Journal of Clinical Research and Ophthalmology
Regenerating Retinal Cells and Restoring Vision
1Riggs Pharmaceuticals, Department of Pharmacy, University of Karachi, Pakistan
2Assistant Professor, Department of Pathology, Dow University of Health Sciences Karachi, Pakistan
Author and article information
Cite this as
Haider R, et al. Regenerating Retinal Cells and Restoring Vision. J Clin Res Ophthalmol. 2026; 13(1): 6-9. Available from: 10.17352/2455-1414.000112
Copyright License
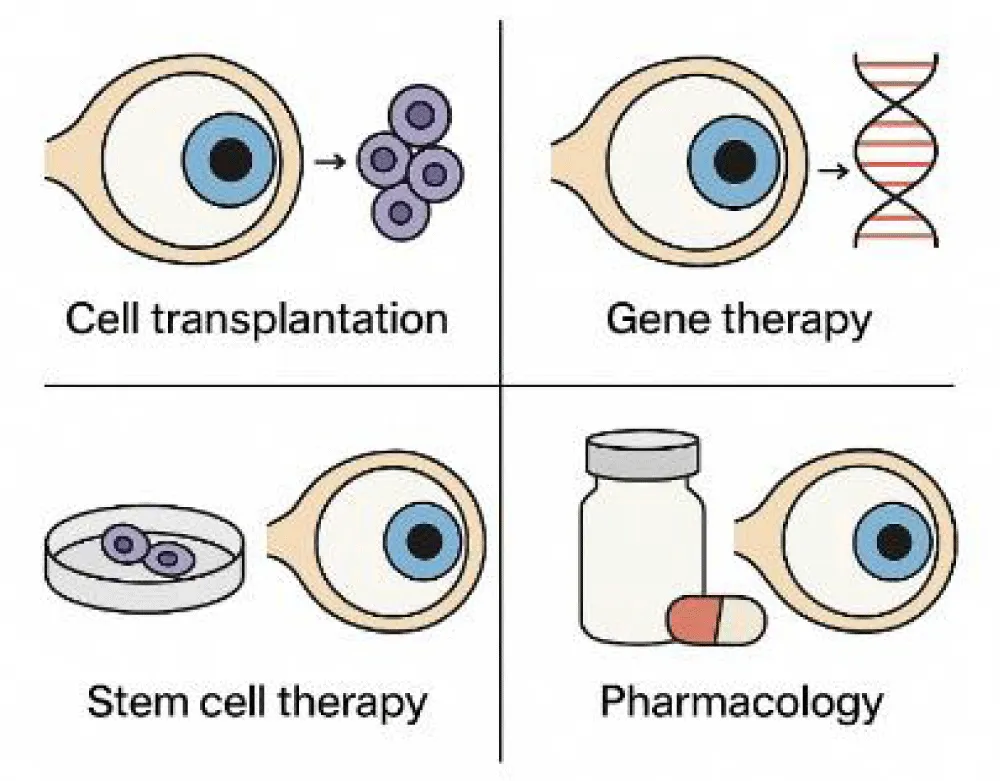
© 2026 Haider R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.The retinal degeneration condition leads to permanent vision loss, which affects millions of people worldwide who suffer from age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, and other inherited retinal disorders. The medical field once believed that damage to photoreceptors and retinal pigment epithelium cells would result in permanent loss because neural retinal tissue cannot repair itself. The medical field has undergone a significant shift in this context due to advances in regenerative medicine. The medical field uses stem cell technologies to create patient-specific retinal cells from Induced Pluripotent Stem Cells (iPSCs). The practice of gene therapy, utilizing viral vector-based gene replacement and CRISPR-based genome editing, has yielded successful results that enhance visual capabilities for certain inherited retinal conditions. The use of retinal tissue engineering methods, combined with biomaterial scaffolds and three-dimensional retinal organoids, enables scientists to create structures that support cell survival as cells adapt to new environments.
