and has been proven to effectively improve insulin resistance in clinical trials. In the future, personalized gut microbiota modulation based on the individual’s gut microbiota composition will become the development trend, and novel microbiota-based drugs (e.g., microbial metabolites, engineered probiotics) are also expected to be developed [102-104].
Precision medicine based on genetic and epigenetic characteristics
Precision medicine is the future development direction of all diseases, and obesity-related T2DM is no exception. With the development of high-throughput sequencing technology and multi-omics analysis, it is possible to formulate individualized prevention and treatment plans for obese and T2DM patients based on their genetic, epigenetic, gut microbiota and metabolic characteristics. For example, patients with different FTO gene variants can be given personalized dietary and exercise intervention plans [105,106]; patients with low adiponectin levels can be prioritized for GLP-1 RAs treatment; patients with specific gut microbiota dysbiosis can receive targeted microbiota modulation [106,107]. Epigenetic therapy is also a potential research direction: drugs that reverse abnormal DNA methylation and histone modification (e.g., DNA methyltransferase inhibitors) are expected to restore the expression of metabolic-related genes and improve metabolic function [108].
Early intervention and prevention of obesity and T2DM
Early intervention and prevention are the most cost-effective strategies to curb the global epidemic of obesity and T2DM. The focus of early intervention is on high-risk populations, including overweight/obese individuals, individuals with a family history of T2DM, prediabetic patients and children and adolescents with obesity [109,110]. For prediabetic patients, lifestyle intervention (weight loss, increased physical activity, healthy diet) can reduce the risk of developing T2DM by 58%, and drug intervention (e.g., metformin, GLP-1 RAs) can also reduce the risk by 30-40%. For children and adolescents with obesity, early lifestyle intervention and behavior correction are crucial to prevent the development of T2DM in adulthood, and family-based intervention is the most effective intervention mode [111,112]. In addition, public health interventions such as health education, food environment regulation (e.g., reducing the content of added sugars in processed foods) and promoting physical activity are important measures to prevent obesity and T2DM at the population level [113-115].
Conclusion
Obesity and T2DM are two closely linked global metabolic diseases with a complex bidirectional interplay: obesity is the primary driver of insulin resistance and pancreatic β-cell dysfunction, the core pathological features of T2DM; while T2DM-induced metabolic disorders further exacerbate adipose tissue dysfunction and abnormal fat metabolism, forming a vicious cycle that promotes the progression of both diseases. Chronic low-grade inflammation, adipose tissue dysfunction, gut microbiota dysbiosis, mitochondrial dysfunction and genetic epigenetic regulation are the key interrelated pathogenic mechanisms linking obesity and T2DM, forming a complex regulatory network.
The clinical management of obesity-related T2DM is a comprehensive project with lifestyle intervention as the foundation, pharmacotherapy as the mainstay and bariatric surgery as the important supplement for severe obesity. The core goal is to break the vicious cycle between obesity and T2DM through moderate and sustained weight loss, improve insulin resistance, protect pancreatic β-cell function, and achieve good glycemic control. GLP-1 RAs combined with SGLT2is is the most recommended pharmacotherapy regimen for obese T2DM patients, with synergistic effects on weight loss and glycemic control, as well as cardioprotective and renoprotective effects. Bariatric surgery can achieve significant and sustained weight loss and induce long-term T2DM remission in severe obesity patients, and is an effective treatment option for patients who fail to respond to conservative treatment.
