International Journal of Dermatology and Clinical Research
Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis Reactions to Ibuprofen and Acetaminophen: Clinical, Laboratory Assessment Using In Vitro Lymphocyte Toxicity Assay to Personalized Therapy
1<em>In Vitro</em> Drug Safety and Biotechnology, Toronto, ON, Canada
2Department of Pharmacology and Toxicology, Faculty of Medicine, University of Toronto, Toronto, ON, Canada
3Professor of Surgery and Emeritus Chief of Plastic Surgery, New York Medical College and Emeritus Director of the Burn Center, New York, USA
4Steven Johnson Consortium, Dallas, Texas, USA
Author and article information
Cite this as
Manuela N, et al. Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis Reactions to Ibuprofen and Acetaminophen: Clinical, Laboratory Assessment Using In Vitro Lymphocyte Toxicity Assay to Personalized Therapy. Int J Dermatol Clin Res. 2026; 12(1): 001-016. Available from: 10.17352/ijdcr.000057
Copyright License
© 2026 Manuela N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Introduction: Stevens-Johnson Syndrome (SJS) and Toxic Epidermal Necrolysis (TEN) are serious cutaneous adverse reactions (SCARs). SJS is defined when 10% of the body’s skin is involved. When epidermal detachment involves 30% or more, it is called TEN. The syndrome affects 1-12 cases per million patients. The risk of death for SJS is 5-10%, and for TEN, 25% -50%. We used our proprietary test lymphocyte toxicity assay (LTA) to identify SJS/TEN to several medications. In this article, we present the role of LTA to personalize the diagnosis of acetaminophen (APAP) and ibuprofen-induced SJS-TEN.
Aims: -to ensure that the cases of SJS and TEN clinically assessed by a burn surgeon and having positive skin biopsies correlate with the positive in vitro LTA results, to ibuprofen/ APAP,
-to identify defects in the mmune-responses of the SJS and TEN patients and confirm the validity of using LTA for the causality assessment for SJS and TEN from acetaminophen and ibuprofen.
Material and Methods: Patients with biopsy-proven SJS and TEN agreeing to send their blood to our laboratory have been enrolled in the study. Each one of the individual samples had a control sample given by one of the parents. The LTA was performed at the same time as the control and the patient, and the results were sent to the clinic. Also, we performed cytokine and chemokine analysis to understand the role of the immune system in the disease.
Results: There were samples of Black (3) and Caucasian (3) children; 2 teenage males (1 Hispanic, 1 Black); 3 females (1 Caucasian, 1 Filipino, 1 Hispanic). The LTA positive results are shown in the table. Analysis of proinflammatory cytokines showed a high level of inflammation.
Discussion: The study found that accurate differential diagnosis is a crucial step in identifying true immune-mediated hypersensitivity reactions (HSR), which differ from pseudo-allergic conditions. This approach minimizes the risks to the patient associated with improper treatment or medications.
Determining the type of HSR, considering immunological reactions and laboratory analysis, permits a clear diagnosis, emphasizing the need for a comprehensive approach to diagnosis of SCAR events, which includes clinical history and the use of personalized laboratory methods.
Conclusion: Treating patients with SCARS is a continuous collaboration between the clinical and laboratory teams. Personalized medicine is the solution to propose to the patients.
AA: Antipyretic Analgesics; ADR: Adverse Drug Reactions; AERS: Adverse Event Reporting System; AGEP: Acute Generalized Exanthematous Pustulosis; ALDEN: Algorithm Diagnostic for Epidermal Necrolysis; APAP: Acetaminophen; CC: Chemokines with two N-terminal Cysteines; CI: Confidence Interval; CXC: Chemokines presenting an amino acid between the two N-terminal cysteine residues; DRESS: Drug Reactions with Eosinophilia and Systemic Symptoms; EBGM: Empirical Bayes Geometric Mean; ECM: Extracellular Matrix; ELISA: Enzyme-linked Immunosorbent Assay; EMM: Erythema Multiforme Major; EN: Epidermal Necrolysis; HLA: Human Leukocyte Antigen; HPLC: High-Performance Liquid Chromatography; HSR: Hypersensitivity Reactions; ICU: Intensive Care Unit; IDHR: Immune-Mediated Drug Hypersensitivity Reactions; IGF: Interferon Growth Factor; IFN: Interferon; IL: Interleukin; INR: International Normalized Ratio; IVIG: Intravenous Immunoglobulin; LTA: Lymphocyte Toxicity Assay; LTT: Lymphocyte Transformation Test; MCP-1: Monocyte Chemoattractant Protein 1; MELD: Model for end-stage Liver Disease; MHC: Major Histocompatibility Complex; MIG/CXCL9: T cell Chemokine Inducible by Interferon; MIP-1: Macrophage Inflammatory Protein 1; MIRM: Mycoplasma – Induced Rash and Mucositis; MLOSCAR: Medium- and Late-Onset Severe Cutaneous Adverse Reactions; MMP: Matrix Metalloproteinases; NK cells: Natural killer Cells; NSAIDs: Non-Steroidal Anti-Inflammatory Drugs; OCD: Over-the-Counter Drugs; PBMC: Peripheral Blood Mononuclear Cells; RANTES: Regulated upon Activation, Normally Expressed by T-Cells, and Presumably Aecreted; RIME: Recurrent Reactive Infectious Mucocutaneous Eruption; SCARS: Severe Cutaneous Adverse Reactions; SJS: Steven-Johnson Syndrome; SOCs: Severe Ocular Surface Complications; SOCS: Suppressor of Cytokine Signaling; STAT: Signal Transducer and Activator of Transcription; TEN: Toxic Epidermal Necrolysis; TIMP: Tissue Inhibitor of Metalloproteinases; Th: T Helper; TLRs: Toll-Like Receptors; TGF-β: Transforming Growth Factor Beta; TNF-α: Tumor Necrosis Factor-Alpha; Treg: T Regulatory
Introduction
Severe Cutaneous Adverse Reactions (SCAR) such as SJS and TEN (SJS/TEN) are associated with significant patient morbidity and mortality. These reactions may result in death or life-threatening conditions, inpatient prolonged hospitalization, or permanent disability. SJS and TEN are caused by medicines in 90% cases2. SJS/TEN leads to disabling complications affecting the eyes, lungs, genito-urinary tract, gastrointestinal epithelium, and other organs. By identifying early diagnostic indicators and understanding the typical progression of the syndrome, clinicians can promptly recognize and manage SCARS like SJS and TEN.
The aims of the study were: 1- to present the clinical and laboratory manifestations of SJS and TEN cases in one U.S. Burn Unit, 2- to investigate the mechanisms of SJS and TEN - diagnostic approaches, 3- to develop clinical and laboratory diagnostic tests to identify SCAR reactions, as well as to develop laboratory recommendations for their personalized treatment.
The methodology included a review of publications and clinical cases, which allowed us to describe types of hypersensitivity according to the Gell and Coombs’ classification. The reactions we analyzed are type IV (T-cells) and were accompanied by skin reactions or systemic symptoms. The study found that accurate differential diagnosis is a crucial step in identifying true immune-mediated hypersensitivity reactions (HSR), which differ from pseudoallergic conditions. A detailed analysis of the mechanisms and clinical manifestations allows accurately determining the type of hypersensitivity, considering both classical immunological reactions and laboratory analysis.
The conclusions emphasize the need for a comprehensive approach to the diagnosis of SCAR events, which includes a thorough drug and clinical history, the use of modern laboratory methods, and differential analysis of clinical symptoms. It is especially important to consider the possibility of mimetic conditions such as DRESS, AGEP (Acute generalized exanthematous pustulosis), MIRM (Mycoplasma – Induced Rash and Mucositis), and recurrent reactive infectious mucocutaneous eruption (RIME), or other pseudo- allergic reactions that require a different therapeutic approach. Assessing immunotoxicity leading to SCAR events should avoid the IDHRs in sensitive individuals. At the same time, the patients who do not present sensitivity to the specific drug should use the medication with confidence.
New Data Reveals Increasing Incidence of SJS and TEN and Adverse Effects on Public Health
New studies have reported higher and rising incidences of SCARs in the U.S., which also include hospitalized cases of erythema multiforme major (EMM), SJS, and TEN. SJS and TEN are life-threatening cutaneous and mucocutaneous skin disorders characterized by widespread epidermal necrosis of the skin and mucosa [1,2]. Studies assessing the public health impact of SJS and TEN found that the mean estimated incidence of SJS had risen in the U.S., reported to be 9.2 per million adults per year. Adults with SJS and TEN had 4-fold longer duration and 5-fold higher mean cost of hospitalization compared with that of an average hospital admission [1].
There has been some recent concern over the rising number of SJS and TEN cases caused by over-the-counter drugs (OCD), such as non-steroidal anti-inflammatory drugs (NSAIDs), such as ibuprofen and acetaminophen (APAP). This generated significant interest in developing an in vitro diagnostic assay to more accurately confirm the etiologic drug of a patient’s SJS and TEN, to prevent recurrent or accidental exposure to ibuprofen or APAP, or both drugs [3]. SJS and TEN are acute, life-threatening, drug-induced reactions that carry an inpatient mortality rate of 35-40% [4]. However, the harmful effects of SJS, SJS-TEN, and TEN on mortality extend beyond the inpatient setting. An international registry study of 460 patients with SJS, SJS/TEN, and TEN found that mortality rates increased over time to 1-year mortality rates of 24%, 43%, and 49%, respectively [5]. These patients may require additional interventions to reduce their excess mortality risk even after discharge from the hospital. This is particularly important given that 44.9% of SJS and TEN patients in the U.S. in the study by Hsu et al. were transferred to short-term hospitals or received home health care. The financial burden of providing treatment for a patient with SJS and TEN is high [1].
In the U.S., recent studies have reported a mean estimated incidence of SJS and TEN ranging from 5.3 to 10 per million children per year [6,7]. In the pediatric population, the total costs for acute care were $63 million for 4 years. Children with SJS-TEN also had up to a 4-fold longer duration and 9-fold higher mean cost of hospitalization in comparison to other pediatric admissions in the U.S.A [6].
The incidence of hospitalized cases of SCARs has been reported to be much higher in U.S. children, with an incidence of 35.5 cases per million per year [7,8]. By contrast, in the U.K., the incidence of SJS and TEN in adults was lower than in the U.S. with an incidence rate of 5.76 SJS/TEN cases per million persons, between 1995 and 2013 [9].
