Global Journal of Anesthesiology
Low-Dose Ketamine and Propofol Combination for Upper Endoscopy in Morbidly Obese Patients
Assistant Professor, Anesthesia and Intensive Care, Faculty of Medicine, Al-azhar University,Cairo,Egypt
Author and article information
Cite this as
Salem HA (2017) Low-Dose Ketamine and Propofol Combination for Upper Endoscopy in Morbidly Obese Patients. Glob J Anesth. 2017; 4(1): 6-12. Available from: 10.17352/2455-3476.000030
Copyright License
© 2017 Salem HA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Aim: The purpose of this randomized double-blinded study is to compare the safety and efficacy of two different concentrations of ketofol on the intraoperative hemodynamics, respiration, bispectral index values and post-anesthesia recovery profiles in morbid obese patients undergoing upper GI endoscopy.
Subjects and methods: Patients were randomly allocated into two groups, group I (k/p 1/2), no = 50, and group II (k/p 1/4), no = 50, after receiving ethics committee approval and informed patient’s consent. Patients in group I received bolus dose of ketamine 0.5 mg/kg + propofol 1 mg/kg, and group II received ketamine 0.25 mg/kg + propofol 1 mg/kg, intravenously in separate syringes (dosed ketamine then propofol). Incremental doses, half the fi rst dose of propofol (0.5 mg/kg) were given to get the desired depth of sedation, modifi ed by aiming at bispectral index (BIS) values between 70–80, and when the sedation was considered as inadequate by the endoscopist. The baseline measurements were obtained just before the administration of the study drugs. The primary outcome was emergence reactions (recovery agitation or hallucination) following the procedure.
Results: There was no signifi cant difference between group I (k/p 1/2) and group II (k/p 1/4) as regard to demographic data (age, gender and BMI) and procedure duration. Discharge time from postanesthesia care unit (PACU) was prolonged in group I (Mean±SD = 38.34±4.28) compared to group II (Mean±SD = 33.11±4.89), p = 0.0001. The number of patients requiring propofol top-up doses was 20 (40%) in group I compared to 35 (70%) in group II (the low-dose ketamine group), p = 0.004. There was a significant difference in the mean propofol top-up dose between the two groups (30±15 mg in group I and 41±13 mg in group II, p = 0.0002). No signifi cant difference between both groups as regard to BIS values and VAS pain score.
Conclusion: The use of ketamine and propofol combination for upper GI endoscopy in morbid obese patients appears to be safe, effective and preserve the hemodynamic and respiratory parameters.
Procedural sedation and analgesia refers to the technique of administering sedatives or dissociative agents with or without analgesics to induce an altered state of consciousness that allows the patient to tolerate painful or unpleasant procedures while preserving cardiorespiratory Function [1]. The aim of PSA is mainly to provide sedation, anxiolysis, analgesia and thereby enhances patient cooperation throughout the procedure. It is important to keep in mind that, the level of sedation can easily and quickly passed from conscious to deep sedation and result in loss of protective reflexes and in problems related to airway control [2]. Agents reported to achieve PSA includes midazolam, ketamine, propofol, fentanyl, remifentanil and dexmedetomidine [3]. The use of ketamine and propofol for procedural sedation and analgesia external to the surgical environment has grown in popularity [4]. Sedative drug selection and the dose depend on the patient’s emotional state, the intensity of pain during the examination, anticipated technical difficulties, surgeon’s experience and hospital-specific policy and procedures. An ideal sedation regimen would provide patient comfort, cooperation, hemodynamic stability, amnesia and maintenance of a patent airway with spontaneous ventilation [5]. Propofol has become a preferred sedative because it offers advantages over benzodiazepines in terms of lack of accumulation, quick onset, easy adjustment, and fast recovery after discontinuation. It has sedative and hypnotic effects that mediate the GABA receptor but has no analgesic action. Adverse effects associated with propofol included pain on injection, hypotension, bradycardia, respiratory depression, and hypertriglyceridemia [6]. Ketamine is classified as an NMDA receptor antagonist and has also been found to bind to opioid receptors and sigma receptors. It induces a state referred to as “dissociative anesthesia” [7]. Ketofol was used for PSA and are physically compatible for 1 hr at 23oC with no increase in particle content at Y site injection [8]. This combination can be mixed in the same syringe or administered independently in the two separate syringes. It can be administered as a bolus or as a continuous infusion for longer procedures [9]. The opposing hemodynamic and respiratory effects of each drug may enhance the utility of this drug combination, increasing both safety and efficacy and allowing reduction in the dose of propofol required to achieve sedation [10]. The combination of the two agents appears to reduce side effects of each medication used alone, and allows for a rapid recovery time [11]. Respiratory problems are the most common adverse events associated with propofol use. However, significant respiratory depression is unlikely at propofol bolus doses less than 0.7 mg/kg [12], and minimal with ketamine doses less than 1 mg/kg [13]. Furthermore, the combination of propofol and ketamine may minimize the need for supplemental opioid analgesics and has the potential to provide better sedation with less toxicity than either drug alone [14]. Mortero et al. 2001 [15], suggested that small-dose ketamine increases thalamic sensory output and arousal and sedative effects of propofol may be partially antagonized by the arousal effects of ketamine. The level of sedation can be assessed clinically or with devices such as the bispectral index (BIS). While clinical estimation of sedation is difficult, the BIS is a processed electroencephalographic measurement that gives objective information about the depth of sedation and anesthesia [16].