In the future, with the in-depth research on the pathogenic mechanisms, novel therapeutic strategies such as targeted therapy for core mechanisms, gut microbiota modulation, precision medicine and early intervention will bring new breakthroughable
- Felix T, Katherine O, Irini G, Carrillo-Larco RM, Varghese JS, Marcus ME, et al. Global, regional, and national estimates of undiagnosed diabetes in adults: findings from the 2025 IDF Diabetes Atlas. Diabetes Care. 2026;49(3):490–496. Available from: https://doi.org/10.2337/dc25-2583
- GBD 2021 Adult BMI Collaborators. Global, regional, and national prevalence of adult overweight and obesity, 1990–2021, with forecasts to 2050: a forecasting study for the Global Burden of Disease Study 2021. Lancet. 2025;406(10505):785–812. Available from: https://pubmed.ncbi.nlm.nih.gov/40049186/
- Pouya S, Inga P, Paraskevi S, Malanda B, Karuranga S, Unwin N, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019;157:107843. Available from: https://doi.org/10.1016/j.diabres.2019.107843
- Salvatore A, Antonietta M, Antonietta M, Casillo M, Sapuppo W, Monda V, et al. Endocrine and metabolic mechanisms linking obesity to type 2 diabetes: implications for targeted therapy. Healthcare (Basel). 2025;13(12). Available from: https://doi.org/10.3390/healthcare13121437
- Abderrahim Z, Noureddine R, Nour El Houda B, Vanderpas J, Hassani B, Boutayeb A, et al. Epidemiology of hypertension and its relationship with type 2 diabetes and obesity in eastern Morocco. SpringerPlus. 2014;3:644. Available from: https://doi.org/10.1186/2193-1801-3-644
- Amélie P, Nadia B, Xavier C, Leterme D, Delattre J, Gauthier V, et al. Obesity and insulinopenic type 2 diabetes differentially impact bone phenotype, bone marrow adipose tissue, and serum levels of the cathelicidin-related antimicrobial peptide in mice. Bone. 2025;193:117387. Available from: https://doi.org/10.1016/j.bone.2024.117387
- Pablo H, Lucía C, Mònica B, Jordi S. Nuts and dried fruits: an update of their beneficial effects on type 2 diabetes. Nutrients. 2017;9(7). Available from: https://doi.org/10.3390/nu9070673
- Asimakidou E, Saipuljumri EN, Lo CH, Zeng J. Role of metabolic dysfunction and inflammation along the liver-brain axis in animal models with obesity-induced neurodegeneration. Neural Regen Res. 2025;20(4):1069–1076. Available from: https://doi.org/10.4103/nrr.nrr-d-23-01770
- Committee ADAPP. Classification and diagnosis of diabetes: standards of medical care in diabetes-2022. Diabetes Care. 2022;45(Suppl 1):S17–S38. Available from: https://doi.org/10.2337/dc22-s002
- Yulia AP, Anton SY, Yulia KK, Morgun AV, Lopatina OL, Malinovskaya NA, et al. Plasticity of adipose tissue-derived stem cells and regulation of angiogenesis. Front Physiol. 2018;9:1656. Available from: https://doi.org/10.3389/fphys.2018.01656
- Alan RS, Jeffrey EP. Insulin signaling pathways in time and space. Trends Cell Biol. 2002;12(2):65–71. Available from: https://doi.org/10.1016/s0962-8924(01)02207-3
- Yi Ning C, Ram Narayanan R, Vetriselvan S. Insulin resistance induced by obesity: mechanisms, metabolic implications and therapeutic approaches. Mol Biol Rep. 2026;53(1):357. Available from: https://doi.org/10.1007/s11033-026-11509-3
- Duygu Tozcu Y, Mehmet Ali G, Mustafa C, Hatice Dortok D, Akin T. Investigation of serum visfatin and chemerin levels in type 2 diabetes and obesity patients: their potential role as clinical and biomarkers. Biomedicines. 2025;13(11). Available from: https://doi.org/10.3390/biomedicines13112619
- Rahmouni K, Haynes WG. Leptin signaling pathways in the central nervous system: interactions between neuropeptide Y and melanocortins. Bioessays. 2001;23(12):1095–1099. Available from: https://doi.org/10.1002/bies.10018
- Park YJ, Warnock GL, Ao Z, Safikhan N, Meloche M, Asadi A, et al. Dual role of interleukin-1β in islet amyloid formation and its β-cell toxicity: implications for type 2 diabetes and islet transplantation. Diabetes Obes Metab. 2017;19(5):682–694. Available from: https://doi.org/10.1111/dom.12873