Diagnosis of SCARs to medications and identification of their pathogenetic mechanisms and at-risk populations improves the management of the patients. Early recognition and diagnosis of SCARs are key to the identification of culprit drugs. SCARS are associated with morbidity during the acute stage of SJS and TEN, drug reactions with eosinophilia and systemic symptoms (DRESS). Early drug withdrawal is mandatory in all SCARs. Physicians’ knowledge and collaboration with the laboratory specialist are essential to the improvement of diagnosis and management, and in the prevention of long-term sequelae of SCARs. The cases involving SJS and TEN include children, teens, adults, and the elderly. The patients are both males and females (2:1) of a variety of races. SJS-TEN cases are considered a medical emergency that requires immediate hospitalization. Countless drugs can precipitate SJS, but the most common medications include non-steroidal anti-inflammatory drugs (e.g. ibuprofen), antipyretics (APAP).
The laboratory can provide personalized medicine for keeping clinicians informed about the safety profiles of medications that can cause SJS and TEN. Strategies for predicting the risks for SJS and TEN and managing treatment-related SCAR adverse events are essential, alongside implementing effective clinical decisions and appropriately communicating, educating, and counseling patients and their families about the risks of SJS and TEN both before and after the event. There is a lack of a standard protocol on how to diagnose and treat SJS and TEN. For example, a common practice for the diagnosis of SJS and TEN includes obtaining a skin biopsy, but this is only performed in 77% of burn intensive care units [10]. A recent survey assessing the type of care being provided for SJS and TEN patients in North America revealed that there was a crucial need for up-to-date treatment guidelines for patients with SJS and TEN. This need was confirmed by 70% of burn centers’ directors [11].
An international expert consensus panel at the International Drug Allergy Symposium held at the Joint Congress of the American Academy of Allergy, Asthma & Immunology/World Allergy Organization (March 1, 2018, in Orlando, Florida, USA) noted that there is a need for clinical care and treatment for SJS and TEN using a standard treatment guidelines, and more importantly a laboratory means of a reliable and accurate in vitro testing that confirms a certain drug is the cause of the inflammatory response, and the patient’s SJS and TEN [12].
We established a specific recognized biomarker, in vitro LTA,that has a high sensitivity and accuracy rate. The biomarker test is employed to determine the culprit drug that provoked the skin damage in patients diagnosed with SJS and TEN, especially for drugs that have been known to have a causal association with SCAR events like SJS/TEN. Laboratory precision analysis, LTA, is inexpensive and can be used to predict drug hypersensitivity of patients before exposure, avoiding any harm to the patient. The in vitro LTA may also increase the accuracy of determining the drug-induced causality for SCAR events and could be used for screening potential “cross-reacting” drugs [13-16].
Using the LTA, we previously established and validated the link between the need for precision laboratory biomarkers and clinical importance in patients who were diagnosed with APAP and ibuprofen-induced SJS - TEN, anticonvulsant, quinolone antibiotics, and sulfonamide-induced SJS-TEN. All the analyzed blood samples from SCAR individuals presented a high level of cytotoxicity to the incriminating drugs over several years after remission. Furthermore, these incriminating drugs can also markedly stimulate the peripheral blood mononuclear cells (PBMC)s to produce a variety of proinflammatory cytokines, leading to cell death [14, 17-20].
Other studies, including pharmaceutical industry-supported clinical interventions, employed in vitro assays to establish the causal role of SJS and TEN associated with the use of APAP, anti-epileptic drugs, or antibiotics [21-22]. Providing an accurate in vitro diagnostic assay to clinicians to integrate these tests into their treatment guidelines for SJS-TEN would help mitigate the healthcare costs associated with the treatment of SJS-TEN by avoiding drugs that were responsible for precipitating the initial SJS-TEN.
SJS and TEN are immune-mediated type IV hypersensitivity reactions (HSR)s encompassing CD8+ T lymphocytes. The regulatory T cells and natural killer (NK) cells are also involved. Inflammatory mediators such as cytokines and chemokines produce inflammatory reactions leading to cell death by apoptosis and necrosis [14]. Genomic variations in human leukocyte antigens (HLA) genes have been implicated in the susceptibility and severity of APAP-induced SJS/TEN [20]. APAP’s use and its associated skin pathologies SJS and TEN, warrant an in-depth examination of the causative processes involved in their pathogenesis. Both physicians and patients must be made aware that while APAP is widely tolerated by most individuals, severe and potentially fatal interactions do occur [21-40].
Mechanism of action
In the figure 1, we explain the mechanism of APAP-induced epidermal cell damage. Acetaminophen is metabolized by cytochrome P450 2E1 (CYP 2E1) to N-acetyl-p-benzoquinone-imine (NAPQI), which is a toxic product. If in the cell there is enough glutathione (GSH), the non-toxic formed NAPQI-GSH is excreted via urine or via bile.
The APAP level in urine is based upon the Rumack–Matthew [37] line that began at 200 µg/mL APAP at four hours post-ingestion. The clinical trials of N-Acetyl Cysteine began, and the FDA required a line 25% low, beginning at 150 µg/mL. In addition, new ways to use the nomogram have been introduced, such as the APAP ratio, which is the first APAP value measured between 4 and 24 h post-ingestion divided by the APAP concentration on the standard nomogram line at the same time point. Moreover, if the person has HSR, NAPQI binds covalently to the mitochondria and leads to toxicity of the skin cell, cell death, and SCARs. In addition, proinflammatory cytokines lead to inflammatory reactions, enhance caspase 3 activation, and cell death.
Biodegradation pathways for ibuprofen have been identified. Hydroxylation is the primary mechanism, which is either via its binding to coenzyme A (CoA) or via the direct tri-hydroxylation of its aromatic ring. Aliphatic monooxygenases perform the hydroxylation of the isobutyl chain to produce 1-hydroxy-ibuprofen or 2-hydroxy-ibuprofen, which are then di-hydroxylated by acyl-CoA synthase to hydroquinone derivatives. Propionic acid hydroxylation creates ibuprofen-CoA, which is then hydroxylated by dioxygenases and deacetylated, adding CoA to propionic acid to produce p-isobutyl catechol. Another route is by reducing propionic acid’s carboxyl group to ibupro-phenol and then ibuprophenol-acetate. Finally, the hydroxylation of ibuprofen’s aromatic ring by aliphatic monooxygenases creates trihydroxy ibuprofen, destabilizing it due to cleavage via the meta-cleavage pathway. Hydroxylation is the main mechanism, which is via its binding to coenzyme A (CoA). Conjugation with glutathione, glucuronic acid, or sulfates is taken out of the body by detoxification.
The low glutathione reserves led to a high level of metabolites and cellular toxicity [38-55]. Significant individual variability has been noted [56]. In many cases, the variability is linked to genetic polymorphisms of drug-metabolizing enzymes. Individuals carrying enzyme-inactivating mutations display impaired drug metabolism, resulting in higher plasma drug concentrations and lower clearance, leading to adverse drug reactions. This effect is of high importance in the case of non-steroidal anti-inflammatory drugs (NSAIDs) because millions of people are treated daily with these drugs [57,58].
Aims/purpose of study
Using Bastuji-Garin clinical diagnostic criteria and strict inclusion criteria of biopsy-proven cases of SJS and TEN, the present study is designed to:
- determine if cases of SJS and TEN correlated with the positive in vitro LTA to APAP or ibuprofen results, clinical assessment by a burn surgeon, and positive skin biopsies,
- to identify defects in the immunoresponses of the SJS and TEN patients and confirm the validity of using LTA for the causality assessment for SJS and TEN from APAP and ibuprofen.
Methodology
All the SJS and TEN cases have been clinically diagnosed by an RN (burn surgeon) in the Burn Unit. The blood was taken from each individual and a control and sent to In Vitro Drug Safety and Biotechnology for analysis.
In the review of the literature, Excerpta Medica Database and PubMed were searched for publications that addressed the diagnosis, causality assessment, and management of SJS and TEN. We also analyzed the FDA’s Adverse Event Reporting System (FAERS), which is a database designed to support the FDA’s post-marketing safety surveillance of prescription and OTC drugs program for all approved drugs and therapeutics to monitor for new adverse events that might occur with both prescription and OTC forms of APAP and ibuprofen marketed products.
- The following terms were employed in the literature search:SJS and TEN and Acetaminophen/Paracetamol, in “epidemiologic studies”, “case control”, cohort, “retrospective study”, longitudinal or “cross-sectional” studies
- SJS and TEN and for “Ibuprofen”, Advil, Motrin, Children’s Advil, Advil Liqui-Gels, Children’s Motrin, Pediacare Children’s Ibuprofen, Nurofen, Calprofen, Pediaprofen, Ibuprofen Migraine, and “propionic-acid non-steroidal drugs.”
Additionally, we use the Canadian Adverse Drug Reaction (Pharmacovigilance) data.
We describe the LTA, our precision biomarker, to diagnose the cause of SJS-TEN adverse reaction in these study patients. In vitro results correlated with the clinical diagnosis. LTA presented a significant difference (P < 0.0001) between control and hypersensitive patients. Moreover, we determine the role of the inflammasome and apoptosome in the possible repair of cellular damage involved with the SJS and TEN. Although both the inflammasome and the apoptosome in SJS-TEN are still in early phases of research, these precision biomarkers offer a scientific personalized treatment. An effective therapy for the treatment of SJS-TEN is the goal of clinicians and laboratories.
We present our own data on patients diagnosed by a clinician dermatologist and burn unit specialist, and a medical clinical biochemist. The cases presented were histologically proven, and LTA was proven to be SJS-TEN. The controls were individuals who were exposed to the same medication and did not present any adverse effects.
The controls agreed to have the analysis performed on the patient. Host-related factors can influence the drug metabolism (slow or rapid acetylation, genetics, use of alcohol that reduces glutathione, diet, drugs of misuse, and their combination).
The proinflammatory cytokines play a role in antigen-stimulating cells. As immune cells are recruited and infiltrate the surrounding area, inflammation ensues, and the altered local environment induces fibroblast activation. Affected cells generate excessive fibrinogen and other extracellular matrix proteins. Inflammation is regulated by several factors, including interleukins (IL1, IL6, IL8). In skin cells, the production of proinflammatory cytokines leads to cell death by apoptosis or by necrosis. This is the reason for measuring the death of cells in the blood from our patients and controls to see if it occurred by necrosis or by apoptosis. In addition, we measured cytokines and chemokines as part of the inflammasome. We analyzed serum apoptosis and necrosis markers to understand if the cell death was programmed (apoptosome) or random (necrosome). For cytokine determinations, we employed commercially available enzyme-linked immunoassay (ELISA) kits.
All the patients were clinically diagnosed by Dr. R.S. The skin biopsy was processed and diagnosed at the same hospital by a certified medical pathologist. Dr. MN at In Vitro Drug Safety and Biotechnology performed laboratory investigations. The laboratory results have been transmitted to the clinical team. Dr. R.S., who either treated the patients or reviewed the medical records, laboratory results, and skin biopsies from outside facilities. The literature search was performed by MMM and MN.
