Global Journal of Anesthesiology
Intravenous General Anesthesia for Patients with Neurological Disorders
Department of Dental Anesthesiology, Assistant professor, Field of Maxillofacial Diagnostic and Surgical Sciences, Faculty of Dental Science, Kyushu University, Graduate School, Japan
Author and article information
Cite this as
Ouchi K (2015) Intravenous General Anesthesia for Patients with Neurological Disorders. Glob J Anesth. 2015; 2(2): 54-56. Available from: 10.17352/2455-3476.000018
Copyright License
© 2015 Ouchi K. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.In dental practice, intravenous general anesthesia is useful for patients who are difficult to treat when not sedated such as those with neurological disorders [1].
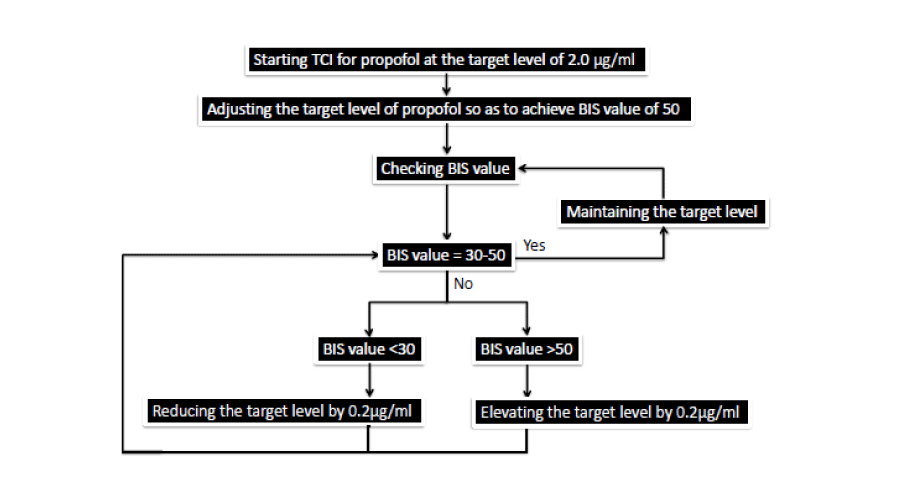
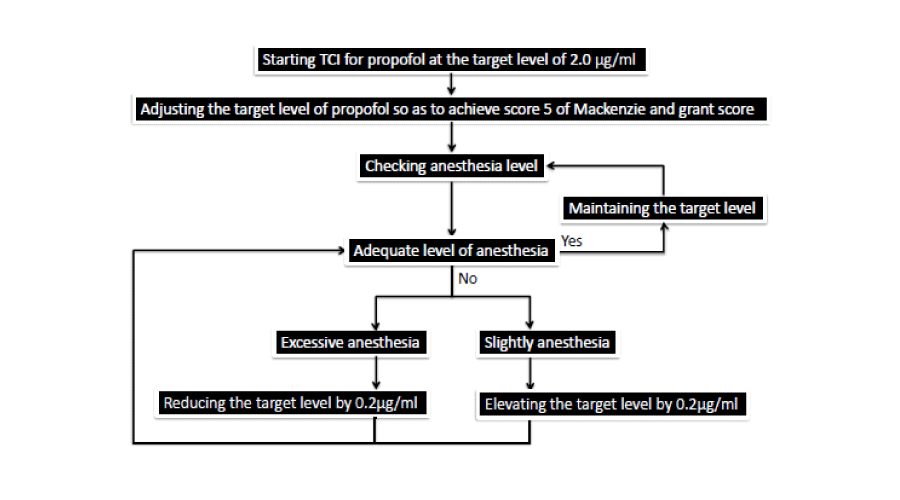
In dental practice, intravenous general anesthesia is useful for patients who are difficult to treat when not sedated such as those with neurological disorders [[4], continuous intravenous infusion of propofol was initiated using the TCI method. The dose of propofol was titrated to achieve a BIS of 50 and achieve an adequate level of anesthesia: asleep but not responding to stimulation. Endotracheal intubation was not performed, and spontaneous breathing was maintained. The level of anesthesia was maintained at BIS 30-50 by adjusting the target propofol level using TCI (Figure 1). Without BIS, The dose of propofol was titrated to achieve a Mackenzie Grant score of 5 and to achieve an adequate level of anesthesia: asleep, but not responding to stimulation. The level of anesthesia was maintained at a Mackenzie and Grant score of 5 by adjusting the target propofol level using the propofol TCI (Figure 2). If respiratory depression was observed or BIS value was less than 30, the target blood concentration of propofol was decreased by 0.2 μg/ml. If the anesthesia level was deemed inadequate BIS value was more than 50, the target blood level of propofol was increased by 0.2 μg/ml. The dental procedure was started after the anesthesia level became stable without respiratory depression. A local anesthetic was used appropriately by the operating dentist. Administration of propofol was discontinued at the end of the dental procedure. Patients were monitored until recovery from anesthesia, when they were fully awake and had stable respiration.