- Osonoi Y, Nakanishi H, Nakamura M. Role of inflammation in progression of chronic kidney disease in type 2 diabetes mellitus: clinical implications. Semin Nephrol. 2023;43(3):151431. Available from: https://doi.org/10.1016/j.semnephrol.2023.151431
- Cipriano R, Fiorentino L, Miele MG, Borriello R, Tota A, Pallozzi M, et al. The crosstalk between gut microbiota, intestinal immunological niche and visceral adipose tissue as a new model for the pathogenesis of metabolic and inflammatory diseases: the paradigm of type 2 diabetes mellitus. Curr Med Chem. 2022;29(18):3189–3201. Available from: https://doi.org/10.2174/0929867329666220105121124
- Cree MG, Newcomer BR, Baumgartner AD, Pyle L, Reusch JE, Nadeau KJ, et al. Insulin resistance in type 2 diabetes youth relates to serum free fatty acids and muscle mitochondrial dysfunction. J Diabetes Complications. 2017;31(1):141–148. Available from: https://doi.org/10.1016/j.jdiacomp.2016.10.014
- Miras AD, le Roux CW. Effects of various gastrointestinal procedures on β-cell function in obesity and type 2 diabetes. Surg Obes Relat Dis. 2016;12(6):1213–1219. Available from: https://doi.org/10.1016/j.soard.2016.02.035
- Perdoncin M, Konrad A, Wyner JR, Lohana S, Pillai SS, Pereira DG, et al. A review of miRNAs as biomarkers and effect of dietary modulation in obesity associated cognitive decline and neurodegenerative disorders. Front Mol Neurosci. 2021;14:756499. Available from: https://doi.org/10.3389/fnmol.2021.756499
- Batista TM, Jayavelu AK, Albrechtsen NJW, Iovino S, Lebastchi J, Pan H, et al. A cell-autonomous signature of dysregulated protein phosphorylation underlies muscle insulin resistance in type 2 diabetes. Cell Metab. 2020;32(5):844–859.e5. Available from: https://doi.org/10.1016/j.cmet.2020.08.007
- Catalano F, Cammarata C, Sardo C, Lippolis P, Arnone S, Sbardella ME. Obesity, insulin resistance, and colorectal cancer: could miRNA dysregulation play a role. Int J Mol Sci. 2019;20(12). Available from: https://doi.org/10.3390/ijms20122922
- Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112(12):1796–1808. Available from: https://doi.org/10.1172/jci19246
- Cárdenas ML, Hernández R. Possible gender influence in the mechanisms underlying the oxidative stress, inflammatory response, and the metabolic alterations in patients with obesity and/or type 2 diabetes. Antioxidants (Basel). 2021;10(11). Available from: https://doi.org/10.3390/antiox10111729
- Kootte RS, Govaert HM, van der Bilt JD, Annema W, Romijn JA. Fetuin B is related to cytokine/chemokine and insulin signaling in adipose tissue and plasma in humans. J Clin Endocrinol Metab. 2025;110(10):e3235–e3244. Available from: https://doi.org/10.1210/clinem/dgaf073
- Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science. 1993;259(5091):87–91. Available from: https://doi.org/10.1126/science.7678183
- Santos GJ, Oliveira CAM, Branco AC, Reis LF. CNTF protects MIN6 cells against apoptosis induced by alloxan and IL-1β through downregulation of the AMPK pathway. Cell Signal. 2011;23(10):1669–1676. Available from: https://doi.org/10.1016/j.cellsig.2011.06.001
- Pradhan AD, Manson JE, Rifai N, Buring JE, Ridker PM. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA. 2001;286(3):327–334. Available from: https://doi.org/10.1001/jama.286.3.327
- Wernstedt U. Adipose tissue expansion in obesity, health, and disease. Front Cell Dev Biol. 2023;11:1188844. Available from: https://doi.org/10.3389/fcell.2023.1188844
- Pasarica V, Cassano S, Veronesi A. Adipose tissue plasticity: how fat depots respond differently to pathophysiological cues. Diabetologia. 2016;59(6):1075–1088. Available from: https://doi.org/10.1007/s00125-016-3933-4
- Kadowaki T, Yamauchi T, Kubota N, Hara K, Ueki K, Tobe K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J Clin Invest. 2006;116(7):1784–1792. Available from: https://doi.org/10.1172/jci29126
- Nawrocki AR, Scherer PE. The delicate balance between fat and muscle: adipokines in metabolic disease and musculoskeletal inflammation. Curr Opin Pharmacol. 2004;4(3):281–289. Available from: https://doi.org/10.1016/j.coph.2004.03.003
- Blaak EE. Fatty acid metabolism in obesity and type 2 diabetes mellitus. Proc Nutr Soc. 2003;62(3):753–760. Available from: https://doi.org/10.1079/pns2003290