Results
In Table 1, we show the personal data, clinical diagnosis, and the skin lesion of the individuals who were diagnosed clinically with SJS-TEN and from medical laboratory results, presented a positive result when their lymphocytes were rechallenged to the drug that produced the HSR. All patients either had cultures, PCR, serology studies, or pathological/laboratory analysis to rule out any infectious or autoimmune diseases as the etiology of their SJS or TEN.
To determine if the patients had a hypersensitivity immune reaction to the specific drug, the polymorphic blood mononuclear cells (PBMC) of every patient and his/her control were exposed to the recommended dose per/age and body mass index. After 24 hours, we determined cell viability by measuring the mitochondrial succinate dehydrogenase (SDH) activity, which determines how many cells survived the therapeutics.
We also measured cytokines and chemokines in the sera of the patients to understand the degree of inflammatory markers in the diseased individuals. Additional markers include apoptosis and necrosis. Apoptosis, as one of the terminal paths of cell death is a programmed cell death involved in elimination of aged or damaged cells to maintain tissue homeostasis. Mitochondrial health can modulate hepatic apoptosis via the mitochondrial pathway.
The phenomenon leading to cellular death is inflammation. Inflammasome-dependent caspase-1 activation promotes cell death and the secretion of proinflammatory cytokines. loss of barrier integrity of the cell membrane, cellular stress, and cellular death. Therefore, it is important to monitor cytokine production and signaling pathways during cell inflammation and repair.
The inflammatory mediators, known as “inflammasome” are a consequence of the metabolic products (metabolome) of cells and commensal or pathogenic bacteria. In response to APAP or Ibuprofen exposure, cells express and secrete chemokines and damage-associated molecular patterns (DAMP), cellular injury, and death. The cellular responses to chronic toxicity enhance the expression of pro- and anti- inflammatory cytokines and chemokines. Moreover, these signals recruit immune cells and activate the inflammatory process [18,20,23]. Inflammation leads to cell death by apoptosis or necrosis. The process opposite to apoptosis is associated with caspase-independent cell death, which leads to necrosis and necroptosis. The dynamic equilibrium between apoptosis and necrosis in the context of disease-related pathogenesis would be essential in targeting the pathway for the treatment of SJS-TEN diseases. To follow the possible inflammatory response to the HSR, we analyzed the markers of inflammatory reactions in serum. In Table 2, we show the levels of proinflammatory cytokines in the present cases.
For the quantitative determination of TNF? in the serum of our patients, we used high sensitivity (Quantikine HS) human TNF/TNFS1A Immunoassay from R & D System. The quantitative determinations of serum VEGF total and VEGF-D we employed the Quantikine human VEGF and VEGF D, from R&D Systems. To quantify interleukins (IL-6 and IL-8) in sera, we used Quantikine human IL-6 and IL-8 from R&D Systems. The measurements of INF-g, Monokine- Induced by Gamma Interferon (MIG)/CXCL9 and RANTES are presented in the Table 3.
The serum levels of IFN-? and RANTES and MIG were measured using the ELISA Human – Quantikine IFN, MIG, and RANTES from InVitrogen, Canada. We consider normal the values of 400 healthy individuals kept in our biobank.
The Interferon and chemokines production during hypersensitivity to APAP and Ibuprofen is significantly higher when compared with the levels in controls (Table 4).
Literature Review and Discussion
A link between genetics and medications has been established. Clinical diagnosis should be confirmed with a biopsy. To prevent an additional SCAR to the individual or to his/her relatives, an LTA to the medication is advisable. The precision medication biomarker is useful to improve patients’ survival.
SJS/TEN Literature case-control studies
The SCAR Study [26] collected data that was obtained from surveillance networks in France, Germany, Italy, and Portugal. Cases included patients admitted to the hospital with a diagnosis of TEN or SJS. Cases were validated by an international group of dermatologists who viewed photographs, pathological slides, and clinical information. Controls were patients admitted to the same hospital for an acute condition. Exposure to medications was limited to the 7 days preceding the date of onset of symptoms or signs that progressed within 3 days to SJS/TEN.
The first study by Roujeau et al. [26], 1995, is a case-control study to quantify the risks associated with SCARS. The third study, by Mockenhaupt et al. [59-70], also used the EuroSCAR study population, which is an international multicenter study aimed at an ongoing surveillance of medication risks for SJS and TEN. This study specifically studied 379 ‘‘community’’ cases of SJS and TEN (i.e., patients who developed SCAR outside the hospital and were admitted to the hospital because of those symptoms) and 1,505 hospitalized controls with acute conditions not associated with chronic conditions. Controls were matched to cases on a 3:1 basis by age, gender, region, and date of interview. Information on medication exposures, clinical data, and medical history was obtained by questionnaire; the relevant exposure period was 7 days preceding the index date (date of diagnosis for cases and controls).
Outcomes were validated by dermatologists, based on clinical data, photographs, and histopathology. The authors found a significant association of APAP with risk of SCAR [adjusted RR = 1.9, 95% CI = (1.2, 2.8)]. This association did not change substantially when the analysis was restricted to patients who were not exposed to any ‘‘highly suspected’’ drugs (adjusted RR=2.4, CI = (1.5, 3.7)) or to any ‘‘suspected drug’’ (adjusted RR=1.9, CI = (0.7, 5.0), although the latter association was no longer statistically significant. However, when the authors attempted to control for what they referred to as confounding by indication, the adjusted RR decreased to less than 1.0 and was not statistically significant (adjusted RR = 0.8, CI = (0.5, 1.3).
- The paper published by Sassolas et al., [23] aimed to evaluate a specific algorithm of drug causality for epidermal necrolysis (EN), called ALDEN, to improve the individual assessment of drug causality in the two main EN diagnoses, SJS and TEN. The investigators used two case-control study populations, SCAR and EuroSCAR, for their analysis. As a first step, an algorithm was developed by a group of experts based on their knowledge of the results of the SCAR study. The algorithm was based on six parameters:time lag from initiation of drug intake to onset of reaction,
- probability that the drug was present in the body at the onset of the reaction,
- prechallenge/rechallenge,
- dechallenge,
- drug notoriety (based on the strength of evidence in previous studies) and
- other possible etiological alternatives.
A numeric scoring system was developed for the 6 parameters, with the highest scores assigned to the highest levels of likelihood. In the second step, the algorithm assessment of drug causality was carried out on all 369 validated SJS/TEN cases enrolled in the EuroSCAR study and a total score was assigned to each drug used by each patient. Scores were pooled into categories reflecting causality: “very probable”, “probable”, “possible”, “unlikely”, and “very unlikely”. Inter-observer agreement on scores was assessed using the kappa statistic. Kappa values of 0.73, 0.71, and 0.71 were determined for each medication, classification in causality groups, and determination of the drug with the highest score for each patient, respectively. Among the drugs for which association with EN was considered doubtful because of a high potential for confounding by indication, four were judged to be probably causal by use of the algorithm. These were APAP (eight exposures), pyrazolone analgesics (six exposures), corticosteroids (seven exposures), and nimesulide (one exposure), for a total of 22 of 233 exposures.
The Adverse Event Reporting System (AERS) is a computerized information database designed to support the FDA’s post-marketing safety surveillance program for all approved drug and therapeutic biologic products. The FDA uses AERS to monitor for new adverse events and medication errors that might occur with marketed products. In the following table, we summarize the data contained in the AERS system regarding cases of APAP – induced SJS and TEN. Pooled ORs and multivariate ORs were estimated using unconditional logistic regression. Multivariate ORs were adjusted for matching variables, highly suspected drugs, fever, and upper respiratory tract infection in the last week [62].
Trujillo et al. presented a previously healthy 11-year-old boy who developed general malaise, fever, and erythematous macules with target-like bulla in the centre, mainly on his left arm and right leg. A few hours later, erosive and hemorrhagic lesions appeared on the lip and spread to the oral and genital mucosa. The rest of the physical examination was normal. These findings were confirmed by a dermatologist who clinically diagnosed erythema multiforme-Stevens-Johnson syndrome. The biopsy was compatible with this disease. Three days before the onset of these symptoms, the patient had started a treatment with paracetamol for a cold [71-75].
Prick and intradermal testing, done 2 years later, was negative for paracetamol; however, an oral test for paracetamol (clinicians were unaware that the original reaction was associated with paracetamol) resulted in lesions that were diagnosed by a dermatologist as erythema multiforme. A biopsy was compatible with erythema multiforme. The authors interpret this as a case of SJS. Dragojevic - Simic et al. present a 20-year-old man who was admitted to an intensive care unit for care of TEN, involving 90% of the skin (clinical description or biopsy are not reported) 75. The man denied use of any drug other than paracetamol, which he had taken for a “cold” three days before the onset of symptoms (two 500 mg tablets) and about 7 days before admission. He did not have human immunodeficiency virus (HIV) infection and denied a history of SLE or malignant diseases. Paracetamol could not be detected by high-performance liquid chromatography (HPLC) in blood or urine. The burn unit assessed paracetamol as the causative agent. He had numerous complications in the hospital, including “struggle against infection and hyperthermia; hyperalimentation; correction of electrolytic imbalance, neutropenia, coagulation and immunological status; plasmapheresis; surgical interventions and physiotherapy.” He was discharged from the hospital 14 weeks after admission.
Verneuil et al. report two cases as part of a case series [76]. For this study of endothelial cell apoptosis, the authors examined skin biopsies from patients with drug-induced skin reactions. The article indicates that the drug involved in two cases (a 51-year-old woman and an 87-year-old man with SJS) was paracetamol; the indication was not specified. Diagnoses had been made for these patients with the aid of biopsies done 3-4 days after the eruption. The authors describe vascular changes observed in biopsies. Both patients had taken paracetamol and developed mucous lesions, erythroderma, and blisters (“>30%” for the 51-year-old woman diagnosed with TEN, “<10%” for the 87-year-old man diagnosed with SJS). The authors do not describe treatments or clinical outcomes.
Yamane et al. [77] report a case of TEN, in a 38-year-old man who took acetaminophen (the “causative drug”). He had 78% skin detachment. The patient was admitted to a hospital and treated with methylprednisolone and intravenous immunoglobulin (IVIG).
Pyo et al. describe a case of a 53-year-old woman who took NSAIDS 1300 mg orally every 8 h for 5 days. The woman had a rash with blisters beginning on her back and progressing to her extremities. After 5 days, she presented with multiple clear-filled bullae and erosion with hyperpigmentation involving 40% of body surface (TEN) and was treated with fluids, cephalosporin, corticosteroids, and wound care. At 7 days, most wounds healed [78].
Yun et al. describe two cases of TEN. Both patients, a 44-year-old and a 42-year-old man, took acetaminophen for an unstated indication. The authors studied lactate dehydrogenase (LDH) levels in patients admitted to a Dermatology department with bullous lesions. The “causative” agent for TEN (both men, one 44 years old, the other 42 years old) was “acetaminophen.” Neither patient had an underlying disease. Both were treated with systemic corticosteroids [79].