Aim of the Study
The purpose of this randomized double-blinded study was to compare the safety and efficacy of two different concentrations of ketofol on the intraoperative hemodynamic and respiratory parameters, BIS values and post-anesthesia recovery profiles in morbid obese patients undergoing upper GI endoscopy. The study hypothesis is that the safety and efficacy of performing sedation with ketofol concentration of (1:4) in morbid obese patients is identical to ketofol concentration of (1:2) with the advantage of less postoperative ketamine side effects.
Subjects and Methods
This study included 100 morbid obese adult subjects, scheduled for upper GI endoscopy performed under topical anesthesia with intravenous sedation, during the period from October, 2015 to July, 2016. Patients selected were classified as American Society of Anesthesiologists (ASA) physical status I and II, aged from 18 to 50 years with a Glasgow Coma Scale score 15. Patients were randomly allocated into two groups, group I (k/p1/2), no = 50, and group II (k/p1/4), no = 50, after receiving ethics committee approval and informed consent from patients. All procedures were performed by the same physician.
Exclusion criteria
ASA more than II, pregnant women, patients with drug abuse or had allergy to egg, hypersensitivity to ketamine or propofol, those who had severe bradycardia or any type of atrioventricular block, heart failure or refused to participate in the study.
In the operating room, patients were breathing spontaneously. Nasal oxygen (3 L/min) was administered and intravenous access was established. Standard monitoring (Infinity Delta Monitor, Drager Medical System En, USA) was used. Heart rate (HR) via ECG, non invasive mean arterial pressure (MAP), pulse oximetry (SpO2), were attached. During the procedure, the BIS Sensor (A-2000XP™, Aspect Medical System) was used to assess the level of sedation. The BIS values, hemodynamics and respiratory parameters (HR, MAP and Spo2) were continuously monitored and recorded at the baseline and then every 5 min thereafter during the procedure and in the post anesthesia care unit (PACU). The baseline measurements were obtained just before the administration of the study drugs. Then, these parameters were compared between the two groups at 5 time points; before induction (at baseline), after induction of sedation (after induction), 5 minutes after induction (during procedure), after 15 minutes from admission to post anesthesia care unit (at PACU), and lastly post-procedure at 90 min after the original baseline measurement where postoperative visit was undertaken (at 90 min). The primary outcome was emergence reactions following the procedure. Secondary outcomes included hemodynamics, respiratory profiles, sedation, analgesia, side effects, supplemental propofol and patient as well as endoscopist satisfaction.
All patients were premedicated with ondansetron 0.1 mg/kg, and glycopyrrolate 4 mcg/kg intravenously before induction. Midazolam 0.02 mg/kg was given as premedication. Fentanyl 0.5 mcg/kg was given IV to patients in both groups before the start of endoscopy together with lidocaine local anesthetic spray and ointment. The upper GI endoscope was commenced after 30 seconds from injection of ketofol. Patients in the group I received bolus dose of ketamine 0.5 mg/kg + propofol 1 mg/kg (k/p 1/2), and group II received ketamine 0.25 mg/kg + propofol 1 mg/kg (k/p 1/4) intravenously in separate syringes (separate syringe strategy). Incremental doses, half the initial dose of propofol (0.5 mg/kg) were given to get the desired depth of sedation, modified by aiming at BIS values between 70–80 [17], and when the sedation was judged as inadequate by the endoscopist. A ketamine and propofol dose was prepared by an assistant who was not involved in the clinical management of the study patients.