- Roussel C, Pelgrim S, Stegen G, Moonen-Kornips E, Meex RC, Phielix E, et al. Relationship of C5L2 receptor to skeletal muscle substrate utilization. PLoS One. 2013;8(2):e57494. Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0057494
- Aydin Ö, Nier A, Gille V. The gut microbiome as a target for the treatment of type 2 diabetes. Curr Diabetes Rep. 2018;18(8):55. Available from: https://doi.org/10.1007/s11892-018-1020-6
- Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444(7122):1022–1023. Available from: https://doi.org/10.1038/4441022a
- Qin J, Li Y, Cai Z, Li S, Zhu J, Zhang F, et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature. 2012;490(7418):55–60. Available from: https://doi.org/10.1038/nature11450
- Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122):1027–1031. Available from: https://doi.org/10.1038/nature05414
- Yokota T, Shimizu T. Core and periphery concentrations of short-chain fatty acids in luminal contents of the rat colon. Comp Biochem Physiol A Comp Physiol. 1992;103(2):353–355. Available from: https://doi.org/10.1016/0300-9629(92)90593-f
- Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, et al. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes. 2008;57(6):1470–1481. Available from: https://doi.org/10.2337/db07-1403
- Everton C, José Carlos P, Elinton Adami C, Cláudio Saddy Rodrigues C, Daniéla Oliveira M. Glucagon-like peptides 1 and 2 are involved in satiety modulation after modified biliopancreatic diversion: results of a pilot study. Obes Surg. 2018;28(2):506–512. Available from: https://doi.org/10.1007/s11695-017-2875-3
- Baklanov A, Nasonova L, Krylov T, Sokolov V, Makarova A, Orlov A. Mitochondrial dysfunction as a factor of energy metabolism disorders in type 2 diabetes mellitus. Front Biosci (Scholar Ed). 2024;16(1):5–14. Available from: https://doi.org/10.31083/j.fbs1601005
- Rebecca P, Kitt Falk P. Mitochondrial dysfunction and type 2 diabetes. Curr Diabetes Rep. 2005;5(3):177–183. Available from: https://doi.org/10.1007/s11892-005-0006-3
- Prasad PD, Miller SM, Hamilton TM. Skeletal muscle nucleo-mitochondrial crosstalk in obesity and type 2 diabetes. Int J Mol Sci. 2017;18(4):831. Available from: https://doi.org/10.3390/ijms18040831
- Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54(6):1615–1625. Available from: https://doi.org/10.2337/diabetes.54.6.1615
- Qu ZY, Zhang M, Dong YM, Wang YM, Zhu GZ, Zhao YP, et al. Metformin prevents LYRM1-induced insulin resistance in 3T3-L1 adipocytes via a mitochondrial-dependent mechanism. Exp Biol Med (Maywood). 2014;239(12):1567–1574. Available from: https://doi.org/10.1177/1535370214537746
- Wang C, Li X, Mu K, Li L, Wang S, Zhu Y, et al. Deficiency of APPL1 in mice impairs glucose-stimulated insulin secretion through inhibition of pancreatic beta cell mitochondrial function. Diabetologia. 2013;56(9):1999–2009. Available from: https://doi.org/10.1007/s00125-013-2971-4
- Kukorowski T, Davis AW, Leone CL. Insights into the genetic susceptibility to type 2 diabetes from genome-wide association studies of obesity-related traits. Curr Diabetes Rep. 2015;15(10):83. Available from: https://doi.org/10.1007/s11892-015-0648-8
- Rees SD, Islam M, Hydrie MZ, Chaudhary B, Bellary S, Hashmi S, et al. An FTO variant is associated with type 2 diabetes in South Asian populations after accounting for body mass index and waist circumference. Diabet Med. 2011;28(6):673–680. Available from: https://doi.org/10.1111/j.1464-5491.2011.03257.x
- Jiao XC, Yang YL, Chen YC, Xia Z, Zhang W, Feng Y, et al. Multivariate analysis of genome-wide data to identify potential pleiotropic genes for type 2 diabetes, obesity and coronary artery disease using MetaCCA. Int J Cardiol. 2019;283:144–150. Available from: https://doi.org/10.1016/j.ijcard.2018.10.102
- Xu ZY, Jiang X, Xu XD. Emerging role and mechanism of the FTO gene in cardiovascular diseases. Biomolecules. 2023;13(5):805. Available from: https://doi.org/10.3390/biom13050850
- Pearson ER. Translating TCF7L2: from gene to function. Diabetologia. 2009;52(7):1227–1230. Available from: https://doi.org/10.1007/s00125-009-1356-1