Gerdts et al. selected patients who had been admitted to a Burn Center with the diagnosis
of TEN between January 1, 1989, and January 1, 2004 [80]. Data describing patients’ physical condition at presentation, such as temperature, the amount of truly detached skin (percentage of total body surface area), and coinciding mucocutaneous lesions, were collected. In both of the following cases, the “probable causative agent” was paracetamol. A 70-year-old woman was referred to a Burn Center only a day after mucocutaneous symptoms. She had an affected BSA of 20% and a temperature > 37.5 °C. She had received “antibiotics, antihistamines”. The Burn Center treated her with “selective digestive decontamination,” enteral nutrition, and “inotropics/vasopressors.” She stayed 14 days in the Burn Center. The article exposed another case of a 34-year-old woman who was referred to the Burn Center 1 day after mucocutaneous symptoms. She had an affected BSA of 90% and a temperature > 37.5 °C. She had stomatitis, conjunctivitis, and vulvitis. No treatment at the referring hospital is stated. A wound culture grew Staphylococcus aureus. The treatment was “selective digestive decontamination,” enteral nutrition, “inotropics/vasopressors”, and IVIG. The patient stayed 12 days in the hospital. She survived.
Prins et al. selected 48 consecutive patients who had been hospitalized in Burn Centers with a diagnosis of TEN by published criteria [81]. The diagnosis was confirmed by histological analysis in 43 cases. All cases had progressive epidermal detachment and were clinically examined by a “senior dermatologist with experience in the evaluation of TEN.” The “putative culprit drug” for 3 patients was paracetamol: 1) a 24-year-old woman with 30% BSA skin detachment, 2) a 4-year-old girl with 26% BSA skin detachment, and 3) a 29-year-old man with 50% skin detachment. Each of these patients was treated with IVIG and survived.
Takeuchi et al. report the case of a 43-year-old woman who took a cephalosporin, acetaminophen, salicylamide, promethazine, methylene-diphenyl-salicylate, and caffeine anhydride. The following day, she developed edematous erythema and bullae on her trunk and limbs, erosion in the mucous membranes of her lips and conjunctiva, and high fever [82]. A biopsy specimen taken from her right forearm revealed subepidermal bulla with scattered necrotic keratinocytes in the epidermis, exocytosis of mononuclear cells, and focal hydropic degeneration. Eruption subsided rapidly and erosions healed within 25 days after administration of systemic corticosteroid therapy, leaving pigmentation. One month after the discontinuation of therapy, a patch test was performed using APAP. Papules and erythema were observed at the site at which APAP had been applied. The diagnosis was TEN due to APAP.
In a letter to the editor, Khare et al. present a case of SJS in a 22-year-old woman who was admitted to a nursing home with “high fever”, chills, and rigor [83]. She was administered paracetamol and intravenous fluids and developed oropharyngeal ulcers and a maculopapular eruption “all over the body” with progression to vesicles, some hemorrhagic. She was treated with systemic and ocular corticosteroids and antibiotics, with resolution of the skin lesions, but keratitis remained.
In their article, Szepietowski and his team describe a case of a 48-year-old man who was admitted for rash and malaise after taking four 500 mg tablets of paracetamol for a toothache [84]. Three days after the last dose, he noticed the first skin lesions and experienced pruritus. A physical examination revealed widespread erythematous rash with large, tense bullae over the trunk and extremities, involving 70% of the body surface. Nikolsky’s sign was positive, and necrosis of the epidermis was evident in some areas. Painful erosions were seen on the oral mucous membranes. The skin biopsy showed marked edema of the dermis with an infiltrate of lymphocytes and histiocytes. The features were consistent with TEN. He received treatment with systemic doxycycline, intensive local skin care, cyclosporine, and prednisone. The lesions “healed.”
Discussion
Seven probable cases in our series contained a confirmed diagnosis of SJS/TEN by a dermatologist and histological findings, which were temporally associated with acetaminophen, and occurred in the absence of other drugs labeled for these events. All but one case involved a single-ingredient acetaminophen product. The outcomes of these cases included 6 hospitalization patients and one death. The fatality was due to SJS and involved a 92-year-old male who received one dose of intravenous acetaminophen approximately 2 days before the onset of severe skin reactions.
The cases with positive histology and dermatologist-validated reports with respect to these rare, serious skin reactions, in which various dosage forms of acetaminophen were taken within short proximities of the events, in patients of various ages. These findings provide evidence that a labeling change to indicate a potential risk of severe skin reactions associated with APAP is merited.
Raksha and Marfatia present a prospective study comprising 200 cases (112 males and 88 females) of cutaneous drug reaction, which was carried out from July 1997 to June 2006 [85]. There was 1 case out of the 6 SJS cases that was caused by paracetamol. The team of Sharma et al. [86] initiated a retrospective analysis of inpatients with this dermatological diagnosis.
Sanmark et al. [87], in a retrospective study, reviewed the medical records of SJS, TEN, and SJS/TEN overlap of inpatients over a period of 10 years in their hospital. There was a total of 46 patients (21 SJS; 21 TEN; 4 SJS/TEN). Males predominated in the SJS group with a ratio of 1.63:1, whereas females predominated in the TEN group with a ratio of 1:2. Drug as an etiology was established in almost all the cases except in one case of SJS, which was due to herpes labialis. Nonsteroidal anti-inflammatory drugs (NSAIDs) were the commonest group of drugs among the SJS group in 5/21 patients (23.8%). There were two cases of TEN due to paracetamol.
Barvaliya M et al., in a tertiary care hospital, conducted a multicentric retrospective study (case series) that analyzed cases of SJS, TEN, and SJS-TEN overlap admitted between January 2006 and December 2009 in four tertiary care hospitals of Gujarat [88]. Among the 32 cases included, there were 11 cases of SJS, 15 cases of TEN, and 6 cases of SJS-TEN overlap. Nonsteroidal anti-inflammatory drugs had a 22.41% occurrence. Paracetamol was the most common cause of the reaction in 8 cases (2 SJS; 4 TEN; 2 SJS/TEN).
In a retrospective study, 59 cases of SJS have been reported at B.J. Medical College, Ahmedabad. Ibuprofen and paracetamol were prescribed to two patients. Amongst the unknown agents, most of the drugs were used for the treatment of fever and were available as over-the-counter drugs [89].
In a review of published case-control studies and case series from 1995 to 2011 describing SJS and TEN in the Indian population, NSAIDs were one of the major causative drugs at 15.93%. Paracetamol is one of the most commonly implicated drugs at 6.17% [90].
Tan and Tay performed a retrospective case series of patients with SJS or TEN from January 2004 to November 2010 treated in a general hospital [91]. There were 18 cases of SJS, seven cases of SJS/TEN overlap, and three cases of TEN, 28 cases total. NSAIDs were implicated in 14.3% of the cases. One 13-year-old female patient took paracetamol and was diagnosed with SJS. Her SCORTEN score was 0, and she stayed in the hospital for 8 days.
Raucci et al., in a case-control study, focused on the attributed drug or vaccine that caused SJS or TEN. There were 29 children with a diagnosis of SJS and 1,362 controls with neurological conditions between November 1, 1999, and October 31, 2012 [92]. Ibuprofen-induced SJS cases were observed in 3 individuals. There were 11 cases of SJS attributed to APAP. Additionally, there were 6 cases of paracetamol and antibiotic combinations implicated. Many patients who took paracetamol were female. Significantly elevated risks were estimated for paracetamol (adjusted OR: 3.2; 95% CI 1.5–6.9). An increased risk was estimated for concomitant use of antibiotics and paracetamol (adjusted OR: 5.1; 95% CI 2.0–13.2).
Guy et al. published a retrospective study that analyzed SJS and TEN cases from the French regional pharmacovigilance and registered in BNP from January 2002 to December 2011. There were 998 cases (409 TEN, 565 SJS, and 24 severe erythema bullous multiforme) concerning 900 adults and 98 children [93]. There were 107 cases of paracetamol as the suspected drug.
Ali presented cases from the FDA adverse event reporting system between 1997 and 2012. Ali, in order to see an association with APAP and SJS/TEN manifestation. A total of 1,295 reports of serious skin reactions were submitted for acetaminophen (592) and acetaminophen-containing products (703) [94]. Empirical Bayes Geometric Mean (EBGM) with 95% confidence interval (EB05-EB95) was calculated as a disproportionality measure. Safety signals with EB 05≥ 2 were considered a significant disproportional increase in event reporting at least twice times higher than expected. Serious skin event signals were detected for acetaminophen (EBGM = 5.03, EB05-EB95 = 4.68-5.40), followed by acetaminophen-containing products (EBGM = 2.93, EB05-EB95 = 2.75-3.13) [94].
Nakamura and his team reported the results of the Japanese SJS/TEN patients that were recruited to a study through the Japan Severe Adverse Reactions (JSAR) research group [95]. The SJS/TEN patients were divided into three groups: (i) patients with severe ocular surface complications (SOCs) who received Antipyretic Analgesics for relieving a common cold before the onset of SJS/TEN (20 cases); (ii) patients without SOCs who received AA for relieving a common cold before the onset of SJS/TEN (16 cases); (iii) patients with SOCs who received causative drug(s) for relieving other than a common cold (38 cases). HLA class I loci (HLA-A, C, and B) were genotyped for these patients and for healthy Japanese volunteers (n = 220). For these HLA genotypes, frequencies in the case groups were compared with those in the healthy controls. Significant association of SJS/TEN with HLA-A*02:06 was found in the group (i).
Significant association with HLA-A*33:03-C*14:03-B*44:03 haplotype in this group, which is the second most frequent haplotype in the Japanese population (5.605%). Fourteen patients of the group (i) had been administered acetaminophen, and these two HLA-type/haplotype associations were still significant in the acetaminophen-administered subgroup of the group (i). HLA-A*02:06 genotype and HLA-A*33:03-C*14:03-B*44:03 haplotype are independently associated with AA-induced SJS/TEN with SOCs. Japanese SJS/TEN patients were recruited through the participating university/hospitals and a nationwide collecting network (as the JSAR research group) from June 2006 to June 2013. A total of 197 SJS/TEN patients were recruited. The SJS/TEN patients with APAP showed a significantly higher rate (55.6%) of severe ocular symptoms than those without this drug (28.0%) (p<0.01). The patients receiving NSAIDs for treatment of cold developed severe ocular symptoms at the rate of 65.4%, while that in the patients administered NSAIDs for other diseases was 17.1%. APAP or NSAIDs for treatment of cold showed high rates for developing severe ocular complications [95].