The patients in the PACU were assessed with the fast–track score. Total score of 14, with minimal score of 12 would be required, with no score below 1 in any individual category [18]. Each patient was informed about how to measure pain intensity on a VAS pain score subjectively using a VAS ruler 0-10, with 0 representing “no pain” and 10 “the worst pain” imaginable. During postoperative visit, satisfaction score was assessed and any adverse events were recorded as pain, nausea, vomiting, shivering, emergence reactions (agitation or hallucination), visual disturbances, myoclonus, seizure, rash, any hypoxic episode or apnea (desaturation was defined as a 10% decrease in peripheral oxygen saturation when compared to baseline, and apnea was defined as cessation of respiration for 15 seconds or more.), any episodes of hypotension (decrease in MAP > 30% of baseline) or bradycardia (decrease in HR > 30% of the initial rate or HR < 55/min). Surgeons and patients were asked to grade their overall satisfaction with sedation technique using a 3 point scale [poor (1), fair (2), and good (3)]. The patients were discharged home with Aldrete’s scores greater than 9 [19].
Statistical analysis
The collected data were statistically analyzed using SPSS version 16.0 (SPSS Inc., USA) for Windows (Microsoft Co, USA). Data were expressed as mean values ± SD or as number and percentages. Fisher’s exact, Student’s t and χ2 tests were used for comparison of the quantitative and qualitative values of the two groups. P < 0.05 was considered statistically significant.
Results
This randomized double-blinded study was conducted during a 10-month period in which 100 morbid obese patients had PSA with ketofol. All patients underwent their planned upper GI endoscopy and received their allocated study drug.
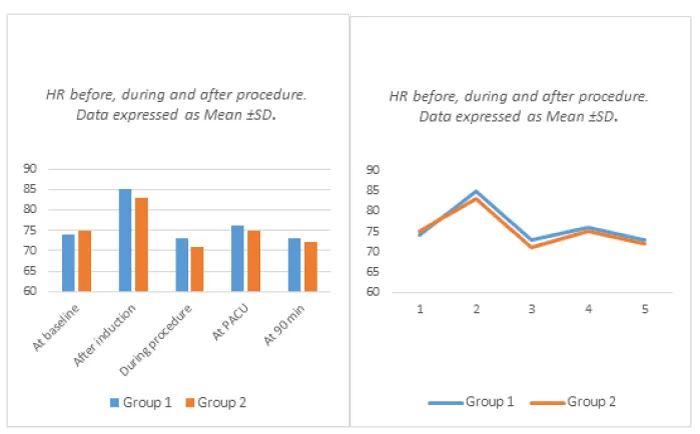
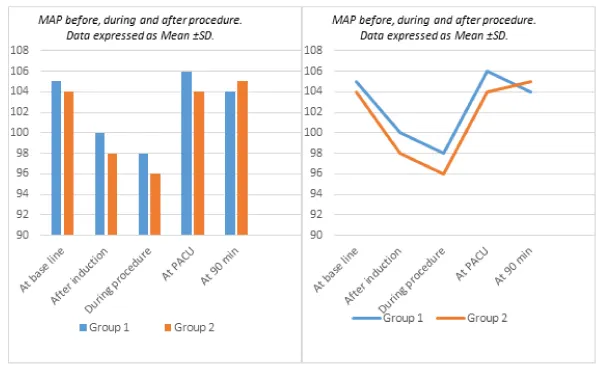
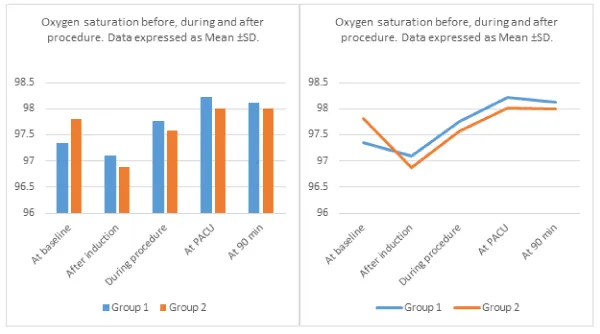
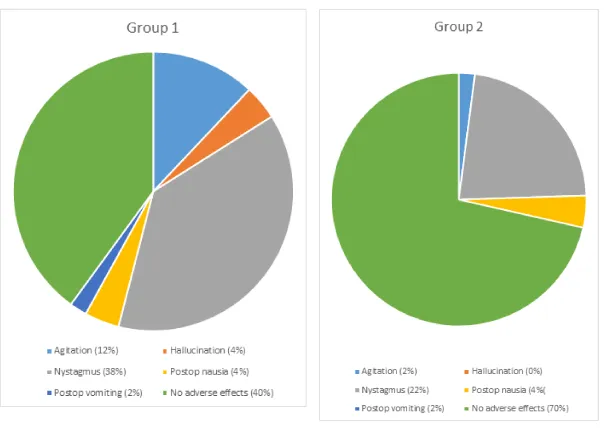
The general characteristics of studied cases and operative data were represented in Table 1. This Table shows that, there was no significant difference between group I (k/p 1/2) and group II (k/p 1/4) as regard to demographic data (age, gender and BMI) and procedure duration. However, discharge time from PACU was prolonged in group I (Mean±SD = 38.34±4.28) compared to group II (Mean±SD = 33.11±4.89), p = 