- Evans DR, Scarpelli BM. PPARγ: a nuclear regulator of metabolism, differentiation, and cell growth. J Biol Chem. 2001;276(41):37731–37734. Available from: https://doi.org/10.1074/jbc.r100034200
- Waterland RA, Jirtle RL. Early nutrition, epigenetic changes at transposons and imprinted genes, and enhanced susceptibility to adult chronic diseases. Nutrition. 2004;20(1):63–68. Available from: https://doi.org/10.1016/j.nut.2003.09.011
- Byung Chul J, Sona K. Epigenetic regulation of inflammatory factors in adipose tissue. Biochim Biophys Acta Mol Cell Biol Lipids. 2021;1866(11):159019. Available from: https://doi.org/10.1016/j.bbalip.2021.159019
- Cleber IC A, Vasques NK C, Silveira PC, Pereira SB M, Garcia MS, de Freitas BS. Exercise training-induced changes in microRNAs: beneficial regulatory effects in hypertension, type 2 diabetes, and obesity. Int J Mol Sci. 2018;19(11):3608. Available from: https://doi.org/10.3390/ijms19113608
- Poston L. Intergenerational transmission of insulin resistance and type 2 diabetes. Prog Biophys Mol Biol. 2011;106(1):315–322. Available from: https://doi.org/10.1016/j.pbiomolbio.2010.11.011
- Scott D. Translating clinical guidelines into clinical practice: role of the pharmacist in type 2 diabetes management. J Am Pharm Assoc. 2009;49(6):e152–e162. Available from: https://doi.org/10.1331/japha.2009.08181
- Look AHEAD Research Group, Wadden RR. Long-term effects of a lifestyle intervention on weight and cardiovascular risk factors in individuals with type 2 diabetes mellitus: four-year results of the Look AHEAD trial. Arch Intern Med. 2010;170(17):1566–1575. Available from: https://doi.org/10.1001/archinternmed.2010.334
- Cassi NU, Jacqueline NS, Robyn EC, Hartman AS, Olson MR, Anderson NL, et al. Consuming mushrooms when adopting a healthy Mediterranean-style dietary pattern does not influence short-term changes of most cardiometabolic disease risk factors in healthy middle-aged and older adults. J Nutr. 2024;154(2):574–582. Available from: https://doi.org/10.1016/j.tjnut.2023.12.026
- Wang MJ, Hsu CH, Yu SB, Chen GS, Huang HJ. Impact of the Mediterranean diet on glycemic control, body mass index, lipid profile, and blood pressure in type 2 diabetes: a meta-analysis of randomized controlled trials. Nutrients. 2025;17(24):4308. Available from: https://doi.org/10.3390/nu17243908
- Sarah P, Stephanie D, Wendy P, Malone A, Ireton-Jones C, Haney A, et al. Enteral and parenteral order writing survey—a collaborative evaluation between the Academy of Nutrition and Dietetics’ Dietitians in Nutrition Support Dietetics Practice Group and the American Society for Parenteral and Enteral Nutrition (ASPEN) Dietetics Practice Section. Nutr Clin Pract. 2020;35(3):377–385. Available from: https://doi.org/10.1016/j.jand.2020.01.015
- Lim SS, Vos T, Flaxman AD, Danaei G, Shibuya K, Adair-Rohani H, et al. A comparative risk assessment of burden of disease and injury attributable to 67 risk factors and risk factor clusters in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2224–2260. Available from: https://doi.org/10.1016/s0140-6736(12)61766-8
- Powell KE, King AC, Buchner DM, Campbell WW, DiPietro L, Erickson KI, et al. The scientific foundation for the Physical Activity Guidelines for Americans, 2nd edition. J Phys Act Health. 2019;16(1):1–11. Available from: https://doi.org/10.1123/jpah.2018-0618
- Eliza GM, Parneet S, Caryl AN, David WD, Robin MD. Effects of progressive resistance training and weight loss versus weight loss alone on inflammatory and endothelial biomarkers in older adults with type 2 diabetes. Eur J Appl Physiol. 2017;117(8):1669–1678. Available from: https://doi.org/10.1007/s00421-017-3657-2
- MHM, AMN, AEH. Different patterns of brisk walking are equally effective in decreasing postprandial lipaemia. Int J Obes Relat Metab Disord. 2000;24(10):1303–1309. Available from: https://doi.org/10.1038/sj.ijo.0801399
- Daniela GL, Luz MTG, Néstor SI, Sergio RVB, Astrid vOG. Effects of break in sedentary behaviour on blood glucose control in diabetic patients: systematic review. Endocrinol Diabetes Nutr. 2022;69(10):888–896. Available from: https://doi.org/10.1016/j.endien.2022.11.027