Ueta and his group investigated the association between HLA genotypes and cold medicine-related SJS/TEN (CM-SJS/TEN), including acetaminophen-related SJS/TEN (AR-SJS/TEN), with severe mucosal involvement, such as severe ocular surface complications (SOC), in Japanese patients. Japanese SJS/TEN patients (n = 236) were recruited at Kyoto Prefectural University of Medicine (Group 1, n = 162) and by the Japan Severe Adverse Reactions Research Group, mainly conducted by the National Institute of Health Sciences (Group 2, n = 74). Of the 162 patients in Group 1, 131 patients had taken cold medicines such as NSAIDs and multi-ingredient cold medications for a few, several days before disease onset for common-cold symptoms. HLA-A*02:06 is rarely a haplotype with HLA-B*44:03 [96]. In all 73 patients with SJS/TEN with SOCs, there was a significant association with both alleles (HLA-A*02:06, p = 2.5 x 10-15, OR = 6.5; HLA-B*44:03, p = 0.0059, OR = 2.2).
A review of previous studies conducted by Ueta summarized the information included in the articles mentioned above and is restated in this ibuprofen review. The additional information not included: HLA-A*02:06 with TLR3 polymorphisms and HLA-A*02:06 with EP3 polymorphisms exerted additive effects in SJS/TEN with SOCs. EP3 is strongly downregulated in the conjunctival epithelium of SJS/TEN. Cold medicines and infectious agents such as viruses or other microbes are both important in triggering the onset of SJS/TEN with SOCs [96,97].
Kanikawa reported that 197 Japanese patients with SJS/TEN (97 females, mean age 56.6 ± 22.3 years) were enrolled. The number of probable SJS, SJS, and TEN cases was 23, 115, and 59, respectively. The frequency of severe ocular surface involvement tended to be higher among females [98]. Patients with SJS/TEN associated with APAP showed a significantly higher rate (55.6%) of experiencing severe ocular surface involvement than those not treated with APAP (27.6%) (p < 0.01). In the SJS group, the rate of APAP-associated severe ocular involvement (7/36 patients, 19.4%) was significantly higher than that of mild involvement (4/79, 5.1%) (p = 0.034), while there was no significant association in the case of the TEN group, although the skin reaction of TEN is more severe than that of SJS. There were 14 of 15 patients with APAP -associated SJS/TEN and severe ocular surface involvement who had not taken other medication [99].
Wakamatsu et al. described in a Brazilian population that HLA class I genes are associated with SJS, leading to severe ocular complications following use of cold medicine [100]. In this case-control study, among 39 patients with cold medicine–associated SJS-TEN and severe ocular complications of 74 SJS-TEN, HLA-A*66:01, HLA-B*44:03, and HLA-C*12:03 were associated with and HLA-A*11:01, HLA-B*08:01, and HLA-B*51:01 were inversely associated with SJS-TEN without severe ocular complications. HLA B*44:03 and HLA-C*12:03 were associated only among individuals with European ancestry. Thirty-nine patients with CM-SJS/TEN of 74 patients with SJS/TEN with SOCs and 133 healthy volunteers were enrolled. Cold medicines were the largest used drug group before the onset of SJS (53%, n = 39). Out of the 39 individuals, 4 had used APAP.
Kaniwa et al, in a single-center retrospective review of pediatric TEN cases admitted to the hospital’s burn intensive care unit, identified 41 patients ≤18 years of age with a diagnosis of TEN confirmed by biopsy. The patients in this study developed an average of 39.7% ± 26 TBSA sloughing, and nearly all (95%) experienced mucosal involvement. Over-the-counter analgesics were used in 17% of cases, and 7 had APAP as the implicated drug. While acute mortality was 0%, 100% of patients experienced complications, and 30% required follow-up procedures [101].
Harimoto et al. published a retrospective study of 30 patients diagnosed with SJS and/or TEN [102]. One of the causative drugs was APAP (10%, 3 cases). Liver damage was seen in 63.3 % of the patients.
Patients with SJS and TEN treated in Dr. Hasan Sadikin General Hospital, Indonesia, were incorporated in a retrospective study. A total of 57 patients were enrolled in the study. Thirty-nine cases of SJS (21 males and 18 females), 7 cases of SJS overlapping TEN (4 males and 3 females), and 11 cases of TEN (5 males and 6 females). All cases of SJS and TEN were caused by drugs, such as paracetamol (16.56%) and ibuprofen (4.46%). Liver involvement was the most common complication [103].
Hirapara et al. analyzed in a cross-sectional study conducted in Gauhati Medical College and Hospital, Northeast India [104]. World Health Organization – and Uppsala Monitoring Center probability assessment system and ALDEN were used for causality assessment of reported SJS/TEN cases. Forty-five cases of drug-induced SJS (42.22%), TEN (55.55%) & SJS-TEN (2.22%) overlap were reported. One of the most offending drugs was antipyretics (17.78%) and NSAIDs (6.67%). In the individual drug category, paracetamol (17.77%, 4 SJS, 4 TEN) was among the commonly reported offending drugs [104].
Byun and his group present a case report of a 24-year-old female patient who presented to the Obstetrics and Gynecology Department (Busan Paik Hospital, Busan, Korea) with dysuria, vulvar irritation, and labial adhesion [105]. The patient had a history of SJS secondary to the administration of medication prescribed to relieve cold symptoms. The patient reported that the lesions appeared within a few hours of taking the medication, which contained acetaminophen. One month later, the patient was complaining of urinary difficulty and vulvar irritation. On vulvovaginal examination, labial adhesion was observed. The vaginal introitus was very narrow due to the labial adhesion observed in the first visual inspection of the vulva.
In Thailand, Paipool and Sriboonnark review that included 38 patients. They consisted of 31 (82%) SJS patients and 7 (18%) TEN patients [106]. Most cases (79%) were caused by drug exposure. One case had ibuprofen, and another case had paracetamol.
Records were collected from the Lombardy Registry of Severe Cutaneous Reactions (REACT) from April 2009 to November 2014. There were 17 cases of TEN and 59 cases of SJS. Overall, 55.4% of cases had a probable or very probable relation with drug exposure. A total of three probable and very probable cases of SJS/TEN were attributed to paracetamol (0.29 (0.10-0.86); 95% CI) and one case to paracetamol/codeine (0.21 (0.04-1.20); 95% CI). In the authors’ opinion, people should be aware of the possible association between paracetamol exposure and SJS/TEN and should use the drug carefully [107,108]. Abe et al. [109] performed a study to determine the relationship between aging and SJS/TEN using the Japanese Adverse Drug Event Report (JADER). Reports were collected between 2004 and 2015 using an adjusted reporting odds ratio (ROR). More than half of the cases of SJS/TEN onset following administration of loxoprofen and APAP occurred within 4 days of the initiation of treatment. The median times-to-onset were 3 days for loxoprofen and 2 days for APAP. There were 498 reported cases of APAP-induced SJS or TEN.
Ban et al. report a case series of six patients diagnosed with SJS and TEN after being prescribed APAP for suspected viral illnesses [110]. All of the five patients with TEN showed monocytosis or marked neutropenia within 1 week after admission.
The report of Schotland et al. [111] describes 456 ADR mentioning APAP-induced SJS and 397 ADR reports mentioning APAP-induced TEN. 939 reports mention APAP and SCARS over a study period of slightly more than 7 years (2004–2011). The authors believe that there was a significant association between APAP and TEN, except for possible concomitant administration of anti-epileptic drugs.
Hirapara shows cases of SJS, SJS–TEN overlaps, and TEN were analyzed for causative drugs, incubation period, a severity-of-illness score for toxic epidermal necrolysis (SCORTEN) score, HIV status, treatment, and outcome. A comparison of parameters between HIV and non-HIV cases was made. Utilization patterns of corticosteroids and their role in outcome were evaluated. Four SJS, 15 SJS-TEN overlap, and 21 TEN cases were evaluated. Paracetamol had 4 cases (1 SJS/TEN overlap; 3 TEN),8.3% of cases [112].
Maggio and his team, working in 4 Israeli Medical centers: Sheba (Tel Hashomer), Tel Aviv, Hadassah Jerusalem, Soroka Beer Sheva, and Assaf Harofeh Tzrifin, searched their databases for SJS- and TEN-related hospitalizations in the years 2005-2015 using the International Classification of Diseases (ICD)-9 codes for SJS, SJS-TEN overlap, and TEN (695.13, 695.14, and 695.15, respectively) [113]. All the drugs listed in the patients’ files were evaluated based on the Algorithm of Drug Causality for Epidermal Necrolysis (ALDEN). Only 26 cases in which any relationship to drug use was “probable” or “very probable” were included in the final analysis. None of the patients was reported as HIV positive. Elevated serum alanine transaminase (ALT) and aspartate transaminase (AST) levels were found in 6 patients (mean 2.1- and 2.6-fold increase compared to reference values, respectively). Renal function was impaired in 4 patients (serum creatinine levels = 1.5, 1.7, 3.7, and 3.8 mg/dL). There was only one case where paracetamol was the causative drug [113].
Rahima et al. analyze all patients diagnosed with SJS and TEN admitted to the Department of Dermatology, Government Medical College in India during two years. There were 31 SJS, 3 had SJS-TEN overlap, and 16 had TEN. There were 6 cases of paracetamol as the causative drug, around 12% of cases [114].
Abdulah and his team described a retrospective study that included 101 patients diagnosed with drug-induced SJS (n = 70), SJS–TEN overlap (n = 16), and TEN (n = 15), from 2009 to 2013 in a referral hospital in West Java Province, Indonesia. Analgesic–antipyretic drugs, including acetaminophen) were reported most frequently, and the major causative drug was at 13.40% [115-122].
Conclusions
Recent studies have reported an increasing incidence of SJS and TEN worldwide. OTC drugs, including as more frequent causes of SJS and TEN, especially in the U.S. Ibuprofen and acetaminophen new data strengthen the causal relationship. Better warnings and patient risk communications on drug labels regarding the increased risks of SJS and TEN associated with ibuprofen and acetaminophen, to at-risk populations, including females and children, are necessary to help to mitigate the incidence and risks of SJS and TEN from these OTC drugs.
Our findings, which used in vitro data, support the assessment of causality using both clinical and pathological data, demonstrating the immune mechanisms of ibuprofen and acetaminophen-induced SJS and TEN in these patients. By using a combination of in vitro analysis using the LTA, serum levels of TNF, Interleukin (IL 6, IL 8), VEGF, IFN-?, RANTES, and MIG, clinical and pathological data in these patients with ibuprofen or acetaminophen-induced SJS and TEN, we provided a robust and accurate assessment for SJS and TEN in our patients.
The use of these in vitro studies, or a combination of these studies and cytokine profiles, can help clinicians and researchers better elucidate the mechanisms of drug-induced SJS and TEN and lead to the identification of at-risk populations and provide personalized medicine. The combination of using clinical data, in vitro data and pathological data can help use new strategies for predicting the risks for SJS and TEN and managing treatment-related SCAR, alongside implementing effective clinical decisions and appropriately communicating, educating, and counseling patients and their families about the risks of SJS and TEN both before, and after the development of SJS and TEN to support medication safety and personalized care.