0.0001. The number of patients requiring propofol top-up doses was 20 (40%) in group I compared to 35 (70%) in group II (the low-dose ketamine group), p = 0.004. There was a significant difference in the mean propofol top-up dose between the two groups (30±15 mg in group I and 41±13 mg in group II, p = 0.0002) as shown in Table 1. Heart rate increased after induction of sedation in both groups. The difference between the groups was statistically insignificant (p = 0.267). The change was least in group II (due to low ketamine dose), but no patient had severe tachycardia requiring treatment in both groups. There was a minimal decrease in MAP from baseline in both groups following the initial dose of ketofol. Heart rate and MAP decreased during procedure in group II compared to group I but this decrease is considered to be not quite statistically significant (as p was 0.077 and 0.076 respectively). No significant differences in SpO2 between the two groups (Table 2, Figure 1-3). No cases needed manual ventilation or artificial airway. Overall, cardiovascular and respiratory adverse events were not significantly different between the two groups. These adverse events were transient and easily treated with no sequelae. There were no significant differences between both groups as regard to BIS values and VAS pain score (Table 3). Table (4) shows that there is no significant difference between the two groups regarding patient as well as surgeon satisfaction. Side effects are listed in (Table 5, Figure 4). The most common side effect was visual disturbances. No difference in the occurrence of PONV between the two groups. Agitation on recovery, although not statistically significant (p value = 0.111); was more commonly reported with group I (6 patients, two of them treated with midazolam 0.02 mg/kg intravenously with prompt resolution of the event) compared to the low-dose ketamine group (group II) in which only one patient developed recovery agitation and needs no treatment). A two patients (4%) in group I and no patient (0%) in group II experienced bad dreams and hallucinations. Side effects such as myoclonus, seizure and rash were not observed in any patient.
- Godwin S, Caro D, Wolf S, et al. (2005) American College of Emergency Physicians. ACEP clinical policy: procedural sedation and analgesia in the emergency department. Ann Emerg Med 45: 177-196.