- Wong D. Motivational interviewing in health care: helping patients change behavior. Drug Alcohol Rev. 2009;28(3):337–338. Available from: https://doi.org/10.1111/j.1465-3362.2009.00073_7.x
- Prakasini S, Pandey P, Abhay MG, Vadia N, Menon SV, Chennakesavulu K, et al. Prevalence and impact of microvascular complications in type 2 diabetes mellitus on cognitive impairment and depression: a systematic review and meta-analysis. Diabetol Metab Syndr. 2025;17(1):187. Available from: https://doi.org/10.1186/s13098-025-01759-9
- Wing RR, Jeffery RW. Benefits of recruiting participants with friends and increasing social support for weight loss and maintenance. J Consult Clin Psychol. 1999;67(1):132–138. Available from: https://doi.org/10.1037//0022-006x.67.1.132
- Emir MM, Špela V, Zibar TK, Janež A, Mikhailidis DP, Rizzo M, et al. Glucagon-like peptide-1 receptor agonists and dual glucose-dependent insulinotropic polypeptide/glucagon-like peptide-1 receptor agonists in the treatment of obesity/metabolic syndrome, prediabetes/diabetes and non-alcoholic fatty liver disease—current evidence. J Cardiovasc Pharmacol Ther. 2022;27:10742484221146371. Available from: https://doi.org/10.1177/10742484221146371
- Rui W, Bo X, Yuting H, Zhou Z, Sun B, Yu L, et al. Effect of semaglutide on arrhythmic, major cardiovascular, and microvascular outcomes in patients with type 2 diabetes: a systematic review and meta-analysis. Front Endocrinol. 2025;16:1554795. Available from: https://doi.org/10.3389/fendo.2025.1554795
- Carlos C, Jeff U. Primary care management of type 2 diabetes: a comparison of the efficacy and safety of glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Postgrad Med. 2021;133(8):843–853. Available from: https://doi.org/10.1080/00325481.2021.1971461
- Huize G, Qian W, Anqi Z, Yu K, Song D, Li J, et al. Evaluation of three mechanisms of action (SGLT2 inhibitors, GLP-1 receptor agonists, and sulfonylureas) in treating type 2 diabetes with heart failure: a systematic review and network meta-analysis of RCTs. Front Endocrinol. 2025;16:1562815. Available from: https://doi.org/10.3389/fendo.2025.1562815
- Silvia PA, Sergio V, Viyey KD. GLP-1 agonists in glycemic and weight control of type 2 diabetes. New perspectives. Med Clin (Barc). 2025;165(3):107042. Available from: https://doi.org/10.1016/j.medcli.2025.107042
- Ping Z, Hai Z, Miaochun H, Wenbin F, Zhixia C. Efficacy and safety of once-weekly semaglutide in adults with overweight or obesity: a meta-analysis. Endocrine. 2022;75(3):718–724. Available from: https://doi.org/10.1007/s12020-021-02945-1
- Gurdeep S, Matthew K, Meghan B. Wegovy (semaglutide): a new weight loss drug for chronic weight management. J Investig Med. 2022;70(1):5–13. Available from: https://doi.org/10.1136/jim-2021-001952
- Robert G. Canagliflozin and cardiovascular and renal events in type 2 diabetes. Postgrad Med. 2018;130(2):149–153. Available from: https://doi.org/10.1056/nejmoa1611925
- Mansi T, Tamara YM, Sophie LS, Richard OD, Jerry RG. Sodium-glucose co-transporter 2 inhibitor therapy: use in chronic kidney disease and adjunctive sodium restriction. Intern Med J. 2022;52(10):1666–1676. Available from: https://doi.org/10.1111/imj.15727
- Bernard Z, Christoph W, John ML, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117–2128. Available from: https://doi.org/10.1056/nejmoa1504720
- Anna N, Johan B, Thomas N, Marcus T, David N, Jan WE. Dapagliflozin and cardiovascular mortality and disease outcomes in a population with type 2 diabetes similar to that of the DECLARE-TIMI 58 trial: a nationwide observational study. Diabetes Obes Metab. 2019;21(5):1136–1145. Available from: https://doi.org/10.1111/dom.13627
- Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, Donato KA, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. J Am Coll Cardiol. 2014;63(25 Pt B):2985–3023. Available from: https://doi.org/10.1161/01.cir.0000437739.71477.ee
- Heck AM, Yanovski JA, Calis KA. Orlistat, a new lipase inhibitor for the management of obesity. Pharmacotherapy. 2000;20(3):270–279. Available from: https://doi.org/10.1592/phco.20.4.270.34882