Clinicians should also be made aware that ibuprofen or APAP can and does cause SJS/TEN. Clinics need to be informed of the higher risks of SJS and TEN from these two OTC drugs. Stronger warnings and risk communications to clinicians that prompt recognition and withdrawal of the culprit drug, along with supportive care, are of utmost importance. Clinicians and laboratory medicine biochemists, or clinical toxicologists, should work together to recognize and prevent drug-adverse reactions. Advances in high-content screening, personalized technologies, such as LTA, the discovery of new proinflammatory biomarkers, and rapid developments in bioinformatics have created new opportunities for cell-based individualization of existing drugs.
Ethical approval statement
All procedures performed in the study were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments. The clinician consulting and diagnosing the patients is Dr. RS. The blood samples of the patients were sent to Dr. MN. All the patients have been treated in the Burn Unit by Dr. RS. All of them agreed to have the LTA and cytokines performed for diagnostic purposes. The analysis was required for diagnosis and treatment, and the patients gave informed consent. They agreed to the procedure.
Moreover, the patients agreed to share their clinical and laboratory data in this article. The data that support the findings of this study are available on request from the corresponding author.
Funding
The author declares that no funds, grants, or other support were received during the preparation of this manuscript. The LTA was performed by In Vitro Drug Safety and Biotechnology Inc., at the request of the clinician and paid by his group. The cytokines, chemokines, apoptosis and necrosis markers were performed by In Vitro Drug Safety and Biotechnology Inc. using the company funds.
Conflict of interest disclosure
The author has no relevant financial or non-financial interests to disclose. All the clinical laboratory analysis and the interpretation belong to Dr. MN. MMM performed literature search.
- Hsu DY, Brieva J, Silverberg NB, et al. Morbidity and mortality of Stevens-Johnson syndrome and toxic epidermal necrolysis in United States adults. J Invest Dermatol. 2016;136:1387–1397. Available from: https://doi.org/10.1016/j.jid.2016.03.023
- Duplisea M, Williams F, Ziemer C, et al. Ten-year mortality trends in Stevens-Johnson syndrome and toxic epidermal necrolysis: single burn center experience. 2020 Mar 17;S:116. Available from: https://doi.org/10.1111/bjd.18360
- Neuman M, Salisbury R. Severe cutaneous adverse reactions to ibuprofen: medical and laboratory causality assessment. Presented at: 40th Anniversary Meeting of the Israel Society of Dermatology & Venereology; 2019 May 15–17; Eilat, Israel.
- Tartarone A, Lerose R. Stevens-Johnson syndrome and toxic epidermal necrolysis: what do we know? Ther Drug Monit. 2010;32(6):669–672. doi:10.1097/FTD.0b013e3181f2f24f. Available from: https://doi.org/10.1097/ftd.0b013e3181f2f24f
- Sekula P, Dunant A, Mockenhaupt M, et al. Comprehensive survival analysis of a cohort of patients with Stevens-Johnson syndrome and toxic epidermal necrolysis. J Invest Dermatol. 2013;133:1197–1204. . Available from: https://doi.org/10.1038/jid.2012.510 .
- Hsu DY, Brieva J, Silverberg NB, et al. Pediatric Stevens-Johnson syndrome and toxic epidermal necrolysis.
- Okubo Y, Nochioka K, Testa MA. Nationwide survey of Stevens-Johnson syndrome and toxic epidermal necrolysis in children in the United States. Pediatr Dermatol. 2017;34:206–208. Available from: https://doi.org/10.1111/pde.13050 .
- Moreau JF, Watson RS, Hartman ME, et al. Epidemiology of ophthalmologic disease associated with erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis in hospitalized children in the United States. Pediatr Dermatol. 2014;31:163–168.Available from: https://doi.org/10.1111/pde.12158
- Frey N, Jossi J, Bodmer M, et al. The epidemiology of Stevens-Johnson syndrome and toxic epidermal necrolysis in the UK. J Invest Dermatol. 2017;137:1240–1247. Available from: https://doi.org/10.1111/pde.1215810.1016/j.jid.2017.01.031 .
- Le HG, Saeed H, Mantagos IS, et al. Burn unit care of Stevens-Johnson syndrome/toxic epidermal necrolysis: a survey. Burns. 2016;42:830–835. Available from: https://doi.org/10.1016/j.burns.2015.12.001
- Dodiuk-Gad RP, Olteanu C, Jeschke MG, et al. Treatment of toxic epidermal necrolysis in North America. J Am Acad Dermatol. 2015;73:876–877.e2. Available from: https://doi.org/10.1016/j.jaad.2015.08.008
- Mayorga C, Ebo DG, Lang DM, et al. Controversies in drug allergy: in vitro testing. J Allergy Clin Immunol. 2019;143:56–65. Available from: https://doi.org/10.1016/j.jaci.2018.09.022
- Neuman MG, Malkiewicz IM, Phillips EJ, et al. Monitoring adverse drug reactions to sulfonamide antibiotics in HIV-infected individuals. Ther Drug Monit. 2002;24(6):728–736. Available from: https://doi.org/10.1097/00007691-200212000-00008 .
- Neuman MG, Malkiewicz IM, Shear NH. A novel lymphocyte toxicity assay to assess drug hypersensitivity syndromes. Clin Biochem. 2000;33:517–524. Available from: https://doi.org/10.1016/S0009-9120(00)00146-6
- Dwivedi R, Gogtay N, Kharkar V, et al. In vitro lymphocyte toxicity to a phenytoin metabolite in phenytoin-induced cutaneous adverse drug eruptions. Indian J Dermatol Venereol Leprol. 2004;70:217–220. Available from: https://pubmed.ncbi.nlm.nih.gov/17642618/
- Neuman MG, Cohen L, Nanau RM, Hwang PA. Genetic and immune predictors for hypersensitivity syndrome to antiepileptic drugs. Transl Res. 2012;159(5):397–406. Available from: https://doi.org/10.1016/S0009-9120(00)00146-610.1016/j.trsl.2012.01.004
- Neuman M, Nicar M. Apoptosis in ibuprofen-induced Stevens-Johnson syndrome. Transl Res. 2007;149:254–259. Available from: https://doi.org/10.1016/j.trsl.2006.12.005
- Neuman MG, Cohen LB, Nanau RM. Quinolones-induced hypersensitivity reactions. Clin Biochem. 2015;48:716–739. Available from: https://doi.org/10.1016/j.clinbiochem.2015.04.006
- Neuman MG, Shear NH, Malkiewicz IM, et al. Predicting possible zonisamide hypersensitivity syndrome. Exp Dermatol. 2008;17:1045–1051. Available from: https://doi.org/10.1111/j.1600-0625.2008.00748.
- Neuman MG, Shear NH, Malkiewicz IM, et al. Immunopathogenesis of hypersensitivity syndrome reactions to sulfonamides. Transl Res. 2007;149:243–253. Available from: https://doi.org/10.1016/j.trsl.2006.12.001
- Fu M, Gao Y, Pan Y, et al. Recovered patients with Stevens-Johnson syndrome and toxic epidermal necrolysis maintain long-lived IFN-γ and sFasL memory response. PLoS One. 2012;7:e45516. Available from: https://doi.org/10.1371/journal.pone.0045516
- Porebski G, Pecaric-Petkovic T, Groux-Keller M, et al. In vitro drug causality assessment in Stevens-Johnson syndrome: alternatives for lymphocyte transformation test. Clin Exp Allergy. 2013;43:1027–1037. Available from: https://doi.org/10.1111/cea.12145
- Sassolas B, Haddad C, Mockenhaupt M, et al. ALDEN, an algorithm for assessment of drug causality in Stevens-Johnson syndrome and toxic epidermal necrolysis: comparison with case-control analysis. Clin Pharmacol Ther. 2010;88(1):60–68. Available from: https://doi.org/10.1038/clpt.2009.252
- Bellón T, Rodríguez-Martín S, Cabañas R, et al. Assessment of drug causality in Stevens-Johnson syndrome/toxic epidermal necrolysis: concordance between lymphocyte transformation test and ALDEN. Allergy. 2020;75:956–959. Available from: https://doi.org/10.1111/all.14062
- Levi N, Bastuji-Garin S, Mockenhaupt M, et al. Medications as risk factors of Stevens-Johnson syndrome and toxic epidermal necrolysis in children: pooled analysis. Pediatrics. 2009;123:e297–e304. Available from: https://doi.org/10.1542/peds.2008-1923
- Roujeau JC, Kelly JP, Naldi L, et al. Medication use and risk of Stevens-Johnson syndrome or toxic epidermal necrolysis. N Engl J Med. 1995;333:1600–1607. Available from: https://doi.org/10.1056/NEJM199512143332404
- Frantz R, Huang S, Are A, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: review of diagnosis and management. Medicina (Kaunas). 2021;57:895. Available from: https://doi.org/10.3390/medicina57090895
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390:1996–2011. Available from: https://doi.org/10.1016/S0140-6736(16)30378-6
- Dibek Misirlioglu E, Guvenir H, Bahceci S, et al. Severe cutaneous adverse drug reactions in pediatric patients: multicenter study. J Allergy Clin Immunol Pract. 2017;5:757–763. Available from: https://doi.org/10.1016/j.jaip.2017.02.013
- Spillers NJ, Luther PM, Talbot NC, et al. Association of acetaminophen with Stevens-Johnson syndrome and toxic epidermal necrolysis: pharmacologic considerations and treatment options. Cureus. 2023;15:e41116.Available from: https://doi.org/10.7759/cureus.41116
- Cartotto R. Burn center care of patients with Stevens-Johnson syndrome and toxic epidermal necrolysis. Clin Plast Surg. 2017;44:583–595. Available from: https://doi.org/10.1016/j.cps.2017.02.016
- Cartotto R, Johnson LS, Savetamal A, et al. American Burn Association clinical practice guidelines on burn shock resuscitation. J Burn Care Res. 2024;45:565–589. Available from: https://doi.org/10.1093/jbcr/irad125
- ISBI Practice Guidelines Committee, et al. ISBI practice guidelines for burn care, part 2. Burns. 2018;44:1617–1706. Available from: https://doi.org/10.1016/j.burns.2018.09.012
- Greenhalgh DG, Hill DM, Burmeister DM, et al. Surviving sepsis after burn campaign. Burns. 2023;49:1487–1524. Available from: https://doi.org/10.1016/j.burns.2023.05.003
- Wolf R, Wolf D, Davidovici B. Classifying severe cutaneous adverse reactions. Clin Dermatol. 2007;25:348–349. Available from: https://doi.org/10.1016/j.clindermatol.2007.01.001
- Kantor TG. Ibuprofen. Ann Intern Med. 1979;91:877–882. Available from: https://doi.org/10.7326/0003-4819-91-6-877
- Rumack BH, Matthew H. Acetaminophen poisoning and toxicity. Pediatrics. 1975;55:871–876. Available from: https://pubmed.ncbi.nlm.nih.gov/1134886/
- Jan-Roblero J, Cruz-Maya JA. Ibuprofen: toxicology and biodegradation. Molecules. 2023;28:2097. Available from: https://doi.org/10.3390/molecules28052097
- Murdoch RW, Hay AG. Biotransformation of ibuprofen in activated sludge. Biodegradation. 2015;26:105–113 . Available from: https://doi.org/10.1007/s10532-015-9719-4
- Cao F, Zhang M, Yuan S, et al. Transformation of acetaminophen during water chlorination. Environ Sci Pollut Res. 2016;23:12303–12311. Available from: https://doi.org/10.1007/s11356-016-6341-x
- Browne GS, Nelson C, Nguyen T, et al. NSAIDs inhibition of mitochondrial oxidation. Biochem Pharmacol. 1999;57:837–844.