- Kremer M, Odom-Forren J, Watson D (2005) Practical guide to moderate sedation/analgesia. St Louis (Mo): Elsevier Mosby. Pharmacology. 53-80. Link: https://goo.gl/3RxSni
- Panzer O, Moitra V, Sladen R (2009) Pharmacology of sedative-analgesic agents: dexmedetomidine, remifentanil, ketamine, volatile anesthetics, and the role of peripheral mu antagonists. Crit Care Clin 25: 451-469. Link: https://goo.gl/eVFm3p
- Willman E Andolfatto G (2007) A Prospective evaluation of Ketofol (Ketamine/Propofol Combination) for procedural sedation and analgesia in the emergency department. Ann Emerg Med 49: 23-30. Link: https://goo.gl/r1xM8L
- Chen, Wei-Xing, Hui-Jia Lin, Wei-Fang Zhang, Qing Gu, et al. (2005) Sedation and safety of propofol for therapeutic endoscopic retrograde cholangiopancreatography. Hepatobiliary Pancreat Dis Int 4: 437-440. Link: https://goo.gl/lFCUFi
- Jacobi J, Fraser G, Coursin D, Riker RR, Fontaine D, et al. (2002) Clinical practice guidelines for the sustained use of sedatives and analgesics in the critically ill adult. Crit Care Med 30: 119-141. Link: https://goo.gl/d1csOQ
- Harrison N, Simmonds M (1985) Quantitative studies on some antagonists of N-methyl D-aspartate in slices of rat cerebral cortex. Br J Pharmacol 84: 381-391. Link: https://goo.gl/bQWFO8
- Trissl L, Gilbert D, Martinez J (1997) Compatibility of propofol injecTable emulsion with selected drugs during simulated Y-site administration, Am J Health-Syst Pharm 54: 1287-1292. Link: https://goo.gl/bjvDuf
- Amornyotin S (2014) Ketofol: A Combination of Ketamine and Propofol. J Anesth Crit Care. Open Access 1: 00031. Link: https://goo.gl/SNuP2S
- Mohamed D, Medhat E, Rashed A (2009) Assessment of different concentration of Ketofol in procedural operation. BJMP 2: 27-31. Link: https://goo.gl/zvMPmN
- Aouad M, Moussa A, Dagher C, Muwakkit SA, Jabbour-Khoury SI, et al. (2008) Addition of ketamine to propofol for initiation of procedural anesthesia in children reduces propofol consumption and preserves hemodynamic stability. Acta Anaesthesiol Scand 52: 561-565. Link: https://goo.gl/7TRRMX
- Smith I, Monk T, White P, Ding Y (1994) Propofol infusion during regional anesthesia: sedative, amnestic and anxiolytic properties. Anesth Analg 79: 313-39. Link: https://goo.gl/f25FKD
- Schuttler J, Zsigmond E, White P (1997) Ketamine and its isomers. In: White PF, ed. Textbook of intravenous anesthesia. Baltimore: Williams & Wilkins 171-188. Link: https://goo.gl/SoEHjC
- Frizelle HP, Duranteau J, Samii K (1997) A comparison of propofol with a propofol-ketamine combination for sedation during spinal anesthesia. Anesth Analg 84: 1318-1322. Link: https://goo.gl/OjrDK3
- Mortero RF, Clark LD, Tolan MM, Metz RJ, Tsueda K, et al. (2001) The Effects of Small-Dose Ketamine on Propofol Sedation: Respiration, Postoperative Mood, Perception, Cognition, and Pain. Anesth Analg 92: 146-159. Link: https://goo.gl/BY1Jwm
- Struys M, Versichelen L, Byttebier G, Mortier E, Moerman A, et al. (1998) Clinical usefulness of the bispectral index for titrating propofol target effect-site concentration. Anaesthesia 53: 4–12. Link: https://goo.gl/8iNT4M
- Scott D Kelley (2007) Monitoring Consciousness using the bispectral Index™ (BIS™) during anesthesia; a pocket guide for Clinicians. Second edition 13-14. Link: https://goo.gl/hEjfjR
- Paul F, Dajun (1999) New Criteria for Fast-Tracking after outpatient anesthesia: A Comparison with the Modified Aldrete’s Scoring System. Anesth Analg 88: 1069-1072. Link: https://goo.gl/h8hcgn
- White P, Song D (1999) New Criteria for Fast-Tracking after outpatient anesthesia: a comparison with the Modified Aldrete’s Scoring System. Anesth Analg 88: 1069-1072. Link: https://goo.gl/h8hcgn
- Ayatollahi V, Vafaiyan M, Hatami M, Behdad S (2016) Two Different Concentrations of Ketofol for Procedural Sedation and Analgesia in Closed Reduction of Nasal Fracture. Journal of Craniofacial Surgery 27: 996–1000. Link: https://goo.gl/tSdYy9