- Sjostrom L, Rissanen A, Andersen T, Boldrin M, Golay A, Koppeschaar HP, et al. Randomised placebo-controlled trial of orlistat for weight loss and prevention of weight regain in obese patients. Lancet. 1998;352(9123):167–172. Available from: https://doi.org/10.1016/s0140-6736(97)11509-4
- Gadde WT, Halpern RD, Lachter M, Gadde KM, Allison DB, Peterson CA, et al. Two-year sustained weight loss and metabolic benefits with controlled-release phentermine/topiramate in obese and overweight adults (SEQUEL): a randomized, placebo-controlled, phase 3 extension study. Am J Clin Nutr. 2012;95(2):297–308. Available from: https://doi.org/10.3945/ajcn.111.024927
- Yaping W, Zhenxing Y, Qingqing C. Efficacy and safety of GLP-1 receptor agonists combined with SGLT-2 inhibitors in elderly patients with type 2 diabetes: a meta-analysis. Am J Transl Res. 2024;16(11):6852–6866. Available from: https://doi.org/10.62347/mcee4840
- Bertram P, Gabriel S, Lawrence AL, Deepak LB. The role of combined SGLT1/SGLT2 inhibition in reducing the incidence of stroke and myocardial infarction in patients with type 2 diabetes mellitus. Cardiovasc Drugs Ther. 2022;36(3):561–567. Available from: https://doi.org/10.1007/s10557-021-07291-y
- Committee ADAPP. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2022. Diabetes Care. 2022;45(Suppl 1):S125–S143. Available from: https://doi.org/10.2337/dc22-s009
- Michalsky M, Reichard K, Inge T, Pratt J, Lenders C. ASMBS pediatric committee best practice guidelines. Surg Obes Relat Dis. 2012;8(1):1–7. Available from: https://doi.org/10.1016/j.soard.2011.09.009
- Philip RS, Deepak LB, John PK, Wolski K, Brethauer SA, Navaneethan SD, et al. Bariatric surgery versus intensive medical therapy for diabetes—3-year outcomes. N Engl J Med. 2014;370(21):2002–2013. Available from: https://doi.org/10.1056/nejmoa1401329
- Buchwald H, Estok R, Fahrbach K, Banel D, Jensen MD, Pories WJ, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122(3):248–256. Available from: https://doi.org/10.1016/j.amjmed.2008.09.041
- Emma O, Rossita MY, Shahjahan K, Breda M, Muhammed AM. Weight loss outcomes in laparoscopic vertical sleeve gastrectomy (LVSG) versus laparoscopic Roux-en-Y gastric bypass (LRYGB) procedures: a meta-analysis and systematic review of randomized controlled trials. Surg Laparosc Endosc Percutan Tech. 2017;27(1):8–18. Available from: https://doi.org/10.1097/sle.0000000000000374
- Kwon Y, Lee S, Kim D, ALRomi A, Park SH, Lee CM, et al. Biliopancreatic limb length as a potential key factor in superior glycemic outcomes after Roux-en-Y gastric bypass in patients with type 2 diabetes: a meta-analysis. Diabetes Care. 2022;45(12):3091–3100. Available from: https://doi.org/10.2337/dc22-0835
- Reem AG, Matthieu B, Karen B, M’harzi L, Zinzindohoue F, Douard R, et al. Is mini-gastric bypass a rational approach for type 2 diabetes. Curr Atheroscler Rep. 2017;19(12):51. Available from: https://doi.org/10.1007/s11883-017-0689-3
- Francesco R, Antonello F, David EC, Vix M, Gnuli D, Mingrone G, et al. The mechanism of diabetes control after gastrointestinal bypass surgery reveals a role of the proximal small intestine in the pathophysiology of type 2 diabetes. Ann Surg. 2006;244(5):741–749. Available from: https://doi.org/10.1097/01.sla.0000224726.61448.1b
- Drab SR. Incretin-based therapies for type 2 diabetes mellitus: current status and future prospects. Pharmacotherapy. 2010;30(6 Pt 2):609–624. Available from: https://doi.org/10.1592/phco.30.6.609
- Maurizio DL, Monica Z, Giulia B, Chiappetta S, Iossa A, Merola G, et al. Metabolic bariatric surgery as a therapeutic option for patients with type 2 diabetes: a meta-analysis and network meta-analysis of randomized controlled trials. Diabetes Obes Metab. 2023;25(8):2362–2373. Available from: https://doi.org/10.1111/dom.15117
- Kim J-H, Wolfe B. Bariatric/metabolic surgery: short- and long-term safety. Curr Atheroscler Rep. 2012;14(6):597–605. Available from: https://doi.org/10.1007/s11883-012-0287-3