Antiepileptics affect for anesthesia
Patients with intellectual disabilities need higher doses of sedatives than those without intellectual disabilities to obtain an adequate level of anesthesia [[6]. Many of neurological disorders patients have epilepsy and are medicated with antiepileptic drugs. In these reports, the group with lower doses of sedatives to obtain an adequate level included those who were given an antiepileptic. In patients not given an antiepileptic, there were no differences in the required dose of propofol and emergence among patients with autism, cerebral palsy, and intellectual disability [8]-[7].
Hepatic enzyme inhibition usually occurs because of competition at the enzyme site and results in a decrease in the rate of metabolism of the affected drug [[12]. Thus, certain antiepileptic drugs have been increased the blood concentration of propofol by inhibiting the action of CYP and UGT. Carbamazepine contributes to the competitive inhibition of hepatic CYP2C9, because metabolism CYP is the same as propofol.(7) In addition, carbamazepine inhibits 2C19 [[7]. In addition, valproate inhibits CYP2C9 in vitro [15]. And valproate inhibits UGT 1A9, which mediates glucuronic acid conjugation, the main metabolic pathway of propofol [20],[17],22]. But, phenobarbital induces CYP2C19 [[24]. But, it is reported that zonisamide does not induce or inhibit the metabolism of other drugs that included drugs metabolize by CYP3A4 or CYP2C19 [2525. Sills G, Brodie M (2007) Pharmacokinetics and drug interactions with zonisamide. Epilepsia 48: 435-441.]. Therefore, zonisamide may not affect the metabolism of propofol.
Therefore, propofol metabolism is affected by antiepileptic use, and is affected by the type of antiepileptic (Table 1). Consequently, in intravenous anesthesia for patients with epilepsy, required propofol dose is lower, emergence from anesthesia is delayed.
- Manley MC, Skelly AM, Hamilton AG
- (2000) Dental treatment for people with challenging behaviour: general anaesthesia or
- sedation? British dental journal 188: 358-360 .
- Keidan I, Gravenstein D,
- Berkenstadt H, Ziv A, Shavit I, et al. (2008) Supplemental oxygen compromises the use of
- pulse oximetry for detection of apnea and hypoventilation during sedation in simulated
- pediatric patients. Pediatrics 122: 293-298 .
- Fu ES, Downs JB, Schweiger JW,
- Miguel RV, Smith RA (2004) Supplemental oxygen impairs detection of hypoventilation by
- pulse oximetry. Chest 126:1552-1558 .
- Marsh B, White M, Morton N, Kenny GN
- (1991) Pharmacokinetic model driven infusion of propofol in children. British journal of
- anaesthesia 67: 41-48 .
- Miyawaki T, Kohjitani A, Maeda S,
- Egusa M, Mori T, et al. (2004) Intravenous sedation for dental patients with intellectual
- disability. Journal of intellectual disability research : JIDR 48: 764-768 .
- Asahi Y, Kubota K, Omichi S (2009)
- Dose requirements for propofol anaesthesia for dental treatment for autistic patients
- compared with intellectually impaired patients. Anaesthesia and intensive care 37: 70-73 .
- Ouchi K, Sugiyama K (2015) Required
- propofol dose for anesthesia and time to emerge are affected by the use of antiepileptics:
- prospective cohort study. BMC Anesthesiol 15: 34 .