- Miner J,Moore J,Austad E,Plummer D,Hubbard L, et al. (2015) Randomized, double-blinded clinical trial of propofol, 1:1 propofol/ketamine, and 4:1 propofol/ketamine for deep procedural sedation in the emergency department. Ann Emerg Med 65: 479-88. Link: https://goo.gl/Dkyats
- Coulter S, Hannam A, Anderson J (2014) Ketofol dosing simulations for procedural sedation. Pediatr emerg care 30: 621-630. Link: https://goo.gl/C3NhtU
- Dal T, Sazak H, Tunc M, Sahin S, Yilmaz A (2014) A comparison of ketamine-midazolam and ketamine-propofol combinations used for sedation in the endobronchial ultrasound guided transbronchial needle aspiration: a prospective, single blind, randomized study. J Thorac Dis 6: 742-751. Link: https://goo.gl/rmdZy2
- Ghadami Yazdi A, Ayatollahi V, Hashemi A, Behdad S, Ghadami Yazdi E (2013) Effect of two different concentrations of propofol and ketamine combinations (Ketofol) in pediatric patients under lumbar puncture or bone marrow aspiration. Iranian J Pediatr Hematol Oncol 3: 187-192. Link: https://goo.gl/Jqghaq
- Wang Y, Jiang X, Pang L, Dong S, Feng Y, et al. (2012) A randomized double-blind controlled study of the efficacy of ketofol with propofol-fentanyl and propofol alone in termination of pregnancy. Afr J Pharm Pharmacol 6: 2510-2514. Link: https://goo.gl/hoitYE
- Amornyotin S, Chalayonnawin W, Kongphlay S (2012) Clinical efficacy of the combination of propofol and ketamine (ketofol) for deep sedation for colonoscopy. Gut 61: A339-A340. Link: https://goo.gl/6hfBgL
- Kayhan G, Yucel A, Colak Y, Ozgul U, Yologlu S, et al. (2012) Ketofol (mixture of ketamine and propofol) administration in electroconvulsive therapy. Anaesth Intensive Care 40: 305-10. Link: https://goo.gl/6cXFi7
- Hashemi A, Ayatolahi V, Ghilian R, Ghadami Y, Fadavi N, et al. (2011) Ketofol for bone marrow aspiration and lumbar puncture in Children with ALL. Iranian journal of pediatric hematology oncology 1: 126–132. Link: https://goo.gl/3v86ko
- Phillips W, Anderson A, Rosengreen M, Johnson J, Halpin J (2010) Propofol versus propofol/ketamine for brief painful procedures in the emergency department: clinical and bispectral index scale comparison. J Pain Palliat Care Pharmacother 24: 349-55. Link: https://goo.gl/fOcQfB
- Akin A, Esmaoglu A, Guler G, R Demircioglu, N Narin, et al. (2005) Propofol and propofol-ketamine in Pediatric patients undergoing cardiac catheterization. Pediatr Cardiol 26: 553-557. Link: https://goo.gl/AWJkea
- Akin A, Guler G, Esmaoglu A, Bedirli N, Boyaci A (2005) A comparison of fentanyl-propofol with a ketamine-propofol combination for sedation during endometrial biopsy. J Clin Anesth 17: 187-190. Link: https://goo.gl/cWgqAe
- Goh PK, Chiu CL, Wang CY, Chan YK, Loo PL (2005) Randomized double-blind comparison of ketamine-propofol, fentanyl-propofol and propofol saline on haemodynamics and laryngeal mask airway insertion conditions. Anaesth Intensive Care 33: 223-228. Link: https://goo.gl/orYmM2
- Furuya A, Matsukawa T, Czaki M, Nishiyama T, Kume M, et al. (2001) Intravenous ketamine attenuates arterial pressure changes during induction of anesthesia with propofol. Eur J Anesthesiol 18: 88-92. Link: https://goo.gl/GTEkxJ
- Badrinath S, Avramov MN, Shadrick M, Witt TR, Ivankovich AD (2000) The use of a ketamine-propofol combination during monitored anesthesia care. Anesth Analg 90: 858-862. Link: https://goo.gl/25sVWU
- Friedberg BL (1999) Propofol-ketamine technique: dissociative anesthesia for office surgery (a 5-year review of 1264 cases). Aesthetic Plast Surg 23: 70-75. Link: https://goo.gl/KGkuDk
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
 Help ?
Help ?

If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."


 Save to Mendeley
Save to Mendeley