- Ljiljana L, Nebojsa ML, Natasa R, Jotic A, Lalic K, Milicic T, et al. Hypertension in obese type 2 diabetes patients is associated with increases in insulin resistance and IL-6 cytokine levels: potential targets for an efficient preventive intervention. Int J Environ Res Public Health. 2014;11(4):3586–3598. Available from: https://doi.org/10.3390/ijerph110403586
- Carey NL, Alan RS. Inflammatory links between obesity and metabolic disease. J Clin Invest. 2011;121(6):2111–2117. Available from: https://doi.org/10.1172/jci57132
- Yamauchi T, Kamon J, Waki H, Terauchi Y, Kubota N, Hara K, et al. The fat-derived hormone adiponectin reverses insulin resistance associated with both lipoatrophy and obesity. Nat Med. 2001;7(8):941–946. Available from: https://doi.org/10.1038/90984
- Bin W, Yakun W, Shoukui X, Yan J, Rong C, Nan H. Alterations of gut microbiome in patients with type 2 diabetes mellitus who had undergone cholecystectomy. Am J Physiol Endocrinol Metab. 2021;320(1):E113–E121. Available from: https://doi.org/10.1152/ajpendo.00471.2020
- Camacho A, Huang JK, Delint-Ramirez I, Yew Tan C, Fuller M, Lelliott CJ, et al. Peroxisome proliferator-activated receptor gamma-coactivator-1 alpha coordinates sphingolipid metabolism, lipid raft composition and myelin protein synthesis. Eur J Neurosci. 2013;38(5/6):2672–2683. Available from: https://doi.org/10.1111/ejn.12281
- Paige VB, Frank AD. Targeting the gastrointestinal tract to treat type 2 diabetes. J Endocrinol. 2016;230(3):R95–R113. Available from: https://doi.org/10.1530/joe-16-0056
- Frank B, Hanna S, Stefan S, Stefan V, Masako T, Stephan S. The dietary fiber pectin: health benefits and potential for the treatment of allergies by modulation of gut microbiota. Curr Allergy Asthma Rep. 2021;21(10):43. Available from: https://doi.org/10.1007/s11882-021-01020-z
- Anne V, Els VN, Frits H, Salojärvi J, Kootte RS, Bartelsman JF, et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143(4):913–916.e7. Available from: https://doi.org/10.1053/j.gastro.2012.06.031
- Sheng-Yuan H, Cheng-Yang L, Hsiu-Chu C, Liang-Ti H, Chung-Ming C. Maternal aspartame exposure induces neonatal pulmonary metabolic dysregulation and redox imbalance: a multiomics investigation of gut microbiota–host interactions. J Agric Food Chem. 2025;73(42):27012–27024. Available from: https://doi.org/10.1021/acs.jafc.5c08819
- Francis SC, Harold V. A new initiative on precision medicine. N Engl J Med. 2015;372(9):793–795. Available from: https://doi.org/10.1056/nejmp1500523
- LY, YL, ZQ, Kumar S, Curran JE, Blangero J, et al. Molecular profiling of human induced pluripotent stem cell-derived hypothalamic neurones provides developmental insights into genetic loci for body weight regulation. J Neuroendocrinol. 2017;29(2). Available from: https://doi.org/10.1111/jne.12455
- Alexios SA, Regent L, Marios M, Charalambos A. Adiponectin as a regulator of vascular redox state: therapeutic implications. Recent Pat Cardiovasc Drug Discov. 2011;6(2):78–88. Available from: https://doi.org/10.2174/157489011795933837
- Ge Y, Jinlong W, Pinyi L, Zhang Q, Tian Y, Hou G, et al. Role of the gut microbiota in type 2 diabetes and related diseases. Metabolism. 2021;117:154712. Available from: https://doi.org/10.1016/j.metabol.2021.154712
- Robert F, Mario FF. Epigenetics and the environment: emerging patterns and implications. Nat Rev Genet. 2012;13(2):97–109. Available from: https://doi.org/10.1038/nrg3142
- Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393–403. Available from: https://doi.org/10.1056/nejmoa012512
- Chiara G, Alice M, Claudio M. Pros and cons of current diagnostic tools for risk-based screening of prediabetes and type 2 diabetes in children and adolescents with overweight or obesity. Horm Res Paediatr. 2023;96(4):356–365. Available from: https://doi.org/10.1159/000528342
- Lindstrom J, Ilanne-Parikka P, Peltonen M, Aunola S, Eriksson JG, Hemiö K, et al. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: follow-up of the Finnish Diabetes Prevention Study. Lancet. 2006;368(9548):1673–1679. Available from: https://doi.org/10.1016/s0140-6736(06)69701-8
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