The review compiles evidence from clinical and preclinical studies published after 2015 that investigate retinal regeneration methods. Results indicate that iPSC-derived RPE transplantation leads to retinal structural restoration, gene therapy enables partial vision recovery for specific genetic mutations, and engineered scaffolds strengthen photoreceptor cells, aiding synaptic connections. While these approaches show promise, challenges remain: developing treatments compatible with the immune system, ensuring long-term efficacy, addressing safety in gene editing, and achieving scalable production.
Current evidence indicates that retinal regeneration research has progressed to early clinical application, offering real possibilities for restoring vision in patients with previously untreatable retinal disorders.
Retinal degenerative diseases represent a significant global public health burden and are among the primary causes of blindness [1,2]. The retinal tissue contains specialized neurosensory components, including photoreceptors, bipolar cells, ganglion cells, and RPE cells, which work together to process light into neural signals for transmission to the brain [3,4]. The visual system experiences progressive and permanent vision loss when photoreceptors and RPE cells sustain damage [5,6].
Current treatment methods aim to prevent disease progression rather than to rebuild damaged retinal cells [2,7]. Anti-vascular endothelial growth factor therapy, together with laser photocoagulation and supportive rehabilitation techniques, enables patients to keep their existing vision but fails to restore their lost photoreceptor function [7].
Recent advances in regenerative medicine have introduced stem cell therapy, gene replacement strategies, and biomaterial-based tissue engineering methods, which scientists now use to achieve both structural restoration and functional recovery [8-10]. These developments provide effective treatment options for medical conditions that doctors previously believed to be impossible to treat.
Literature review
Stem cell–based therapies
The invention of iPSCs enables scientists to create retinal pigment epithelium and photoreceptor cells, which function as transplantable tissues for medical use. As a key development, initial clinical tests have shown that transplanted RPE cells can survive and partially integrate into the surrounding tissue in the subretinal space. Transitioning from these cell therapies, retinal organoids can reproduce retinal development using pluripotent stem cells as a source, and they may be used as transplantable tissue constructs.
Gene therapy
Adeno-associated viral (AAV) vector–mediated gene therapy has shown success in treating inherited retinal dystrophies. Clinical trials have reported improvements in light sensitivity and visual acuity in selected patients. CRISPR-based gene editing introduces precise genomic correction, which expands therapeutic possibilities for monogenic retinal diseases.
Tissue engineering
Biomaterial scaffolds improve cellular survival rates while helping cells maintain their proper orientation, together with their synaptic connections, which occur after transplantation. Three-dimensional engineered retinal tissues enhance the alignment of photoreceptors, and they enable photoreceptors to make proper contact with host neural circuits.
Neuroprotection and regenerative signaling
Neurotrophic factors have demonstrated protective effects against photoreceptor degeneration in experimental models [11,12]. These approaches may complement cell-based regenerative therapies.
Research methodology
The study performed a systematic, structured review that used three databases: PubMed, Scopus, and Web of Science. The researchers examined studies that were published between 2015 and 2025. The researchers included studies that involved human clinical trials or validated animal studies that tested stem cell therapy, gene therapy, and tissue engineering methods for retinal regeneration [8-10,13-18].
The researchers examined 312 records and found 38 studies that matched the inclusion requirements for qualitative analysis.
Statistical analysis
The team gathered quantitative data from eligible clinical trials, which were selected for their specific requirements. The study measured best-corrected visual acuity (BCVA) changes through ETDRS chart assessment when data became accessible to researchers [9,19]. Researchers established statistical significance at a p < 0.05 threshold. The research team decided against conducting a meta-analysis because of the study design discrepancies and different outcome assessment methods used in the various studies.
Results
The clinical research on iPSC-derived RPE transplantation showed both structural stabilization and specific areas of functional enhancement [14,20]. The gene therapy studies showed that patients with inherited retinal disorders achieved statistically significant improvements in both visual sensitivity and acuity measurements [9,19]. The preclinical research on tissue engineering demonstrated that biodegradable scaffolds enhanced both photoreceptor survival and synaptic integration capabilities [10,21] (Table 1, Figure 1).