- Bédouet L, Pascale F, Bonneau M, et al. In vitro evaluation of ibuprofen toxicity on joint cells. Toxicol In Vitro. 2011;25:1944–1952. Available from: https://doi.org/10.1016/j.tiv.2011.06.018
- Gliszczynska A, Sánchez-López E. Dexibuprofen therapeutic advances. Pharmaceutics. 2021;13:414. Available from: https://doi.org/10.3390/pharmaceutics13030414
- Żur J, Piński A, Marchlewicz A, et al. Paracetamol and ibuprofen toxicity and biodegradation. Environ Sci Pollut Res Int. 2018;25:21498–21524. Available from: https://doi.org/10.1007/s11356-018-2517-x
- Angadi SS, Karn A. Ibuprofen-induced Stevens-Johnson syndrome/toxic epidermal necrolysis. Asia Pac Allergy. 2016. Available from: https://doi.org/10.5415/apallergy.2016.6.1.70
- Kim HY, Yang HK, Kim SH, Park JH. Ibuprofen-associated TEN in infant. Yonsei Med J. 2014;55:834–837. doi:10.3349/ymj.2014.55.3.834. Available from: https://doi.org/10.5415/apallergy.2016.6.1.70
- Balint B, Stepic N, Todorovic M, et al. Ibuprofen-induced TEN: case report. Blood Transfus. 2014;12:438–439. Available from: https://doi.org/10.5415/apallergy.2016.6.1.7010.2450/2014.0065-14
- Zyryanov S, Asetskaya I, Butranova O, et al. Stevens-Johnson syndrome database analysis. Pharmaceuticals (Basel). 2024;17:675. Available from: https://doi.org/10.3390/ph17060675
- Brockow K, Wurpts G, Trautmann A, et al. Guideline for allergological diagnosis of drug hypersensitivity reactions. Allergol Select. 2023;7:122–139. Available from: https://doi.org/10.5414/ALX02422E
- Atanasković-Marković M, Medjo B, Gavrović-Jankulović M, et al. Stevens-Johnson syndrome in children. Pediatr Allergy Immunol. 2013;24:645–649. Available from: https://doi.org/10.1111/pai.12121
- Suwarsa O, Yuwita W, Dharmadji HP, et al. Stevens-Johnson syndrome in Indonesia. Asia Pac Allergy. 2016;6:43–47. Available from: https://doi.org/10.5415/apallergy.2016.6.1.43
- Middendorf MM, Busaileh AZ, Babakhani A, et al. TEN treatment case report. BMJ Case Rep. 2019;12:e230538.
- Kheir K, Abdallah RM, Sleiman Z, et al. Case report. Cureus. 2023;15:e49608.
- Szentes V, Gazdag M, Szokodi I, Dézsi CA. CXCR3 chemokines in disease. Front Immunol. 2018;9:1932.
- Nanau RM, Neuman MG. Ibuprofen-induced hypersensitivity syndrome. Transl Res. 2010;155:275–293. Available from: https://doi.org/10.1016/j.trsl.2010.01.005
- Evans AM. Comparative pharmacology of ibuprofen. Clin Rheumatol. 2001;20(Suppl 1):S9–S14. Available from: https://doi.org/10.1007/bf03342662
- Karaźniewicz-Łada M, Luczak M, Główka F. Pharmacokinetics of ibuprofen enantiomers. Xenobiotica. 2009;39:476–485. Available from: https://doi.org/10.1023/a:1015811428066
- Williams K, Day R, Knihinicki R, Duffield A. Uptake of ibuprofen enantiomers. Biochem Pharmacol. 1986;35:3403–3405. Available from: https://doi.org/10.1016/0006-2952(86)90443-0
- López-Rodríguez R, Novalbos J, Gallego-Sandín S, et al. CYP polymorphisms and ibuprofen PK. Pharmacol Res. 2008;58:77–84. Available from: https://doi.org/10.1007/s00228-020-03046-0
- Hamman MA, Thompson GA, Hall SD. Metabolism of ibuprofen. Biochem Pharmacol. 1997;54:33–4. Available from: https://doi.org/10.3109/00498257309151547
- Kirchheiner J, Meineke I, Freytag G, et al. CYP2C9 variants and ibuprofen. Clin Pharmacol Ther. 2002;72:62–75. Available from: https://doi.org/10.1067/mcp.2002.125726
- Martínez C, García-Martín E, Blanco G, et al. CYP2C8 polymorphism and ibuprofen. Br J Clin Pharmacol. 2004;59:62–68. Available from: https://doi.org/10.1111/j.1365-2125.2004.02183.x
- Pilotto A, Seripa D, Franceschi M, et al. NSAID-related bleeding and CYP2C9. Gastroenterology. 2007;133:465–471. Available from: https://doi.org/10.1053/j.gastro.2007.10.015
- Evans AM. Pharmacodynamics of profens. Clin Pharmacokinet. 1996;36:S7–S15. Available from: https://pubmed.ncbi.nlm.nih.gov/9013378/
- Evans AM. Enantioselective NSAID pharmacology. Eur J Clin Pharmacol. 1992;42:237–256.
- Rudy AC, Knight PM, Brater DC, Hall SD. Ibuprofen metabolism. J Pharmacol Exp Ther. 1991;259:1133–1139.
- Bygum A, Gregersen JW, Buus S. Acetaminophen-induced TEN. Pediatr Dermatol. 2004;21:236–238. Available from: https://doi.org/10.1111/j.0736-8046.2004.21309.x
- Halevi A, Ben-Amitai D, Garty B. TEN with acetaminophen. Ann Pharmacother. 2000;34:21–24. Available from: https://doi.org/10.1345/aph.19064
- Castellana E, Chiappetta MR, et al. Database report. Hosp Pharm. 2025. Available from: https://doi.org/10.1136/ejhpharm-2025-004663
- Mockenhaupt M, Viboud C, Dunant A, et al. EuroSCAR study on SJS/TEN drug risks. J Invest Dermatol. 2008;128:35–44. Available from: https://doi.org/10.1038/sj.jid.5701033
- Sidoroff A, Dunant A, Viboud C, Halevy S, Bouwes Bavinck JN, Naldi L, Mockenhaupt M, Fagot JP, Roujeau JC. Risk factors for acute generalized exanthematous pustulosis (AGEP): results of a multinational case-control study (EuroSCAR). Br J Dermatol. 2007;157:989–996. Available from: https://doi.org/10.1111/j.1365-2133.2007.08156.
- Gau SS, Chao PF, Lin YJ, et al. Association between carbamazepine and valproate and adverse cutaneous drug reactions in patients with bipolar disorder: a nested case-control study. J Clin Psychopharmacol. 2008;28:509–517. Available from: https://doi.org/10.1097/jcp.0b013e3181845610
- Reese D, Henning JS, Rockers K, et al. Cyclosporine for Stevens-Johnson syndrome/toxic epidermal necrolysis: case series and literature review. Cutis. 2011;87:24–29. Available from: https://pubmed.ncbi.nlm.nih.gov/21323097/
- Trujillo C, Gago C, Ramos S. Stevens-Johnson syndrome after acetaminophen ingestion confirmed by challenge test in an 11-year-old patient. Allergol Immunopathol (Madr). 2010;38:99–100. Available from: https://doi.org/10.1016/j.aller.2009.06.009
- Dragojevic-Simic V, Vukovic J, Kovacevic A. Paracetamol-induced toxic epidermal necrolysis in adults: case report. Basic Clin Pharmacol Toxicol. 2009;105(Suppl 1):98.