- Oda Y, Hamaoka N, Hiroi T, Imaoka
- S, Hase I, et al. (2001) Involvement of human liver cytochrome P4502B6 in the metabolism
- of propofol. Br J Clin Pharmacol 51: 281-285 .
- Court MH, Duan SX, Hesse LM,
- Venkatakrishnan K, Greenblatt DJ (2001) Cytochrome P-450 2B6 is responsible for
- interindividual variability of propofol hydroxylation by human liver microsomes.
- Anesthesiology 94: 110-119.
- Guitton J, Buronfosse T, Desage M,
- Flinois JP, Perdrix JP, et al. (1998) Possible involvement of multiple human cytochrome
- P450 isoforms in the liver metabolism of propofol. Br J Anaesth 80: 788-795 .
- Perucca E (2002) Pharmacological
- and therapeutic properties of valproate: a summary after 35 years of clinical experience.
- CNS drugs 16: 695-714 .
- Anderson GD (1998) A mechanistic
- approach to antiepileptic drug interactions. Ann Pharmacother. 32: 554-563.
- Yap KY, Chui WK, Chan A (2008) Drug
- interactions between chemotherapeutic regimens and antiepileptics. Clin Ther. 30:
- 1385-1307 .
- Sachdeo RC, Sachdeo SK, Levy RH,
- Streeter AJ, Bishop FE, et al. (2002) Topiramate and phenytoin pharmacokinetics during
- repetitive monotherapy and combination therapy to epileptic patients. Epilepsia 43:
- 691-696 .
- Wen X, Wang JS, Kivisto KT,
- Neuvonen PJ, Backman JT (2001) In vitro evaluation of valproic acid as an
- inhibitor of human cytochrome P450 isoforms: preferential inhibition of cytochrome P450
- 2C9 (CYP2C9). Br J Clin Pharmacol 52: 547-553 .
- Ethell BT, Anderson GD, Burchell B
- (2003) The effect of valproic acid on drug and steroid glucuronidation by expressed human
- UDP-glucuronosyltransferases. Biochem Pharmacol 65: 1441-1449 .
- Mutlib AE, Goosen TC, Bauman JN,
- Williams JA, Kulkarni S, et al. (2006) Kinetics of acetaminophen glucuronidation by
- UDP-glucuronosyltransferases 1A1, 1A6, 1A9 and 2B15. Potential implications in
- acetaminophen-induced hepatotoxicity. Chem Res Toxicol 19: 701-709 .
- Levy RH BM (1995) Phenytoin: interactions with other drugs: mechanistic aspects. In: Levy
- RH MR, Meldrum BS editor. Antiepileptic drugs. Volume. 4 ed. New York: Raven Press 329-344.
- Walzer M, Bekersky I, Blum RA,
- Tolbert D (2012) Pharmacokinetic drug interactions between clobazam and drugs metabolized
- by cytochrome P450 isoenzymes. Pharmacotherapy 32: 340-353 .
- Sankar R (2012) GABA(A) receptor
- physiology and its relationship to the mechanism of action of the 1,5-benzodiazepine
- clobazam. CNS drugs 26: 229-244 .
- Wheless JW, Phelps SJ (2013)
- Clobazam: a newly approved but well-established drug for the treatment of intractable
- epilepsy syndromes. J Child Neurol 28: 219-229 .
- Kostrubsky SE, Sinclair JF, Strom
- SC, Wood S, Urda E, et al. (2005) Phenobarbital and phenytoin increased acetaminophen
- hepatotoxicity due to inhibition of UDP-glucuronosyltransferases in cultured human
- hepatocytes. Toxicol Sci 87: 146-155 .
- Zhou SF, Zhou ZW, Yang LP, Cai JP
- (2009) Substrates, inducers, inhibitors and structure-activity relationships of human
- Cytochrome P450 2C9 and implications in drug development. Curr Med Chem 16: 3480-3675 .
- Nakasa H, Nakamura H, Ono S, Tsutsui
- M, Kiuchi M, et al. (1998) Prediction of drug-drug interactions of zonisamide metabolism
- in humans from in vitro data. Eur J Clin Pharmacol 54: 177-183 .
- Sills G, Brodie M (2007)
- Pharmacokinetics and drug interactions with zonisamide. Epilepsia 48: 435-441 .
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