Discussion
The development of new treatment approaches in ophthalmology has resulted from the combined research efforts in stem cell biology, gene editing technology, and biomaterials science [8,10,15]. Researchers are currently studying two different aspects of the study, which include its safety and efficacy through early-phase trials, while they still need to determine its sustained effectiveness and immune system response [11,22]. The successful implementation of photoreceptor transplants into existing neural pathways represents a major scientific obstacle that researchers still need to overcome [6].
Gene editing procedures require both ethical oversight and regulatory frameworks as necessary components [16]. The worldwide implementation of the project will depend on its cost-effectiveness and accessibility to different populations [23-25].
Conclusion
Researchers are moving retinal regeneration research from the experimental stage to clinical testing. The combination of stem cell therapy with gene replacement and tissue engineering research enables researchers to establish realistic methods for restoring retinal structure and function. The scientific community needs to conduct interdisciplinary research together with extensive clinical studies to determine the long-term safety and effectiveness of medical treatments.
Acknowledgement
The accomplishment concerning this research project would not have happened without the plentiful support and help of many things and arrangements. We can no longer express our genuine appreciation to all those who risked a function in the progress of this project.
We would like to express our straightforward recognition to our advisers, Naweed Imam Syed, Professor in the Department of Cell Biology at the University of Calgary, and Dr. Sadaf Ahmed, from the Psychophysiology Lab at the University of Karachi, for their priceless counseling and support during the whole of the wholeness of the research process. Their understanding and knowledge assisted in forming the management of this project.
Declaration of interest
I here with acknowledge that: I have no economic or added individual interests, straightforwardly or obliquely, in any matter that conceivably influences or biases my trustworthiness as a journalist concerning this book.
Financial Support and Protection: No external funding for a project was taken to assist with the preparation of this manuscript.
- Wong WL, Su X, Li X, Cheung CMG, Klein R, Cheng CY, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014;2(2):e106-16. Available from: https://doi.org/10.1016/s2214-109x(13)70145-1
- Bourne RRA, Flaxman SR, Braithwaite T, Cicinelli MV, Das A, Jonas JB, et al. Magnitude, temporal trends, and projections of global prevalence of blindness and distance and near vision impairment. Lancet Glob Health. 2017;5(9):e888-97. Available from: https://doi.org/10.1016/s2214-109x(17)30293-0
- Strauss O. The retinal pigment epithelium in visual function. Physiol Rev. 2005;85(3):845-81.
- Jones BW, Pfeiffer RL, Ferrell WD, Watt CB, Marmor M, Marc RE. Retinal remodeling in inherited photoreceptor degenerations. Prog Retin Eye Res. 2016;50:1-36.
- Klein R, Klein BEK. The epidemiology of age-related eye disease. Am J Ophthalmol. 2013;155(3):431-7. Available from: https://doi.org/10.1167/iovs.13-12789
- MacLaren RE, Pearson RA, MacNeil A, Douglas RH, Salt TE, Akimoto M, et al. Retinal repair by transplantation of photoreceptor precursors. Nature. 2006;444(7116):203-7. Available from: https://doi.org/10.1038/nature05161
- Rosenfeld PJ, Brown DM, Heier JS, Boyer DS, Kaiser PK, Chung CY, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med. 2006;355(14):1419-31. Available from: https://doi.org/10.1056/nejmoa054481
- Takahashi K, Yamanaka S. Induced pluripotent stem cells in medicine and biology. Cell. 2016;164(5):847-56. Available from: https://doi.org/10.1242/dev.092551
- Russell S, Bennett J, Wellman JA, Chung DC, Yu ZF, Tillman A, et al. Efficacy and safety of voretigene neparvovec (AAV2-hRPE65v2) for RPE65-mediated inherited retinal dystrophy. Lancet. 2017;390(10097):849-60. Available from: https://doi.org/10.1016/s0140-6736(17)31868-8
- Luo Z, Zhong X. Emerging advances in biomaterials for retinal tissue engineering. Acta Biomater. 2018;84:1-15.