- Verneuil L, Ratajczak P, Allabert C, et al. Endothelial cell apoptosis in severe drug-induced bullous eruptions. Br J Dermatol. 2009;161:1371–1375. Available from: https://doi.org/10.1111/j.1365-2133.2009.09357.x
- Yamane Y, Aihara M, Tatewaki S, et al. Analysis of treatments and fatal cases of severe adverse drug reactions: study of 46 SJS/TEN cases. Arerugi. 2009;58:537–547. Available from: https://pubmed.ncbi.nlm.nih.gov/19487835/
- Pyo KS, Heung YD, Jung KS, et al. Toxic epidermal necrolysis induced by NSAIDs: case report. Toxicol Lett. 2008;180(Suppl 5):S140. Available from: https://doi.org/10.1007/s00393-003-0535-6
- Yun SJ, Choi MS, Piao MS, et al. Serum lactate dehydrogenase as a marker of disease severity in toxic epidermal necrolysis. Dermatology. 2008;217:254–259. Available from: https://doi.org/10.1159/000148255
- Gerdts B, Vloemans AF, Kreis RW. Toxic epidermal necrolysis: 15 years’ experience in a Dutch burns centre. J Eur Acad Dermatol Venereol. 2007;21:781–788. Available from: https://doi.org/10.1111/j.1468-3083.2006.02082
- Prins C, Kerdel FA, Padilla RS, Hunziker T, Chimenti S, Viard I, et al. Treatment of toxic epidermal necrolysis with high-dose intravenous immunoglobulins: multicenter retrospective analysis. Arch Dermatol. 2003;139:26–32 https://doi.org/10.1001/archderm.139.1.26
- Takeuchi Y, Katagiri K, Hatano Y, et al. Toxic epidermal necrolysis due to acetaminophen: case report. J Dermatol. 2004;31:1–10
- Khare KC, Khare S, Mathew G. Stevens-Johnson syndrome with keratitis following paracetamol injection. Indian J Dermatol Venereol Leprol. 1997;63:209
- Szepietowski J, Wasik F, Szybejko-Machaj G, et al. Toxic epidermal necrolysis treated with cyclosporine: report of three cases. J Eur Acad Dermatol Venereol. 1997;9:169–172
- Raksha MP, Marfatia YS. Clinical study of cutaneous drug eruptions in 200 patients. Indian J Dermatol Venereol Leprol. 2008;74:80 https://doi.org/10.4103/0378-6323.42883
- Sharma VK, Sethuraman G, Minz A. Stevens-Johnson syndrome, toxic epidermal necrolysis and overlap: retrospective study. Indian J Dermatol Venereol Leprol. 2008;74:238–240 https://doi.org/10.4103/0378-6323.41369
- Sanmarkan AD, Sori T, Thappa DM, Jaisankar TJ . Retrospective analysis of Stevens-Johnson syndrome and toxic epidermal necrolysis over 10 years. Indian J Dermatol. 2011;56:25–29 https://doi.org/10.4103/0019-5154.77546
- Barvaliya M, Sanmukhani J, Patel T, Paliwal N, Shah H, Tripathi C.. Drug-induced Stevens-Johnson syndrome and toxic epidermal necrolysis: multicentric retrospective study. J Postgrad Med. 2011;57:115–119 https://doi.org/10.4103/0022-3859.81865
- Patel PP, Gandhi AM, Desai CK, Desai MK, Dikshit RK. Analysis of drug-induced Stevens-Johnson syndrome. Indian J Med Sci. 2012;136:1051–1053 https://pmc.ncbi.nlm.nih.gov/articles/PMC3612312/
- Patel TK, Barvaliya MJ, Sharma D, Tripathi C. A . Systematic review of drug-induced Stevens-Johnson syndrome and TEN in Indian population. Indian J Dermatol Venereol Leprol. 2013;79:389–398 https://doi.org/10.4103/0378-6323.110749
- Tan SK, Tay YK. Profile of Stevens-Johnson syndrome and toxic epidermal necrolysis in Singapore. Acta Derm Venereol. 2012;92:62–66 https://doi.org/10.2340/00015555-1169
- Raucci U, Rossi R, Da Cas R, Rafaniello C, Mores N, Bersani G, et al. Stevens-Johnson syndrome associated with drugs and vaccines in children: case-control study. PLoS One. 2013;8:e68231 https://doi.org/10.1371/journal.pone.0068231
- Guy C, Lebrun-Vignes B, Jean-Pastor MJ. Drug-induced Stevens-Johnson syndrome and TEN: analysis of French pharmacovigilance database. 2014
- Ali AK. Acetaminophen and serious skin reactions. Value Health. 2014;17:A283 https://doi.org/10.1016/j.jval.2014.03.1648
- Nakamura R, Kaniwa N, Ueta M, Sotozono C, Sugiyama E, Maekawa K, et al. HLA association with analgesic-induced SJS/TEN. EAACI Drug Hypersensitivity Meeting; 2014 https://doi.org/10.1186/2045-7022-4-S3-P89
- Saito Y, Ueta M, Nakamura R, et al. Medication trends causing severe ocular complications in SJS/TEN. EAACI Meeting; 2014
- Ueta M, Kaniwa N, Sotozono C, et al. HLA associations in SJS/TEN with ocular complications. Sci Rep. 2014;4:4862
- Ueta M, Kannabiran C, Wakamatsu TH, Kim MK, Yoon KC, Seo KY, et al. Trans-ethnic HLA associations in SJS/TEN. Sci Rep. 2014;4:5981 https://doi.org/10.1038/srep05981
- Ueta M. Genetic predisposition to Stevens-Johnson syndrome with ocular complications. Cornea. 2015;34(Suppl 11):S158–S165 https://doi.org/10.1097/ico.0000000000000605
- Wakamatsu TH, Ueta M, Tokunaga K, , Okada Y, Loureiro RR, Costa KA et al. HLA genes and SJS in Brazilian population. JAMA Ophthalmol. 2017;135:355–360 https://doi.org/10.1001/jamaophthalmol.2017.0074
- Kaniwa N, Ueta M, Nakamura R, Okamoto-Uchida Y, Sugiyama E, Maekawa K, et al. Drugs causing ocular complications in SJS/TEN. Allergol Int. 2015;64:379–381 https://doi.org/10.1016/j.alit.2015.05.002
- Harimoto N, Wang H, Ikegami T, et al. Acetaminophen-induced SJS with liver failure. J Gastroenterol Hepatol. 2015;30:656
- Suwarsa O, Yuwita W, Dharmadji HP, Sutedja E. Stevens-Johnson syndrome in Indonesia. Asia Pac Allergy. 2016;6:43–47 https://doi.org/10.5415/apallergy.2016.6.1.43
- Hirapara HN, Patel TK, Barvaliya MJ, Tripathi C. Drug-induced SJS in Indian population. Niger J Clin Pract. 2017;20:978–983 https://doi.org/10.4103/njcp.njcp_122_16
- Jung Mi Byun, Dae Hoon Jeong, Young Nam Kim, Kyung Bok Lee, Moon Su Sung, Kitae Kim, Labial adhesions due to Stevens-Johnson syndrome. Dermatol Sin. 2015;33:239–240 https://doi.org/10.1016/j.dsi.2015.02.004
- Paipool W, Sriboonnark L. SJS/TEN in children: retrospective study. Asian Biomed. 2015;9:193–196
- Gamba C, Schroeder J, Citterio A, Cazzaniga S, Rivolta AL, Vighi G; . Surveillance of severe cutaneous drug reactions. Recenti Prog Med. 2014;105:379–384 https://doi.org/10.1701/1626.17670
- Diphoorn J, Cazzaniga S, Gamba C, Schroeder J, Citterio A, Rivolta AL, et al. Incidence and mortality of SJS/TEN in Italy. Pharmacoepidemiol Drug Saf. 2016;25:196–203 https://doi.org/10.1002/pds.3937
- Abe J, Umetsu R, Mataki K, Kato Y, Ueda N, Nakayama Y, et al. Analysis of SJS/TEN using Japanese database. J Pharm Health Care Sci. 2016;2:14 https://doi.org/10.1186/s40780-016-0048-5
- Ban GY, Ahn SJ, Yoo HS, Park HS, Ye YM. SJS/TEN associated with acetaminophen during infections. Immune Netw. 2016;16:256–260 https://doi.org/10.4110/in.2016.16.4.256
- Schotland P, Bojunga N, Zien A, Trame MN, Lesko LJ. Systems pharmacology approach to drug safety. Eur J Pharm Sci. 2016;94:84–92 https://doi.org/10.1016/j.ejps.2016.06.009
- Suwarsa O, Yuwita W, Dharmadji HP, Sutedja E.SJS/TEN study Indonesia (duplicate). Asia Pac Allergy. 2016;6:43–47 https://doi.org/10.5415/apallergy.2016.6.1.43
- Maggio N, Firer M, Zaid H, Bederovsky Y, Aboukaoud M, Gandelman-Marton R, et al. Causative drugs of SJS/TEN in Israel. J Clin Pharmacol. 2017 https://doi.org/10.1002/jcph.873
- Rahima S, Abdul Latheef EN, Pavithran K, et al. Clinical study of SJS/TEN in South India. Int J Res Dermatol. 2017;3:134–139
- Abdulah R, Suwandiman TF, Handayani N, et al. Incidence and economic impact of SJS/TEN. Ther Clin Risk Manag. 2017;13:919–925
- Alouani I, et al. Toxic epidermal necrolysis in child: case report. Reanimation. 2017;26:440–442
- Jongkhajornpong P, Lekhanot K, Pisuchpen P, Puangsricharern V, Prabhasawat P, Chantaren P, et al. HLA haplotype and SJS/TEN in Thailand. Br J Ophthalmol. 2018 https://doi.org/10.1136/bjophthalmol-2020-317315
- Sullivan KJ, Jeffres MN, Dellavalle RP, et al. Medication use in SJS/TEN patients. Antibiotics. 2018;7:1–7
- Wang YH, Chen CH, Tassaneeyakul W, Saito Y, Aihara M, Choon SE et al. Drug risk of SJS/TEN in Asians. Clin Pharmacol Ther. 2019;105:112–120 https://doi.org/10.1002/cpt.1071
- Rodríguez-Martín S, Martín-Merino E, Lerma V, Rodríguez-Miguel A, González O, González-Herrada C, et al. Active surveillance of severe cutaneous reactions. Pharmacoepidemiol Drug Saf. 2018 https://doi.org/10.1002/pds.4622
- Nguyen KD, Tran TN, Nguyen ML, Nguyen HA, Nguyen HA Jr, Vu DH, et al. Drug-induced SJS/TEN in Vietnam database. J Clin Pharm Ther. 2018 https://doi.org/10.1111/jcpt.12754
- Barry RJ, Zanetto U, Kolli S, Morjaria R. Toxic epidermal necrolysis: clinical case. BMJ Case Rep. 2018;2018:bcr2018225861 https://doi.org/10.1136/bcr-2018-225861
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
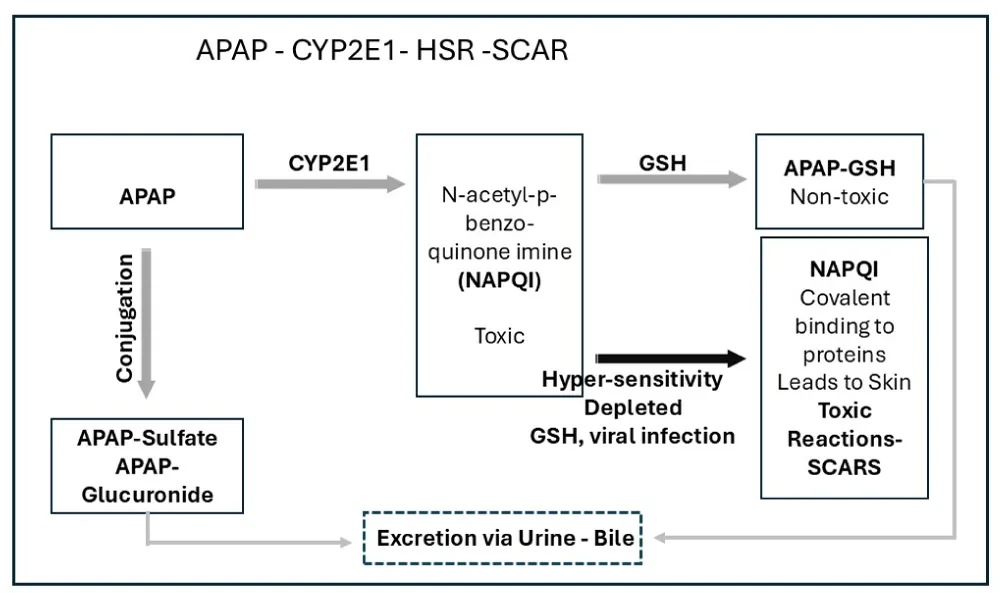


 Save to Mendeley
Save to Mendeley