- Hartong DT, Berson EL, Dryja TP. Retinitis pigmentosa. N Engl J Med. 2006;355(5):522-37. Available from: https://doi.org/10.1016/s0140-6736(06)69740-7
- Lamba DA, McUsic A, Hirata RK, Wang PR, Russell D, Reh TA. Generation, purification and transplantation of photoreceptors derived from human induced pluripotent stem cells. PLoS One. 2010;5(1):e8763. Available from: https://doi.org/10.1371/journal.pone.0008763
- Zhong X, Gutierrez C, Xue T, Hampton C, Vergara MN, Cao LH, et al. Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nat Commun. 2014;5:4047. Available from: https://doi.org/10.1038/ncomms5047
- Mandai M, Watanabe A, Kurimoto Y, Hirami Y, Morinaga C, Daimon T, et al. Autologous induced stem-cell–derived retinal cells for macular degeneration. N Engl J Med. 2017;376(11):1038-46. Available from: https://doi.org/10.1056/nejmoa1608368
- Maeder ML, Stefanidakis M, Wilson CJ, Baral R, Barrera LA, Bounoutas GS, et al. Development of a gene-editing approach to restore vision loss in Leber congenital amaurosis type 10. Nat Med. 2019;25(2):229-33. Available from: https://doi.org/10.1038/s41591-018-0327-9
- Doudna JA, Charpentier E. The new frontier of genome engineering with CRISPR-Cas9. Science. 2014;346(6213):1258096. Available from: https://doi.org/10.1126/science.1258096
- Ovando-Rivera P, Thomas BB, Nandrot EF, Hikita ST. Retinal organoids: a window into human retinal development and disease. Stem Cell Reports. 2021;16(5):1039-52. Available from: https://doi.org/10.1242/dev.189746
- Shirai H, Mandai M, Matsushita K, Kuwahara A, Yonemura S, Nakano T, et al. Transplantation of human embryonic stem cell–derived retinal tissue in two primate models of retinal degeneration. Proc Natl Acad Sci U S A. 2016;113(1):E81-90. Available from: https://doi.org/10.1073/pnas.1512590113
- Bainbridge JWB, Mehat MS, Sundaram V, Robbie SJ, Barker SE, Ripamonti C, et al. Long-term effect of gene therapy on Leber congenital amaurosis. N Engl J Med. 2015;372(20):1887-97. Available from: https://doi.org/10.1056/nejmoa1414221
- Schwartz SD, Hubschman JP, Heilwell G, Franco-Cardenas V, Pan CK, Ostrick RM, et al. Embryonic stem cell trials for macular degeneration. Lancet. 2012;379(9817):713-20. Available from: https://doi.org/10.1016/s0140-6736(12)60028-2
- Palanker D, Le Mer Y, Mohand-Said S, Muqit M, Sahel JA. Photovoltaic restoration of central vision in atrophic age-related macular degeneration. Ophthalmology. 2020;127(8):1097-104. Available from: https://doi.org/10.1016/j.ophtha.2020.02.024
- Cehajic-Kapetanovic J, Eleftheriou C, Allen AE, Milosavljevic N, Pienaar A, Bedford R, et al. Restoration of vision with ectopic expression of human rod opsin. Curr Biol. 2015;25(16):2111-22. Available from: https://doi.org/10.1016/j.cub.2015.07.029
- Nakano T, Ando S, Takata N, Kawada M, Muguruma K, Sekiguchi K, et al. Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell. 2012;10(6):771-85. Available from: https://doi.org/10.1016/j.stem.2012.05.009
- Acland GM, Aguirre GD, Ray J, Zhang Q, Aleman TS, Cideciyan AV, et al. Gene therapy restores vision in a canine model of childhood blindness. Nat Genet. 2001;28(1):92-5. Available from: https://doi.org/10.1038/ng0501-92
- Palanker D. Retinal prosthetics and hybrid regenerative approaches. Ophthalmol Sci. 2022;2(3):100154. Available from: https://doi.org/10.1101/cshperspect.a041525
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
